Transcatheter Arterial Embolisation of Acute Nonvariceal Upper Gastrointestinal Bleeding Refractory to Endoscopic Haemostasis
REVIEW ARTICLE CME
Transcatheter Arterial Embolisation of Acute Nonvariceal Upper Gastrointestinal Bleeding Refractory to Endoscopic Haemostasis
JH Kwon, JS Kim
Department of Radiology, Dongguk University Ilsan Hospital, Goyang-si, Republic of Korea
Correspondence: Prof JH Kwon, Department of Radiology, Dongguk University Ilsan Hospital, Goyang-si, Republic of Korea. Email: jhkwon17@naver.com
Submitted: 6 Aug 2018; Accepted: 21 Aug 2018.
Contributors: JHK contributed to the concept of study, acquisition and analysis of data, and wrote the manuscript. All authors had critical revision of the manuscript for important intellectual content. All authors had full access to the data, contributed to the study, approved the final
version for publication, and take responsibility for its accuracy and integrity.
Conflicts of Interest: The authors declare that there is no conflict of interest.
Funding/Support: The authors received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval: This study was approved by the institutional review board of Dongguk University Ilsan Hospital (Ref 2016-33). The
requirement for informed consent was waived for inclusion in this retrospective study.
Abstract
Upper gastrointestinal tract bleeding (UGIB) originates in the distal oesophagus, stomach, and duodenum (proximal to
the ligament of Treitz). The most common cause of nonvariceal UGIB is peptic ulcer disease, but it is associated with
many different diagnoses, including benign and malignant tumours, ischaemia, gastritis, arteriovenous malformations
such as Dieulafoy’s lesions, Mallory-Weiss tears, trauma, and iatrogenic causes. Endoscopic haemostasis remains
the initial treatment modality, but when endoscopic treatment fails to control bleeding, transcatheter arterial
embolisation is a safe, effective, and minimally invasive treatment compared with surgery. Advances in catheterbased
techniques and embolic agents have expanded the role of interventional radiology in UGIB treatment. This
article discusses the aetiologies of UGIB, methods of embolisation, characteristics of embolic agents, and evidence
in the literature regarding the technical and clinical outcomes of transcatheter arterial embolisation in patients with
acute nonvariceal UGIB.
Key Words: Angiography; Embolization, therapeutic; Gastrointestinal hemorrhage
中文摘要
經內鏡止血困難時以經導管動脈栓塞治理急性非靜脈曲張上消化道出血
JH Kwon, JS Kim
上消化道出血源於食道遠端、胃和十二指腸(Treitz靭帶近端十二指腸)。非曲張性上消化道出血的
最常見原因為消化性潰瘍,其他原因包括良性和惡性腫瘤、缺血、胃炎、動靜脈畸形(如Dieulafoy
病灶、賁門粘膜撕裂、創傷和醫源性原因)。內鏡止血仍然是初步治療方式。然而,當內窺鏡治療
無法控制出血時,與手術相比,經導管動脈栓塞術是一種安全有效且微創的治療方法。基於導管的技術和栓塞劑的進步加強了介入放射學在上消化道出血治療中的作用。本文討論上消化道出血的病
因、栓塞方法、栓塞劑的特性以及有關急性非靜脈曲張上消化道出血患者進行經導管動脈栓塞的技
術及臨床結果的文獻資料。
INTRODUCTION
Upper gastrointestinal (GI) bleeding (UGIB) is defined
as bleeding originating in the distal oesophagus,
stomach, and duodenum (proximal to the ligament of
Treitz). Upper GI bleeding is 5 times more common
than lower GI bleeding. This distinction is important,
as localisation of the bleeding source determines the
therapeutic approach.[1] [2] [3] The most common cause of
nonvariceal UGIB is peptic ulcer disease. Other less
common causes include benign and malignant tumours,
ischaemia, gastritis, Mallory-Weiss tears, trauma,
iatrogenic factors, and arteriovenous malformations such
as Dieulafoy’s lesions.[4] [5]
Effective treatment requires an accurate diagnosis
including the location and aetiology. Unlike the case
with lower GI bleeding, most patients have undergone
endoscopic examination and treatment before being
referred for interventional radiology.[6] However,
endoscopic evaluation and potential treatment
are sometimes not technically feasible because of
oesophageal strictures, altered anatomy from prior
surgery, or profuse bleeding.[3] Of the small group of
patients whose bleeding fails to respond to endoscopic
therapy, some are treated surgically, but the majority
have been increasingly referred for transcatheter arterial
embolisation (TAE).[6] Surgical treatment is typically
reserved for patients whose bleeding has failed to
respond to all previous treatments. In such cases,
conservative surgical techniques that focus on the source
of the bleeding are usually preferred to conventional
surgery.[6] The development of newer catheter techniques
and embolic devices over the last three decades has made
percutaneous TAE the standard management technique
for UGIB in patients with unfeasible or failed endoscopic
management of bleeding.[3] [6] The purpose of this review
is to summarise the data on the aetiologies, indications,
techniques, and outcomes of TAE for acute nonvariceal
UGIB
CAUSES OF ACUTE NONVARICEAL
UPPER GASTROINTESTINAL BLEEDING
Erosive processes, such as peptic ulcers, oesophagitis,
gastritis, duodenitis, and Zollinger-Ellison syndrome, account for 70% of UGIB cases.[6] [7] [8] The incidence of
bleeding from these complications among elderly patients
has increased because of improved life expectancy and
the expanded use of nonsteroidal anti-inflammatory
drugs.[8] Cyclooxygenase-2 inhibitors, which are used for
both their anti-inflammatory and analgesic properties, are
associated with an overall increased use of nonsteroidal
anti-inflammatory medications and subsequent UGIB.[9]
GI bleeding can be accelerated if a patient is in an
endogenous coagulopathic state or on anticoagulation
therapy.[6]
Oesophageal varices are the second most common cause
of UGIB, accounting for 5% to 14% of cases.[10] These
patients present with portal hypertension and may have
underlying alcoholic liver disease or hepatitis-induced
cirrhosis.[8] However, the management of oesophageal
varices and portal hypertension is generally distinct from
the conditions described herein and is excluded from this
review.
A Mallory-Weiss tear is a mucosal laceration at the
gastroesophageal junction or in the cardia region of the
stomach.[11] These lesions are associated with repeated
retching or vomiting and are another important cause
of nonvariceal UGIB. It is estimated that 5% to 15% of
all cases of acute UGIB are secondary to Mallory-Weiss
tears.[11] [12]
Dieulafoy’s lesion is characterised by a large aberrant
arteriole in the mucosa that has the potential to rupture
spontaneously.[13] It is a calibre-persistent gastric artery or
aneurysm and may be considered as an aberrant large
arteriole. These arterioles, which are approximately
1 mm in diameter, can reach the submucosa and invade
through the mucosal surface.[13] [14] Dieulafoy’s lesion is
relatively rare and is thought to cause fewer than 5% of
all cases of GIB.[7]
Both malignant and benign neoplasms involving or
originating from the upper GI tract may cause 2% to 5%
of cases of UGIB. Although only a small proportion of
UGIB cases are of neoplastic aetiology, UGIB may be
the only presenting symptom of a neoplasm and therefore
should be included in the differential diagnosis.[6] [7]
Vascular ectasias, also referred to as angiomas,
arteriovenous malformations, and angiodysplasia, are
another source of UGIB. Angiodysplasia or arteriovenous
malformations are vascular anomalies characterised by
an abnormal tangle of vessels with a prominent draining
vein or veins that typically exhibit early and prolonged
opacification.[7] These vascular anomalies are seen more
frequently in lower GI bleeding but also occur rarely in
UGIB.[15]
Other rare causes of nonvariceal UGIB should also be
considered in any differential diagnosis. Aortoenteric
fistula is a rare but potentially catastrophic cause of
UGIB. Communication between the aorta and bowel
can develop from pathologic processes at either site.
The most common causes of aortoenteric fistula are
aortic aneurysm, infectious aortitis due to syphilis or
tuberculosis, and surgical repair of an abdominal aortic
aneurysm; surgical intervention is necessary in almost
all such cases, as mortality without surgical intervention
has been reported to be as high as 100%.[6] [7] [16] Haemobilia,
a pathologic process of the liver or recent hepatobiliary
tree instrumentation, is another rare cause of UGIB that
should be considered in trauma and surgery settings.[6] [8] [17]
Haemosuccus pancreaticus refers to bleeding from the
pancreatic duct and should be considered in patients
with chronic pancreatitis or pseudocysts. Bleeding in
these patients can be secondary to a pseudoaneurysm
in peripancreatic blood vessels as a complication of
pancreatic pseudocysts.[18] Finally, iatrogenic injuries
secondary to biopsies or endoscopic procedures, such as
the placement of percutaneous endoscopic gastrostomy
tubes, are also rare but potential causes of nonvariceal
UGIB.[4] [18]
INDICATIONS FOR ANGIOGRAPHY
Since TAE was introduced as an alternative to surgery
for control of UGIB, embolotherapy has become a useful
diagnostic and therapeutic tool in selected patients.[19]
The typical candidate patient presents with the following:
(1) massive bleeding (transfusion requirement of at
least 4 U blood in 24 h) or haemodynamic instability
(hypotension with systolic pressure <100 mmHg and
heart rate >100 beats/min or clinical shock secondary
to blood loss), (2) bleeding that has failed to respond
to conservative medical therapy, including volume
replacement, antacids, H2 receptor blocking agents, or
proton pump inhibitors, and (3) bleeding that has failed
to respond to at least one, and sometimes two, attempts
at endoscopic control.[6] [20] At that point, low-risk patients
are offered the option of surgical intervention, whereas high-risk patients are offered TAE. Finally, TAE can
be used after open surgical intervention has failed
and bleeding has recurred, even after percutaneous
embolotherapy.[6] [21] It is important to perform angiography
while the patient is bleeding, rather than waiting until the
patient is hypotensive or unstable.[22]
CONTRA-INDICATIONS TO
ANGIOGRAPHY
There are no absolute contra-indications to angiography
and embolisation, as these can be lifesaving procedures.
Thus, contra-indications to TAE in patients with UGIB
are only relative. These include renal insufficiency,
contrast allergy, and uncorrectable coagulopathy. For
patients with severe reactions to iodinated contrast
media, alternative contrast agents such as carbon dioxide
can be used. There is an increased risk of gastric or
duodenal infarction following TAE in patients who
have received previous extensive upper GI surgery or
radiotherapy.[6] [21] [23] [24]
DIAGNOSTIC EVALUATION
Successful treatment of UGIB depends on accurate initial
assessment of the patient’s condition and on prompt
localisation of the bleeding site. Upper endoscopy is the
initial diagnostic modality of choice for acute UGIB.10
It is highly accurate at determining a diagnosis and is
very useful for formulating treatment strategies and
therapeutic interventions.[25] The sensitivity and specificity
of endoscopy have been reported as 92% to 98% and 33%
to 100%, respectively.[8] Endoscopy can also help with
planning the timing of and approach to angiography.[24]
For example, the ability to find the source of bleeding
helps to guide the choice of which artery to cannulate
first during angiography. Endoscopic information, such
as excluding oesophageal bleeding sources, is valuable
to angiographers.[24] Nevertheless, endoscopy may miss
up to 10% of lesions if they are within the reach of the
endoscope and 18% if they are out of reach.[26]
Technical improvements in multidetector computed
tomography (MDCT) technology, such as higher
temporal resolution, have expanded the application of
CT angiography for evaluating patients with vascular
disease, including acute GI bleeding.[27] Temporally
resolved MDCT angiography allows the identification
of active extravasation of contrast material and accurate
identification of the source of haemorrhage.[27] MDCT
angiography is a rapid and easy-to-perform diagnostic
method for fast and accurate detection and localisation
of acute GI bleeding in the emergency setting.[27] [28] The sensitivity of MDCT angiography for diagnosing the
source of GI bleeding has been reported as 91% to 92%
and 45% to 47% for active and obscure GI bleeding,
respectively.[28] [29]
Scintigraphy and enteroscopy using technetium
99m–labelled red blood cells or technetium 99m–sulfur
colloid, as well as video capsule endoscopy, are not
universally accessible in the emergency setting.[27]
Scintigraphy imaging has advantages including its
non-invasiveness and its ability to image for long periods
of time. Its disadvantages include difficulty determining
the precise location of the site of bleeding. Generally,
scintigraphy plays a minor role in UGIB diagnosis
because upper endoscopy has a high accuracy rate for
detection of UGIB.[8]
ANGIOGRAPHY AND
EMBOLISATION
Angiography
By the time a patient with UGIB reaches the
interventional room, fluid resuscitation and correction of
coagulopathy should have been initiated. Blood products
such as packed red blood cells, fresh frozen plasma, or
platelets may also be given intraoperatively.[4] [23] Bladder
catheter insertion is desirable.[23] During the procedure,
blood pressure, heart rate, oxygen saturation, and
electrocardiogram are monitored. It is desirable to have
anaesthesiologist and intensive care physician support,
particularly with patients who are haemodynamically
unstable.[23] It is desirable to correct any coagulopathy prior to embolisation because achieving haemostasis
depends on both technically successful embolisation
and the patient’s ability to form a clot. However,
angiography with embolisation should be promptly
performed in patients who are acutely bleeding, followed
by coagulopathy correction.[6] Endoscopic diagnosis
and therapy can render angiography unnecessary or
inform the timing or planning of the angiographic
approach. Even negative endoscopic information,
including exclusion of oesophageal bleeding, is
particularly valuable to angiographers.[24] Increased time
to angiography is a predictor of early re-bleeding after
embolisation; therefore, angiography and embolisation
should be performed soon after the onset of bleeding.[30] [31]
Angiography is used most often when endoscopy fails
to detect UGIB. The main goals of angiography are
to (1) accurately confirm the diagnosis of bleeding,
(2) accurately localise the site of bleeding, and
(3) provide transcatheter therapy as required.[8]
Angiography is very specific (100% specificity) and
highly sensitive (90% sensitivity) for UGIB.[8] It was
used successfully to detect bleeding in a canine model
when the rate of bleeding was >0.5 mL/min, and in-vitro
studies have suggested that digital subtraction
angiography is five- to nine-fold more sensitive than
film-screen angiography for detecting haemorrhage.[8] [32]
One definitive sign of GI bleeding in angiography is
extravasation of contrast medium into the bowel lumen
(Figure 1).[8] [22] Indirect signs of GI bleeding include
aneurysm/pseudoaneurysm, arteriovenous fistula, vessel irregularity, vessel cut-off, hyperaemia, or neovascularity
on imaging (Figure 2).[8] [22] Interventionists should
recognise digital subtraction artefacts in angiograms that
mimic contrast extravasation, such as bowel peristalsis
and adrenal gland blush.[7]
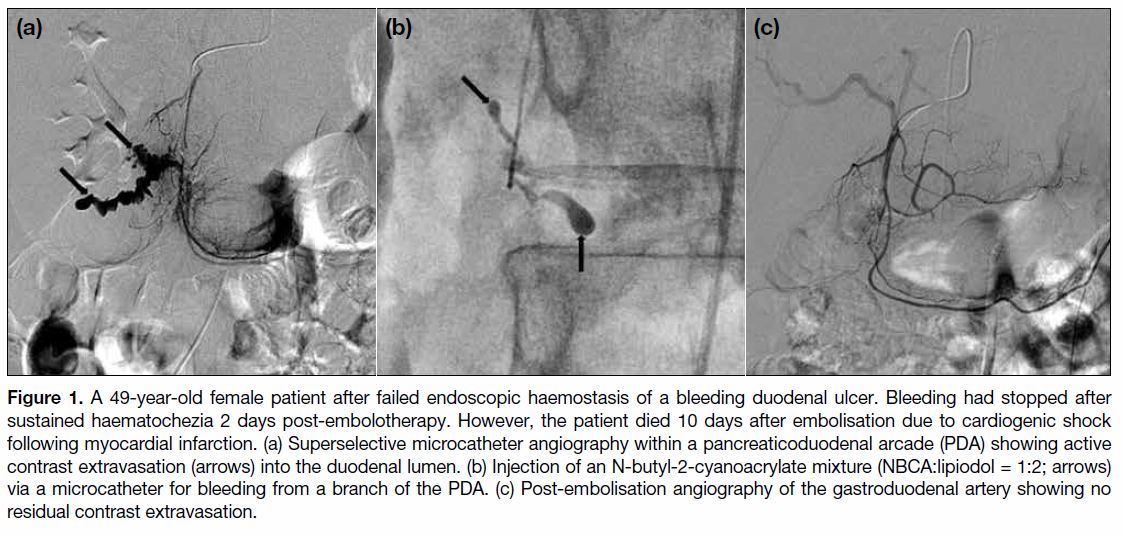
Figure 1. A 49-year-old female patient after failed endoscopic haemostasis of a bleeding duodenal ulcer. Bleeding had stopped after
sustained haematochezia 2 days post-embolotherapy. However, the patient died 10 days after embolisation due to cardiogenic shock
following myocardial infarction. (a) Superselective microcatheter angiography within a pancreaticoduodenal arcade (PDA) showing active
contrast extravasation (arrows) into the duodenal lumen. (b) Injection of an N-butyl-2-cyanoacrylate mixture (NBCA:lipiodol = 1:2; arrows)
via a microcatheter for bleeding from a branch of the PDA. (c) Post-embolisation angiography of the gastroduodenal artery showing no
residual contrast extravasation.
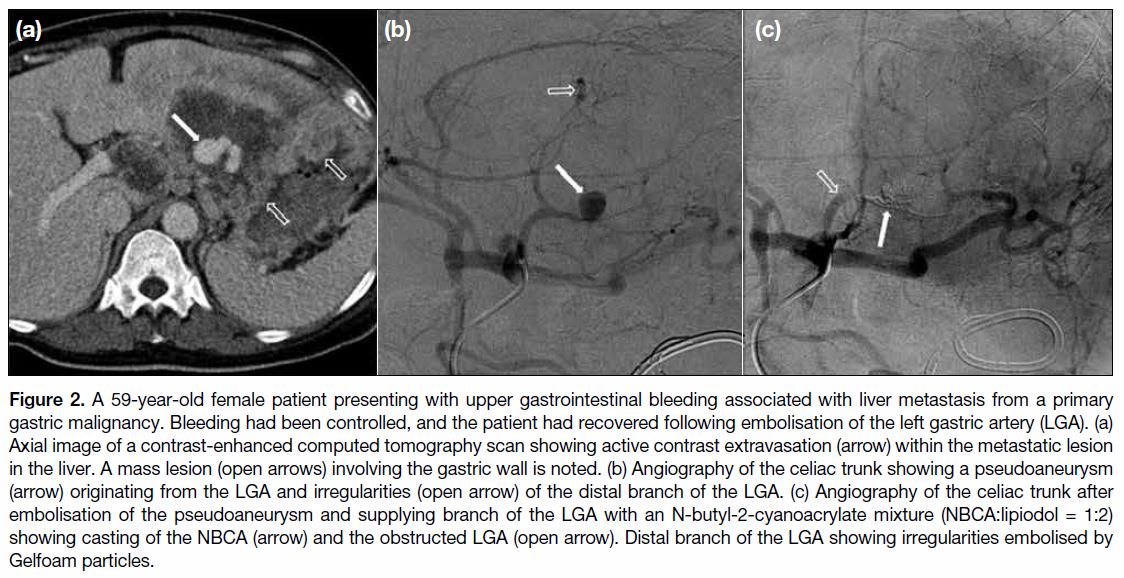
Figure 2. A 59-year-old female patient presenting with upper gastrointestinal bleeding associated with liver metastasis from a primary
gastric malignancy. Bleeding had been controlled, and the patient had recovered following embolisation of the left gastric artery (LGA). (a)
Axial image of a contrast-enhanced computed tomography scan showing active contrast extravasation (arrow) within the metastatic lesion
in the liver. A mass lesion (open arrows) involving the gastric wall is noted. (b) Angiography of the celiac trunk showing a pseudoaneurysm
(arrow) originating from the LGA and irregularities (open arrow) of the distal branch of the LGA. (c) Angiography of the celiac trunk after
embolisation of the pseudoaneurysm and supplying branch of the LGA with an N-butyl-2-cyanoacrylate mixture (NBCA:lipiodol = 1:2)
showing casting of the NBCA (arrow) and the obstructed LGA (open arrow). Distal branch of the LGA showing irregularities embolised by
Gelfoam particles.
In the majority of cases, transfemoral arterial access
is used for initial catheter introduction. Abdominal
aortography is generally unnecessary, as it requires
a large bolus of contrast medium and has a low
likelihood of identifying active bleeding.[7] Glucagon
and scopolamine butylbromide may be given before
the procedure to decrease bowel motility and motion
artefacts during digital subtraction angiography.[21]
The first vessels selected for angiography should be
determined following an accurate history and clinical
exam and guided by CT, scintigraphic images, or
endoscopic findings.[21] In patients with UGIB, the celiac
and superior mesenteric arteries are the main target
vessels. If routine angiography of the celiac artery or
superior mesenteric artery does not identify a bleeding
focus, superselective catheterisation of the smaller
branches should be performed. Angiographic images
should be obtained until the venous phase has cleared
out to distinguish contrast extravasation from venous
opacification.[21] If the bleeding site is still not identified
by angiography, the bleeding site may be small and
therefore difficult to visualise, or the offending bleeding
vessel may not have been catheterised.[8] When two bleeding sources are suspected, both arterial sources
need to be embolised to assure that all inflow ceases.
This is typically seen when an ulcer invades into the
gastroduodenal artery.[6] [24]
The use of carbon dioxide as a contrast medium can
improve angiography’s sensitivity for small bleeds.[6] [22]
Endoscopic clips placed around the bleeding area to
achieve pre-embolisation endoscopic haemostasis can
help to accurately localise the site of bleeding. If no
extravasation is seen despite contrast injection, then
the branches terminating at the clip are superselected
and embolised using microcatheter techniques.[33] [34]
Provocative angiography following infusion of tolazoline
(a vasodilator), heparin, or thrombolytics such as tissue
plasminogen activator can encourage bleeding. However,
not enough data are available to generate definitive
guidelines for performing provocative mesenteric
angiography or pharmacoangiography.[24] Those
techniques are mainly used to induce lower GI bleeding,
which is more challenging to localise than UGIB. Most
UGIB cases require endoscopy to identify, localise, and
treat the source of bleeding. Several prior studies have
shown that in the absence of contrast extravasation,
empiric embolisation based on endoscopic findings can
be performed safely and successfully.[24][35] The absence of
contrast extravasation in angiography is less problematic
in the upper GI tract and does not prevent embolisation
of the artery supplying the bleeding site.[24]
Embolisation
The goal of TAE is selective reduction of blood flow
at the bleeding source while maintaining sufficient
collateral flow for intestinal viability.[7] [24] Despite the
fact that vessel embolisation carries some risk of bowel
ischaemia or infarction, selective catheterisation and
precise embolisation minimise these complications.[7]
In addition, the GI tract has a rich collateral blood
supply with extensive vascular arcades that allow
safe embolisation.[7] Contemporary TAE techniques
generally involve the placement of diagnostic 4- to
5-French catheters into the main trunk of the feeding
artery, followed by coaxial introduction of a 3-French
or smaller microcatheter. The microcatheter can then be
used to superselectively catheterise the target vessel as
close as possible to the source of bleeding. It therefore
must have an internal diameter that allows introduction
of the chosen embolic agent.[3] [7]
Embolisation can be classified as localised, proximal,
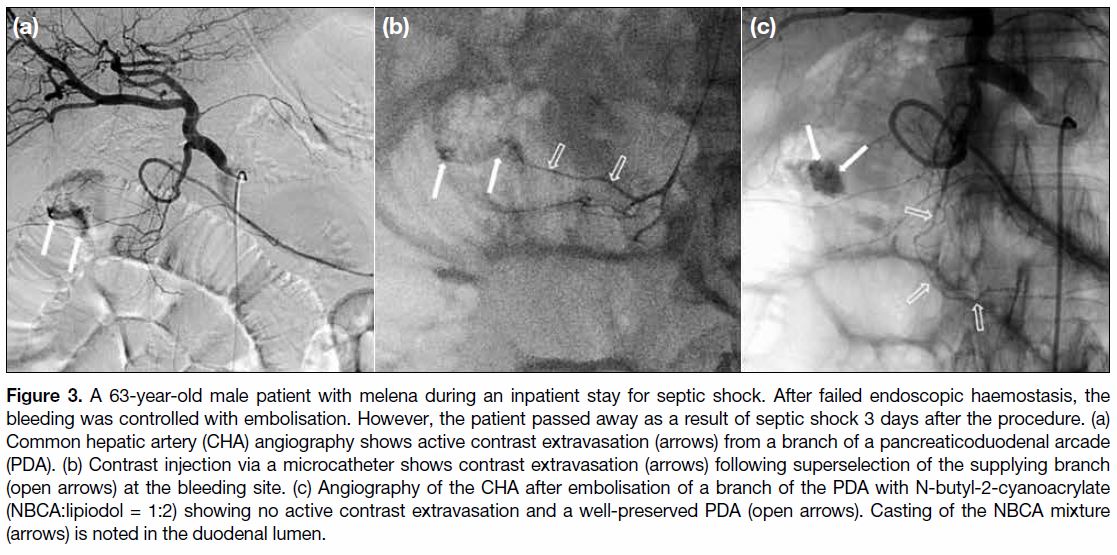
or segmental.[21] In localised embolisation, superselective
embolisation at the site of bleeding is performed without
embolising other non-target arteries (Figure 3). Proximal
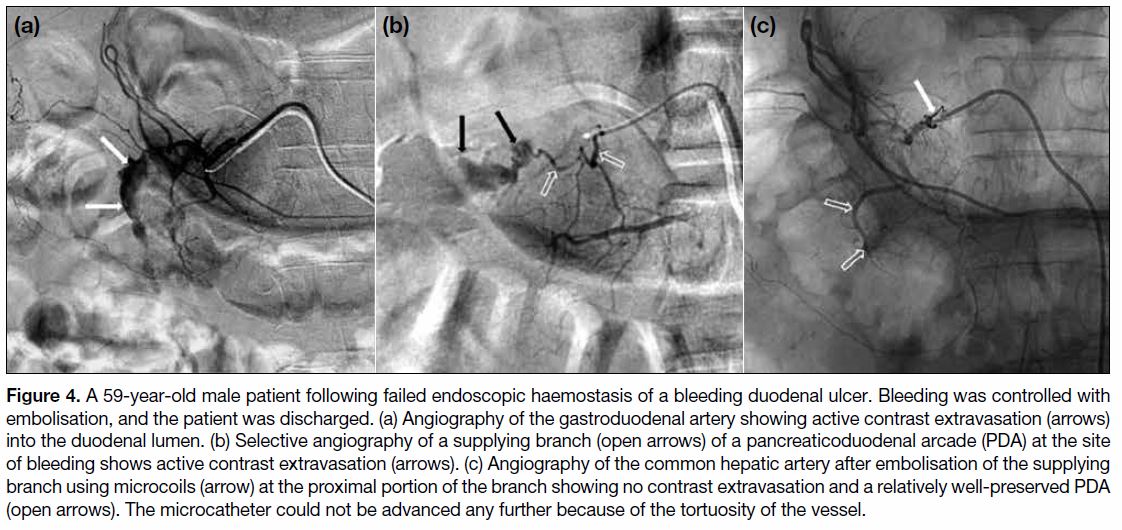
embolisation is required when a microcatheter cannot
enter the bleeding vessel and embolisation must be
performed in its parent artery, leaving the actual bleeding
site without embolisation (Figure 4). With proximal
embolisation, recanalisation of the bleeding artery
can occur as a result of distal back flow. Segmental
embolisation is the inclusion of an adjacent branch artery or arteries in addition to the segment responsible
for the bleeding (Figure 5). With excessive segmental
embolisation, ischaemic complications of the involved
bowel can occur.
Figure 3. A 63-year-old male patient with melena during an inpatient stay for septic shock. After failed endoscopic haemostasis, the
bleeding was controlled with embolisation. However, the patient passed away as a result of septic shock 3 days after the procedure. (a)
Common hepatic artery (CHA) angiography shows active contrast extravasation (arrows) from a branch of a pancreaticoduodenal arcade
(PDA). (b) Contrast injection via a microcatheter shows contrast extravasation (arrows) following superselection of the supplying branch
(open arrows) at the bleeding site. (c) Angiography of the CHA after embolisation of a branch of the PDA with N-butyl-2-cyanoacrylate
(NBCA:lipiodol = 1:2) showing no active contrast extravasation and a well-preserved PDA (open arrows). Casting of the NBCA mixture
(arrows) is noted in the duodenal lumen.
Figure 4. A 59-year-old male patient following failed endoscopic haemostasis of a bleeding duodenal ulcer. Bleeding was controlled with
embolisation, and the patient was discharged. (a) Angiography of the gastroduodenal artery showing active contrast extravasation (arrows)
into the duodenal lumen. (b) Selective angiography of a supplying branch (open arrows) of a pancreaticoduodenal arcade (PDA) at the site
of bleeding shows active contrast extravasation (arrows). (c) Angiography of the common hepatic artery after embolisation of the supplying
branch using microcoils (arrow) at the proximal portion of the branch showing no contrast extravasation and a relatively well-preserved PDA
(open arrows). The microcatheter could not be advanced any further because of the tortuosity of the vessel.
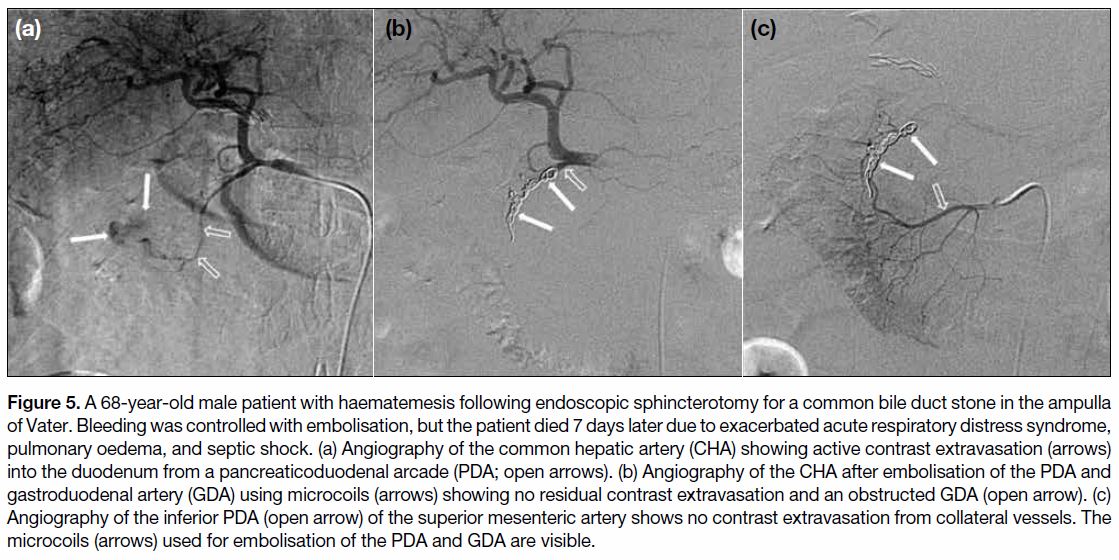
Figure 5. A 68-year-old male patient with haematemesis following endoscopic sphincterotomy for a common bile duct stone in the ampulla
of Vater. Bleeding was controlled with embolisation, but the patient died 7 days later due to exacerbated acute respiratory distress syndrome,
pulmonary oedema, and septic shock. (a) Angiography of the common hepatic artery (CHA) showing active contrast extravasation (arrows)
into the duodenum from a pancreaticoduodenal arcade (PDA; open arrows). (b) Angiography of the CHA after embolisation of the PDA and
gastroduodenal artery (GDA) using microcoils (arrows) showing no residual contrast extravasation and an obstructed GDA (open arrow). (c)
Angiography of the inferior PDA (open arrow) of the superior mesenteric artery shows no contrast extravasation from collateral vessels. The
microcoils (arrows) used for embolisation of the PDA and GDA are visible.
Many embolic agents have been used successfully,
including coils, microcoils, gelatine sponge particular
materials, polyvinyl alcohols (PVA), and trisacryl
gelatin particles. Liquid embolic materials such as
N-butyl-2-cyanoacrylate (NBCA) or ethylene-vinyl
copolymer (Onyx; Microtherapeutics, Inc., Irvine [CA],
United States) are less frequently used.[6] [24] [30] [36] [37] [38] There
is still debate about which embolic material is ideal.
Embolotherapy needs to be performed quickly, feasibly,
and effectively in emergency cases. Each embolic
material has particular advantages and disadvantages, and
the choice of embolic material is usually made based on
bleeding location, aetiology, and operator preference.[3] [4] [24]
Coils are used for the embolisation of macroscopic
vessels and must be used cautiously and precisely as
they permanently occlude the bleeding vessel (Figure 6).
Embolisation of UGIB using coils acts to decrease
arterial pressure in the involved vascular bed, resulting
in a more effective clotting cascade and reduction of
bleeding.[4] Particulate agents can be used together with
coils to embolise distal vessels. The main advantage of
coils is the lower risk of ischaemic complications, as
they are used for focal occlusion of macroscopic arteries,
whereas the more distal microvasculature is maintained through collateral circulation. Coils can be placed
more precisely than can liquid agents, which are more
difficult to control.[1] The main disadvantage of coils is
that they occlude the artery permanently and preclude
re-intervention if further embolotherapy is required.[4]
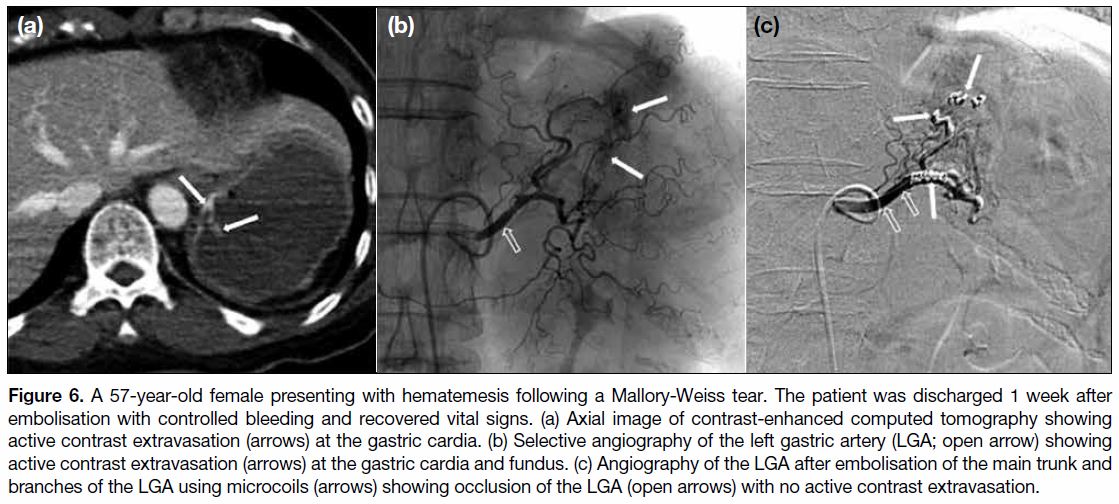
Figure 6. 6. A 57-year-old female presenting with hematemesis following a Mallory-Weiss tear. The patient was discharged 1 week after
embolisation with controlled bleeding and recovered vital signs. (a) Axial image of contrast-enhanced computed tomography showing
active contrast extravasation (arrows) at the gastric cardia. (b) Selective angiography of the left gastric artery (LGA; open arrow) showing
active contrast extravasation (arrows) at the gastric cardia and fundus. (c) Angiography of the LGA after embolisation of the main trunk and
branches of the LGA using microcoils (arrows) showing occlusion of the LGA (open arrows) with no active contrast extravasation.
Gelatine sponges are one of the oldest flow-directed
embolic agents (Figure 7). Reduction in pressure
proximal to the bleeding site using sponges can be sufficient to stop the bleeding.[4] Gelatine sponges are
biodegradable and re-absorbable, allowing recanalisation
of embolised vessels 2 to 6 weeks postoperatively.[3]
The advantages of Gelfoam particles are that they are
readily available, inexpensive, and unlikely to cause
ischaemia, allowing future access to embolised vessels
after re-absorption.[3] The disadvantages include the
time-consuming preparation of appropriately sized
particles and the unpredictable pace of recanalisation.[1] [4]
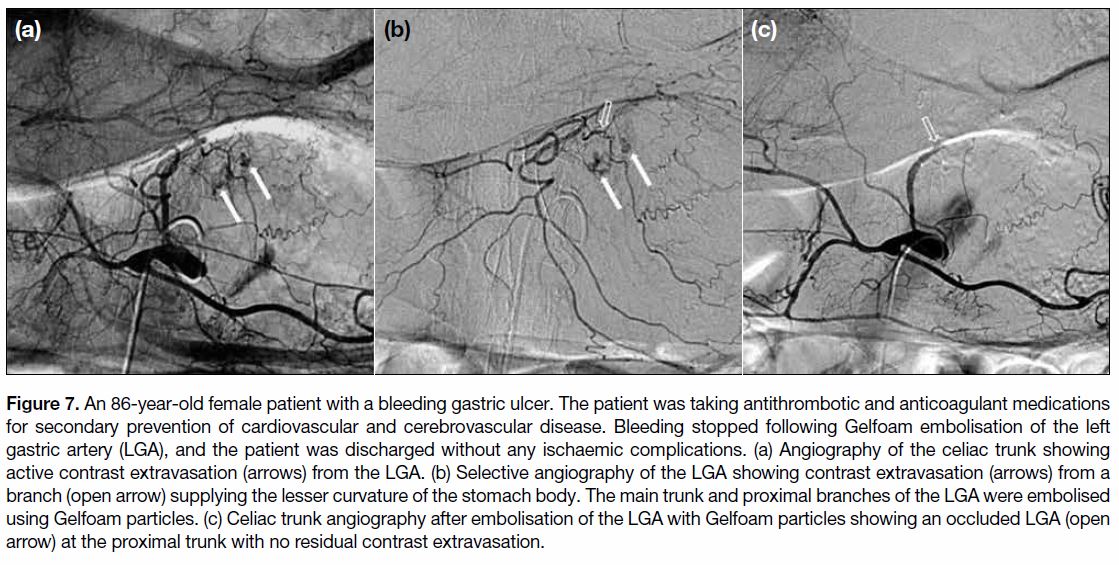
Figure 7. 7. An 86-year-old female patient with a bleeding gastric ulcer. The patient was taking antithrombotic and anticoagulant medications
for secondary prevention of cardiovascular and cerebrovascular disease. Bleeding stopped following Gelfoam embolisation of the left
gastric artery (LGA), and the patient was discharged without any ischaemic complications. (a) Angiography of the celiac trunk showing
active contrast extravasation (arrows) from the LGA. (b) Selective angiography of the LGA showing contrast extravasation (arrows) from a
branch (open arrow) supplying the lesser curvature of the stomach body. The main trunk and proximal branches of the LGA were embolised
using Gelfoam particles. (c) Celiac trunk angiography after embolisation of the LGA with Gelfoam particles showing an occluded LGA (open
arrow) at the proximal trunk with no residual contrast extravasation.
Lang et al[39] reported that a higher rate of re-bleeding
in UGIB was noted when gelatine sponges were used
alone than in combination with other embolic agents.
Encarnacion et al[40] reported a low success rate in their
study, which included patients embolised with gelatine
sponges alone, suggesting that the use of gelatine sponges
as the only embolic agent assures only short-term
results and should be avoided.[24] Recently, premade and
precisely calibrated gelatine sponge particles (Caligel;
Hangzhou Alicon Pharm Sci & Tec Co., Zhejiang, China) have become available, and these do not require
time-consuming preparation and are more predictable in
size. Large pledgets of gelatin sponges created by cutting
individual sponge pieces can also be used as torpedoes
to occlude larger vessels.[4] In addition, gelatine powder is
also commercially available as an embolic agent, but it
results in higher incidence of ischaemic complications.[4]
Particles of PVA vary in size and are supplied as either
irregular or spherical particles suspended in sterile saline.[3] They are inexpensive and easy to use but have
a similar precision to that of coils (Figure 8). The
particles are mixed with contrast medium immediately
before injection. Their irregular surface may cause
clumping or aggregation of particles, resulting in
catheter obstruction or large vessel occlusion.[41] The
use of PVA particles is reserved for areas where
permanent embolisation down to the arteriolar bed is
required in the bleeding lesion. These agents have been
used successfully to treat GI bleeding. Usually, larger
particles (>500 μm) are used to decrease the risk of
ischaemic complications.[3] [4] [24]
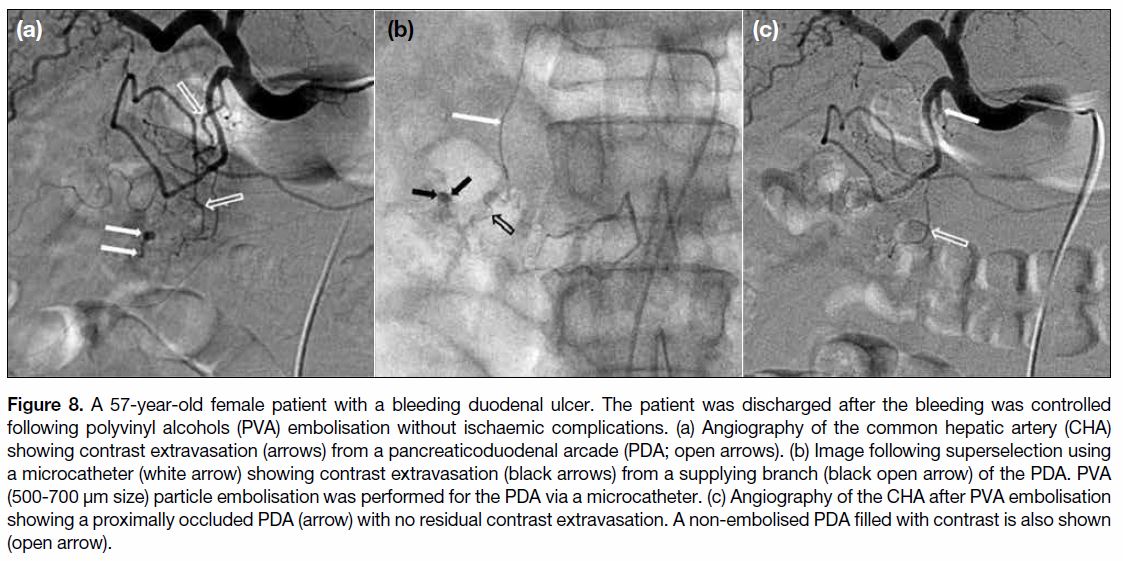
Figure 8. A 57-year-old female patient with a bleeding duodenal ulcer. The patient was discharged after the bleeding was controlled
following polyvinyl alcohols (PVA) embolisation without ischaemic complications. (a) Angiography of the common hepatic artery (CHA)
showing contrast extravasation (arrows) from a pancreaticoduodenal arcade (PDA; open arrows). (b) Image following superselection using
a microcatheter (white arrow) showing contrast extravasation (black arrows) from a supplying branch (black open arrow) of the PDA. PVA
(500-700 μm size) particle embolisation was performed for the PDA via a microcatheter. (c) Angiography of the CHA after PVA embolisation
showing a proximally occluded PDA (arrow) with no residual contrast extravasation. A non-embolised PDA filled with contrast is also shown
(open arrow).
The main liquid embolic agents include dehydrated
ethanol and NBCA. Dehydrated ethanol, which is
cytotoxic, is generally used to induce necrosis throughout
the entire vascular bed. The use of dehydrated ethanol is
reserved for treatment of arteriovenous malformations.[4]
NBCA is mixed with an oily contrast medium (Lipiodol;
Guerbet, Aulnay-sous-Bois, France) to make it visible
and viscous, and it polymerises upon contact with
circulating ions. Depending on the volume and solubility
of the dilutional agent, the material can be designed
to solidify rapidly or more gradually.[4] Embolisation
by NBCA is quick and permanent, with the degree of
embolisation controlled by titrating the viscosity of
the mixture. Embolisation with NBCA can be useful
in haemodynamically unstable patients and in cases of
underlying coagulopathy.[24] Recently, good results using NBCA to control GI bleeding have been reported.[42] [43] [44] [45]
Embolisation using NBCA is effective in cases with
involvement of fine and tortuous vessels, which are
difficult to embolise using coils. The embolisation of
both feeding arteries and collateral vessels is often too
difficult and time-consuming to catheterise selectively
(Figure 9).[45] The main disadvantage of NBCA is that
reflux of even small amounts of NBCA can result in
non-target embolisation and complete occlusion of a
non-target vessel.[4] The use of NBCA requires training
and considerable experience to prevent bowel ischaemia
or infarction caused by reflux of NBCA into non-target
vessels.[24]
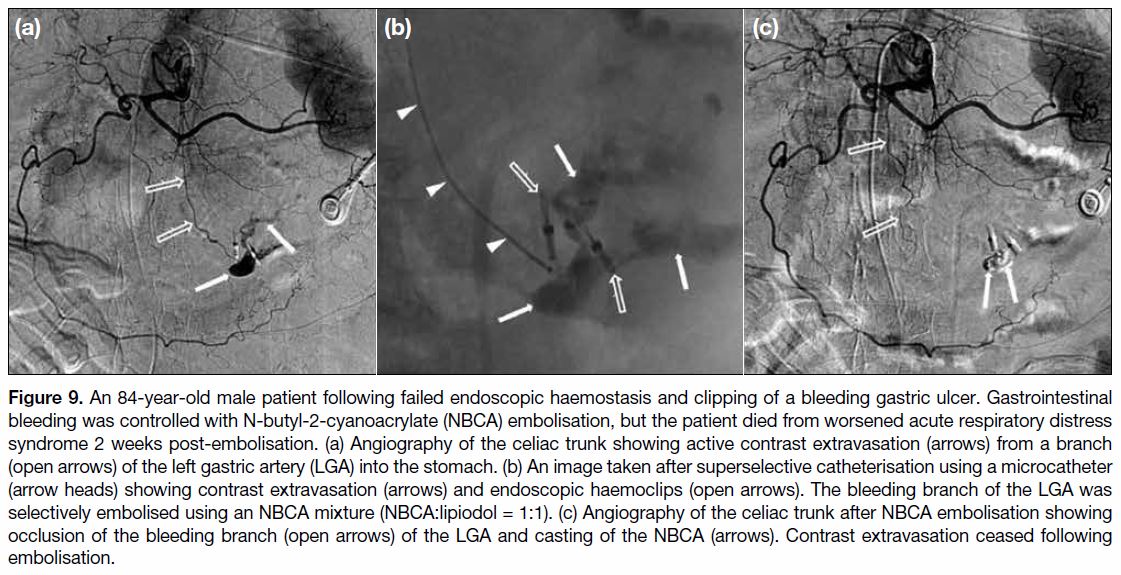
Figure 9. An 84-year-old male patient following failed endoscopic haemostasis and clipping of a bleeding gastric ulcer. Gastrointestinal
bleeding was controlled with N-butyl-2-cyanoacrylate (NBCA) embolisation, but the patient died from worsened acute respiratory distress
syndrome 2 weeks post-embolisation. (a) Angiography of the celiac trunk showing active contrast extravasation (arrows) from a branch
(open arrows) of the left gastric artery (LGA) into the stomach. (b) An image taken after superselective catheterisation using a microcatheter
(arrow heads) showing contrast extravasation (arrows) and endoscopic haemoclips (open arrows). The bleeding branch of the LGA was
selectively embolised using an NBCA mixture (NBCA:lipiodol = 1:1). (c) Angiography of the celiac trunk after NBCA embolisation showing
occlusion of the bleeding branch (open arrows) of the LGA and casting of the NBCA (arrows). Contrast extravasation ceased following
embolisation.
Because of the often intermittent nature of GI bleeding,
the incidence of normal angiographic findings in patients
with acute UGIB and lower GI bleeding has been
observed as 52%.[46] If the site of bleeding is not identified,
empiric embolisation is an alternative that can be guided
by endoscopic evidence.[22] Empiric embolisation for
UGIB is sometimes encouraged because GI bleeding is
often intermittent and accompanied by high re-bleeding
and mortality rates if left untreated. Angiographic
confirmation of a bleeding site is not a prerequisite for
TAE in UGIB. Several studies have shown no differences
in clinical outcomes between patients with negative and
positive angiographic findings after TAE.[47] [48] [49] In the case
of gastric bleeding that is visible on endoscopy, the left
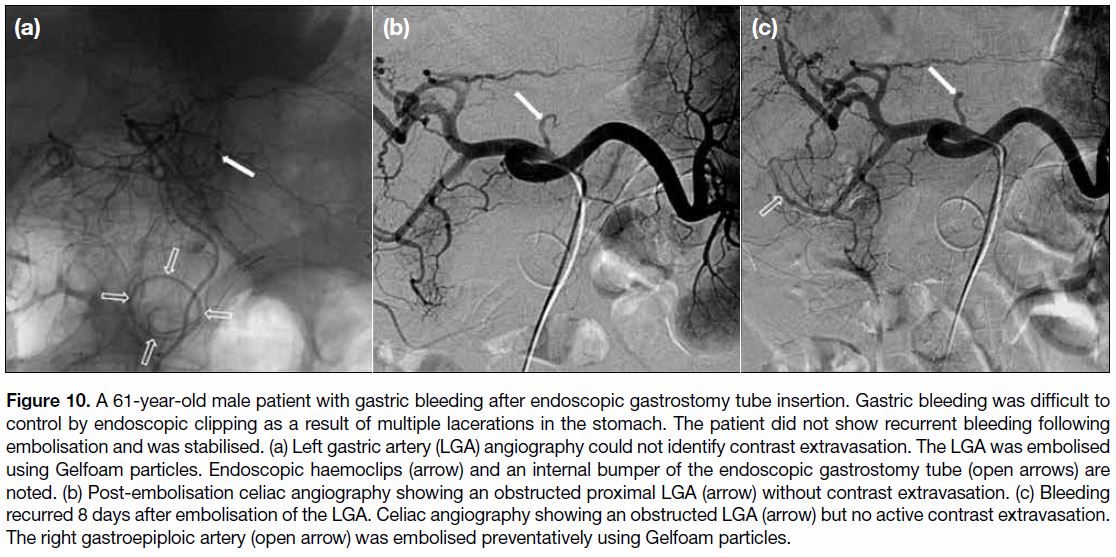
gastric artery is often embolised (Figure 10).
Figure 10. A 61-year-old male patient with gastric bleeding after endoscopic gastrostomy tube insertion. Gastric bleeding was difficult to
control by endoscopic clipping as a result of multiple lacerations in the stomach. The patient did not show recurrent bleeding following
embolisation and was stabilised. (a) Left gastric artery (LGA) angiography could not identify contrast extravasation. The LGA was embolised
using Gelfoam particles. Endoscopic haemoclips (arrow) and an internal bumper of the endoscopic gastrostomy tube (open arrows) are
noted. (b) Post-embolisation celiac angiography showing an obstructed proximal LGA (arrow) without contrast extravasation. (c) Bleeding
recurred 8 days after embolisation of the LGA. Celiac angiography showing an obstructed LGA (arrow) but no active contrast extravasation.
The right gastroepiploic artery (open arrow) was embolised preventatively using Gelfoam particles.
OUTCOMES
In a review by Loffroy et al[6] of 15 studies (involving
819 patients, mean age 65 years), the technical success
rate of endovascular embolisation of intractable
nonvariceal UGIB was 93%. Endoscopic haemostasis
had failed in 99% of the patients. The causes for the
failure of TAE to control UGIB included difficult
vascular anatomy, arterial dissection, vasospasm, misinterpretation of angiography findings, multiple
bleeding sites, and haemorrhage from malignant
processes. The majority of the patients in that series who
underwent TAE had significant co-morbidities and high
levels of risk associated with operative intervention.
Active contrast extravasation was observed at the time
of TAE in only 54% of patients. Consequently, 46% of
the patients underwent embolisation, guided by either endoscopy findings or clip placement around the area
of the bleeding vessel.[6] The clinical success rate was
67% in patients who underwent technically successful
embolisation. Possible predictors of re-bleeding were
the presence of multiple or large duodenal ulcers, longer
time to angiography, massive transfusion requirements,
previous surgery, bleeding secondary to trauma,
cancer-associated bleeding, the use of coils as the sole
embolic agent, gastritis, coagulopathy, and multi-organ
failure.[6] [21] [22] Continued bleeding was observed in 33% of
patients, but almost half of those patients responded to
repeat embolisation. Finally, 20% of the patients required
open surgical intervention for definitive management of
bleeding.[6] The overall 30-day mortality rate was 28%,
with bleeding being the underlying cause of death in most
cases.[6] [24] The presence of uncorrectable coagulopathy
was the most significant predictive factor for recurrent
bleeding and mortality.[23] Other significant predictive
factors included older age, cirrhosis, oncologic disease,
multiple organ failure, and frequent corticosteroid
treatment.[23]
COMPLICATIONS
Periprocedural complications occur with the same
frequency in TAE as in other endovascular interventions.
These complications, which often have no clinical
consequences and are mostly preventable, include
haematoma, pseudoaneurysm, arterial dissection,
contrast allergic reactions, and nephrotoxicity.[21] [22] [23] TAE
for UGIB is considered safe because of the abundant
collateral blood supply to the stomach and duodenum.
The risk of ischaemic complications increases when
collateral blood supplies are damaged by an earlier
surgical procedure of the upper abdomen, radiotherapy,
severe atherosclerosis, or when liquid embolic agents
or tiny particulate agents permeate far into the vascular
bed.[1] [21] [22] [23] [50] Duodenal strictures resulting from ischaemia
after embolisation are rare and have been reported in
<7% of cases.[39] [50] Other rare complications include
unintentional embolisation of the main hepatic artery
resulting in liver failure.[23] The overall complication rate is approximately 9%.[23]
CONCLUSION
Management of acute nonvariceal UGIB remains a
challenge. A multidisciplinary approach involving skilled
endoscopists, intensive care specialists, experienced
upper GI surgeons, and interventional radiologists is
required. For the past three decades, the techniques
and devices used for endovascular treatment of acutely
haemorrhagic patients have significantly improved. The gold standard for patients who fail endoscopic
haemostasis is now TAE, which is widely accepted as
a safe and effective treatment for life-threatening acute
nonvariceal UGIB. Endovascular embolisation may
be sufficient even for the most gravely ill patients who
are not candidates for surgical treatment, even when
angiography cannot visualise active contrast extravasation
during empiric embolisation. Interventional radiologists
should be aware of the technical and clinical factors that
affect outcomes following embolotherapy. Embolisation
should be performed as early as possible after the onset
of bleeding with concurrent correction of coagulopathy.
In addition, careful selection of the appropriate embolic
agents according to the characteristics of the bleeding
vessel and the patient’s circumstances may improve
overall technical and clinical outcomes.
REFERENCES
1. Walker TG. Acute gastrointestinal hemorrhage. Tech Vasc Interv
Radiol. 2009;12:80-91. Crossref
2. Green BT, Rockey DC. Lower gastrointestinal bleeding —
management. Gastroenterol Clin North Am. 2005;34:665-78. Crossref
3. Abdel-Aal AK, Bag AK, Saddekni S, Hamed MF, Ahmed FY.
Endovascular management of nonvariceal upper gastrointestinal
hemorrhage. Eur J Gastroenterol Hepatol. 2013;25:755-63. Crossref
4. Frisoli JK, Sze DY, Kee S. Transcatheter embolization for the
treatment of upper gastrointestinal bleeding. Tech Vasc Interv
Radiol. 2004;7:136-42. Crossref
5. Huang CS, Lichtenstein DR. Nonvariceal upper gastrointestinal
bleeding. Gastroenterol Clin North Am. 2003;32:1053-78. Crossref
6. Loffroy R, Rao P, Ota S, De Lin M, Kwak BK, Geschwind JF.
Embolization of acute nonvariceal upper gastrointestinal
hemorrhage resistant to endoscopic treatment: results and predictors
of recurrent bleeding. Cardiovasc Intervent Radiol. 2010;33:1088-
100. Crossref
7. Zurkiya O, Walker TG. Angiographic evaluation and management
of nonvariceal gastrointestinal hemorrhage. AJR Am J Roentgenol.
2015;205:753-63. Crossref
8. Lee EW, Laberge JM. Differential diagnosis of gastrointestinal
bleeding. Tech Vasc Interv Radiol. 2004;7:112-22. Crossref
9. Mamdani M, Juurlink DN, Kopp A, Naglie G, Austin PC, Laupacis A.
Gastrointestinal bleeding after the introduction of COX 2 inhibitors:
ecological study. BMJ. 2004;328:1415-6. Crossref
10. Jutabha R, Jensen DM. Management of upper gastrointestinal
bleeding in the patient with chronic liver disease. Med Clin North
Am. 1996;80:1035-68. Crossref
11. Llach J, Elizalde JI, Guevara MC, Pellisé M, Castellot A, Ginès A,
et al. Endoscopic injection therapy in bleeding Mallory-Weiss
syndrome: a randomized controlled trial. Gastrointest Endosc.
2001;54:679-81. Crossref
12. Morales P, Baum AE. Therapeutic alternatives for the Mallory-Weiss tear. Curr Treat Options Gastroenterol. 2003;6:75-83. Crossref
13. Romãozinho JM, Pontes JM, Lérias C, Ferreira M, Freitas D.
Dieulafoy’s lesion: management and long-term outcome.
Endoscopy. 2004;36:416-20. Crossref
14. Sone Y, Kumada T, Toyoda H, Hisanaga Y, Kiriyama S, Tanikawa M.
Endoscopic management and follow up of Dieulafoy lesion in the
upper gastrointestinal tract. Endoscopy. 2005;37:449-53. Crossref
15. Meyer CT, Troncale FJ, Galloway S, Sheahan DG. Arteriovenous malformations of the bowel: an analysis of 22 cases and a review
of the literature. Medicine (Baltimore). 1981;60:36-48. Crossref
16. Cumpa EA, Stevens R, Hodgson K, Castro F. Primary aortoenteric
fistula. South Med J. 2002;95:1071-3. Crossref
17. Chapman WC, Abecassis M, Jarnagin W, Mulvihill S, Strasberg SM.
Bile duct injuries 12 years after the introduction of laparoscopic
cholecystectomy. J Gastrointest Surg. 2003;7:412-6. Crossref
18. Loffroy R, Guiu B, Cercueil JP, Lepage C, Cheynel N, Steinmetz E,
et al. Transcatheter arterial embolization of splenic artery aneurysms
and pseudoaneurysms: short- and long-term results. Ann Vasc Surg.
2008;22:618-26. Crossref
19. Gomes AS, Lois JF, McCoy RD. Angiographic treatment of
gastrointestinal hemorrhage: comparison of vasopressin infusion
and embolization. AJR Am J Roentgenol. 1986;146:1031-7. Crossref
20. Parente F, Anderloni A, Bargiggia S, Imbesi V, Trabucchi E, Baratti C,
et al. Outcome of non-variceal acute upper gastrointestinal
bleeding in relation to the time of endoscopy and the experience
of the endoscopist: a two-year survey. World J Gastroenterol.
2005;11:7122-30. Crossref
21. Shin JH. Recent update of embolization of upper gastrointestinal tract bleeding. Korean J Radiol. 2012;13 Suppl 1:S31-9. Crossref
22. Shin JH. Refractory gastrointestinal bleeding: role of angiographic
intervention. Clin Endosc. 2013;46:486-91. Crossref
23. Valek V, Husty J. Quality improvement guidelines for transcatheter
embolization for acute gastrointestinal nonvariceal hemorrhage.
Cardiovasc Intervent Radiol. 2013;36:608-12. Crossref
24. Loffroy R, Favelier S, Pottecher P, Estivalet L, Genson PY, Gehin S,
et al. Transcatheter arterial embolization for acute nonvariceal upper
gastrointestinal bleeding: indications, techniques and outcomes.
Diagn Interv Imaging. 2015;96:731-44. Crossref
25. Palmer K. Management of haematemesis and melaena. Postgrad
Med J. 2004;80:399-404. Crossref
26. Kovacs TO, Jensen DM. Recent advances in the endoscopic
diagnosis and therapy of upper gastrointestinal, small intestinal,
and colonic bleeding. Med Clin North Am. 2002;86:1319-56. Crossref
27. Artigas JM, Marti M, Soto JA, Esteban H, Pinilla I, Guillén E.
Multidetector CT angiography for acute gastrointestinal bleeding:
technique and findings. Radiographics. 2013;33:1453-70. https://doi.org/10.1148/rg.335125072Crossref
28. Geffroy Y, Rodallec MH, Boulay-Coletta I, Jullès MC,
Ridereau-Zins C, Zins M. Multidetector CT angiography in acute
gastrointestinal bleeding: why, when, and how. Radiographics.
2011;31:E35-46. Crossref
29. Soto JA, Park SH, Fletcher JG, Fidler JL. Gastrointestinal
hemorrhage: evaluation with MDCT. Abdom Imaging.
2015;40:993-1009. Crossref
30. Loffroy R, Guiu B, D’Athis P, Mezzetta L, Gagnaire A, Jouve JL,
et al. Arterial embolotherapy for endoscopically unmanageable
acute gastroduodenal hemorrhage: predictors of early rebleeding.
Clin Gastroenterol Hepatol. 2009;7:515-23. Crossref
31. Walsh RM, Anain P, Geisinger M, Vogt D, Mayes J, Grundfest-
Broniatowski S, et al. Role of angiography and embolization
for massive gastroduodenal hemorrhage. J Gastrointest Surg.
1999;3:61-5. Crossref
32. Krüger K, Heindel W, Dölken W, Landwehr P, Lackner K.
Angiographic detection of gastrointestinal bleeding. An
experimental comparison of conventional screen-film angiography
and digital subtraction angiography. Invest Radiol. 1996;31:451-7. Crossref
33. Song JS, Kwak HS, Chung GH. Nonvariceal upper gastrointestinal
bleeding: the usefulness of rotational angiography after endoscopic
marking with a metallic clip. Korean J Radiol. 2011;12:473-80. Crossref
34. Eriksson LG, Sundbom M, Gustavsson S, Nyman R. Endoscopic
marking with a metallic clip facilitates transcatheter arterial
embolization in upper peptic ulcer bleeding. J Vasc Interv Radiol.
2006;17:959-64. Crossref
35. Padia SA, Geisinger MA, Newman JS, Pierce G, Obuchowski NA,
Sands MJ. Effectiveness of coil embolization in angiographically
detectable versus non-detectable sources of upper gastrointestinal
hemorrhage. J Vasc Interv Radiol. 2009;20:461-6. Crossref
36. Defreyne L, Vanlangenhove P, Decruyenaere J, Van Maele G,
De Vos M, Troisi R, et al. Outcome of acute nonvariceal
gastrointestinal haemorrhage after nontherapeutic arteriography
compared with embolization. Eur Radiol. 2003;13:2604-14. Crossref
37. Aina R, Oliva VL, Therasse E, Perreault P, Bui BT, Dufresne MP,
et al. Arterial embolotherapy for upper gastrointestinal hemorrhage:
outcome assessment. J Vasc Interv Radiol. 2001;12:195-200. Crossref
38. Lenhart M, Paetzel C, Sackmann M, Schneider H, Jung EM,
Schreyer AG, et al. Superselective arterial embolisation with
a liquid polyvinyl alcohol copolymer in patients with acute
gastrointestinal haemorrhage. Eur Radiol. 2010;20:1994-9. Crossref
39. Lang EV, Picus D, Marx MV, Hicks ME. Massive arterial
hemorrhage from the stomach and lower esophagus: impact of
embolotherapy on survival. Radiology. 1990;177:249-52. Crossref
40. Encarnacion CE, Kadir S, Beam CA, Payne CS. Gastrointestinal
bleeding: treatment with gastrointestinal arterial embolization.
Radiology. 1992;183:505-8. Crossref
41. Miller M Jr., Smith TP. Angiographic diagnosis and endovascular
management of nonvariceal gastrointestinal hemorrhage.
Gastroenterol Clin North Am. 2005;34:735-52. Crossref
42. Park JH, Kim HC, Chung JW, Jae HJ, Park JH. Transcatheter
arterial embolization of arterial esophageal bleeding with the use
of N-butyl cyanoacrylate. Korean J Radiol. 2009;10:361-5. Crossref
43. Lee CW, Liu KL, Wang HP, Chen SJ, Tsang YM, Liu HM.
Transcatheter arterial embolization of acute upper gastrointestinal
tract bleeding with N-butyl-2-cyanoacrylate. J Vasc Interv Radiol.
2007;18:209-16. Crossref
4. Mine T, Murata S, Nakazawa K, Onozawa S, Ueda T, Miyauchi M,
et al. Glue embolization for gastroduodenal ulcer bleeding:
contribution to hemodynamics and healing process. Acta Radiol.
2013;54:934-8. Crossref
45. Jae HJ, Chung JW, Jung AY, Lee W, Park JH. Transcatheter arterial
embolization of nonvariceal upper gastrointestinal bleeding with
N-butyl cyanoacrylate. Korean J Radiol. 2007;8:48-56. Crossref
46. Kim JH, Shin JH, Yoon HK, Chae EY, Myung SJ, Ko GY, et al.
Angiographically negative acute arterial upper and lower
gastrointestinal bleeding: incidence, predictive factors, and clinical
outcomes. Korean J Radiol. 2009;10:384-90. Crossref
47. Poultsides GA, Kim CJ, Orlando R 3rd, Peros G, Hallisey MJ,
Vignati PV. Angiographic embolization for gastroduodenal
hemorrhage: safety, efficacy, and predictors of outcome. Arch
Surg. 2008;143:457-61. Crossref
48. Loffroy R, Lin M, Thompson C, Harsha A, Rao P. A comparison
of the results of arterial embolization for bleeding and non-bleeding
gastroduodenal ulcers. Acta Radiol. 2011;52:1076-82. Crossref
49. Arrayeh E, Fidelman N, Gordon RL, LaBerge JM, Kerlan RK Jr,
Klimov A, et al. Transcatheter arterial embolization for upper
gastrointestinal nonvariceal hemorrhage: is empiric embolization
warranted? Cardiovasc Intervent Radiol. 2012;35:1346-54. Crossref
50. Loffroy R, Guiu B. Role of transcatheter arterial embolization
for massive bleeding from gastroduodenal ulcers. World J
Gastroenterol. 2009;15:5889-97. Crossref