Prophylactic Caesarean Iliac Artery Balloon Insertion in Patients with Abnormal Placental Implantation
ORIGINAL ARTICLE CME
Prophylactic Caesarean Iliac Artery Balloon Insertion in Patients with Abnormal Placental Implantation
CSC Tsai1, SSM Wong1, CMY Chung2, SCH Yu1
1 Department of Imaging and Interventional Radiology, Prince of Wales Hospital, Hong Kong
2 Department of Obstetrics and Gynaecology, Prince of Wales Hospital, Hong Kong
Correspondence: Prof SCH Yu, Department of Imaging & Interventional Radiology, Prince of Wales Hospital, Hong Kong. Email: simonyu@cuhk.edu.hk
Submitted: 6 Aug 2019; Accepted: 10 Dec 2019.
Contributors: All authors designed the study. CSCT, SSMW and CMYC acquired the data. CSCT and SSMW analysed the data and drafted the
manuscript. All authors critically revised the manuscript for important intellectual content. All authors had full access to the data, contributed to
the study, approved the final version for publication, and take responsibility for its accuracy and integrity.
Conflicts of Interest: All authors have disclosed no conflicts of interest.
Funding/Support: This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Data Availability: All data generated or analysed during the present study are available from the corresponding author on reasonable request.
Ethics Approval: This study was approved by the Joint Chinese University of Hong Kong–New Territories East Cluster Clinical Research Ethics Committee (Ref 2019.577). The requirement for patient consent was waived for this retrospective study.
Abstract
Introduction
We evaluated the safety and obstetric outcomes of patients with abnormal placental implantation who underwent prophylactic Caesarean iliac balloon insertion.
Methods
Clinical and procedural records of patients with abnormal placental implantation (i.e., high-grade
placenta praevia and/or placenta accreta) undergoing prophylactic Caesarean iliac artery balloon insertion in a
tertiary referral hospital from September 2009 to April 2016 were reviewed. Patients’ demographics, procedural
complications (e.g., dissection and thromboembolism) and outcomes (estimated blood loss, transfusion requirements,
immediate/delayed hysterectomy rate, postoperative sepsis, and immediate maternal/fetal mortality) were analysed.
Results
Twenty-three cases were included in the study. The median age of the patients was 36 years (range,
28-47 years). A total of 91.3% (21/23) were high-grade placenta praevia (34.8% grade III and 56.5% grade IV)
with 69.6% (16/23) co-existing placenta accreta. All prophylactic iliac balloon insertion procedures were uneventful
without major complications such as dissection or thromboembolic events. The median blood loss was 1700 mL
(100-8000 mL). The mean units of packed cells, platelets, and fresh frozen plasma transfused were 2.5, 2.2, and
2.0, respectively. The immediate and delayed hysterectomy rates were 34.8% (8/23) and 13.0% (3/23), respectively.
Postoperative sepsis incidence was 8.7% (2/23). No immediate maternal or fetal mortality was recorded. Overall,
the obstetric outcomes were comparable to data published in the literature.
Conclusion
Prophylactic Caesarean iliac artery balloon insertion for patients with abnormal placental implantation
is feasible and safe. The obstetric outcomes were comparable to data published in the literature.
Key Words: Balloon occlusion; Iliac artery; Placenta accreta; Placenta previa; Radiology, interventional
中文摘要
異常胎盤植入患者預防性剖宮產髂動脈球囊置入術
蔡紹俊、王先民、鍾汶欣、余俊豪
引言
評估接受預防性剖宮產髂骨球囊置入術的異常胎盤植入患者的安全性和產科結果。
方法
回顧2009 年 9 月至 2016 年 4 月期間在三級轉介醫院接受預防性剖宮產髂動脈球囊置入術的異常胎盤植入患者(即第三級或第四級前置胎盤以及/或者植入性胎盤)的臨床和手術記錄。分析患者的基本資料訊息、手術併發症(如撕裂和血栓栓塞)和結果(估計失血量、輸血需求、即時/延遲子宮切除率、術後敗血症和產婦/胎兒即時死亡率)。
結果
共納入23例。患者年齡中位數為36歲(介乎28-47歲)。23名患者中,21名(91.3%)出現嚴重前置胎盤(第三級佔34.8%,第四級佔56.5%),16名(69.6%)併有胎盤植入。所有預防性髂骨球囊置入術均成功,沒有出現撕裂或血栓栓塞事件等重大併發症。失血量中位數為1700 毫升(介乎100-8000毫升)。 紅血球濃厚液、血小板和新鮮冷凍血漿的平均用量分別為2.5、2.2和2.0。即時和延遲子宮切除率分別為34.8%(8例)和13.0%(3例)。術後敗血症發生率為 8.7%(2例)。沒有錄得產婦/胎兒即時死亡。總體而言,產科結果與文獻數據相若。
結論
胎盤植入異常患者進行預防性剖宮產髂動脈球囊置入術是可行且安全的。 產科結果與文獻數據相若。
INTRODUCTION
Abnormal placental implantation represents a major
challenge to obstetricians in terms of its high risks
of massive haemorrhage, disseminated intravascular
coagulopathy, sepsis, and the resulting maternal and/or
fetal mortality. Risk factors for placenta accreta include
placenta praevia, previous Caesarean section, uterine
surgery, multiparity, and advanced maternal age.[1] With
a rising incidence of abnormal placental implantation
in recent years due to increasing surgical delivery rates
worldwide,[2] multidisciplinary input from obstetricians,
obstetric anaesthetists, and interventional radiologists
are recognised as pivotal to improve obstetric outcomes.
An interventional radiology approach for management
of patients with placenta accreta and its variants was
first described by Dubois et al in 1997.[3] It involved
prophylactic balloon occlusion of the anterior division
of the internal iliac arteries followed by uterine artery
embolisation. Since then, investigations have been carried
out by various groups looking into its clinical efficacy. In
a retrospective study by Tan et al,[4] preoperative internal
iliac artery balloon occlusion reduced intraoperative
blood loss and transfusion requirements in patients with
placenta accreta and its variants undergoing Caesarean
delivery. Other studies by Shrivastava et al[5], Bodner et al,[6] and Salim et al[7] failed to show benefit of prophylactic
balloon occlusion in terms of blood loss reduction
or transfusion requirements. Therefore, its role in
management of abnormal placental implantation remains
controversial. Our study aimed to further evaluate the
safety and obstetric outcomes of patients who received
prophylactic Caesarean iliac artery balloon insertion.
METHODS
Study Design and Patients
This was a retrospective case series study, reported in
accordance with the Strengthening the Reporting of
Observational Studies in Epidemiology (STROBE)
guidelines.[8] The clinical records and procedural details of
23 consecutive patients with a high suspicion of abnormal
placental implantation, based on clinical risk factors
and ultrasonographic and magnetic resonance imaging
findings, that underwent prophylactic Caesarean iliac
artery balloon insertion in the period from September
2009 to April 2016 were retrospectively reviewed.
Procedure
The internal iliac balloon insertions were performed
on an elective basis in the angiography suite of the
radiology department. Balloon insertion procedures were
performed by a senior interventional radiologist with 18 years of experience (SCHY). Bilateral femoral
punctures were performed and 7-Fr femoral sheaths
(Radifocus; Terumo Corporation, Tokyo, Japan)
introduced. Both internal iliac arteries were sequentially
cannulated with 5-Fr catheters (RIM; Cook Medical,
Bloomington [IN], US) over a guidewire (Terumo)
using the contralateral approach (i.e., cannulation of the
left internal iliac artery via right femoral access and vice
versa). A 6-Fr compliance balloon catheter (Berenstein
Occlusion Balloon, Boston Scientific, Natick [MA], US)
was placed in the main trunk or anterior division of each
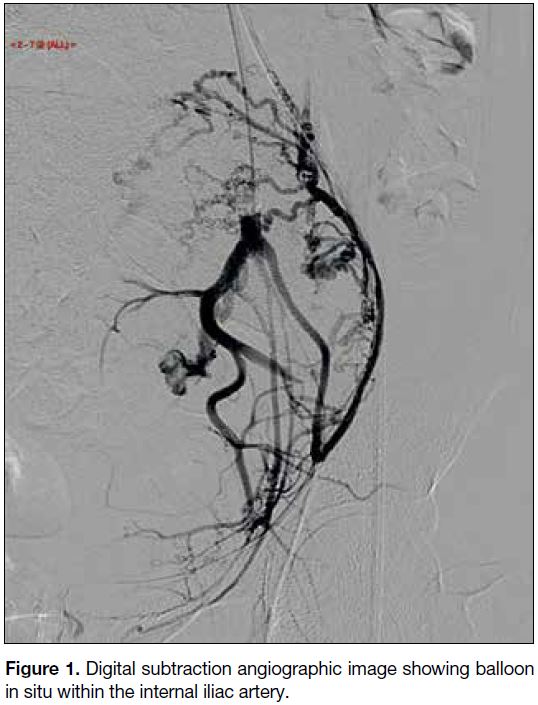
internal iliac artery (Figure 1). The balloon catheters
were connected to a container with 0.4 to 0.7 mL contrast
(Omnipaque, GE Healthcare). The catheters were then
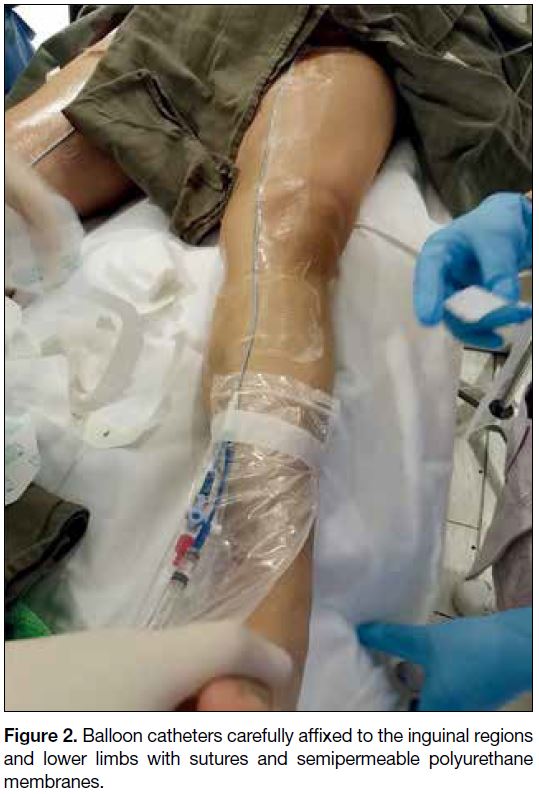
carefully anchored to the inguinal region with sutures and
semipermeable polyurethane membranes (Tegaderm,
3M Healthcare, St. Paul [MN], US) and taped to the lower
limbs (Figure 2). In order to reduce procedural radiation
dose, a low pulse rate (≤6.25 frames/s), coning/shielding,
and avoidance of digital subtraction angiography (with
roadmap only) were adopted.
Figure 1. Digital subtraction angiographic image showing balloon in situ within the internal iliac artery.
Figure 2. Balloon catheters carefully affixed to the inguinal regions
and lower limbs with sutures and semipermeable polyurethane
membranes.
Patients were then transferred to the obstetric theatre for
Caesarean delivery. Inflation of the balloon catheters was performed by the attending obstetricians after
the fetus was fully delivered and the umbilical cord
clamped. The need for balloon inflation would be
assessed by the obstetrician depending on the degree
of postpartum haemorrhage, which also determined
subsequent management with options including primary
hysterectomy, a conservative approach with uterus
and placenta in situ or uterine artery embolisation
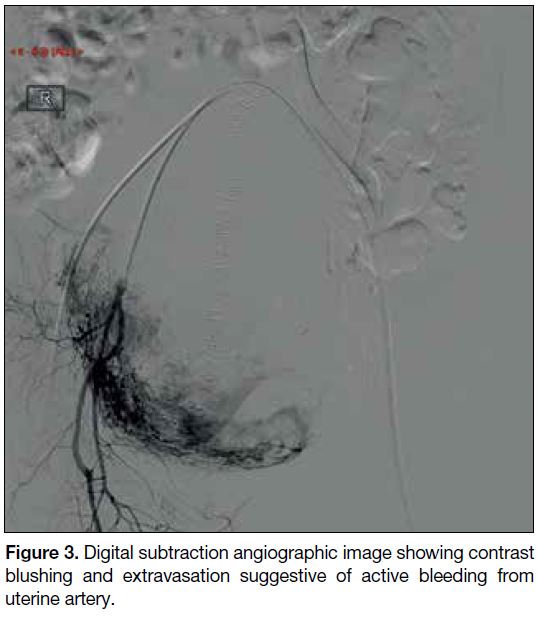
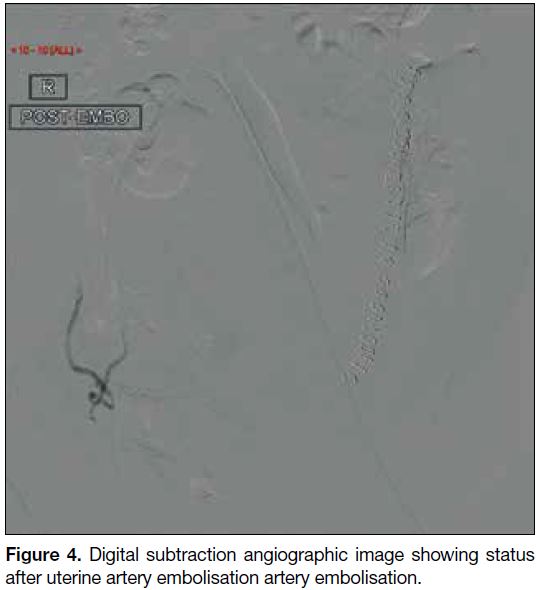
(Figures 3 and 4) using an absorbable gelatine sponge
slurry (Gelfoam; Upjohn, Kalamazoo [MI], US). The
obstetricians had the option of checking the balloon
catheters’ position intraoperatively using a C-arm if
necessary. Intravenous antibiotics were not routinely
administered unless there were signs of endometritis.
Figure 3. Digital subtraction angiographic image showing contrast
blushing and extravasation suggestive of active bleeding from
uterine artery.
Figure 4. Digital subtraction angiographic image showing status
after uterine artery embolisation artery embolisation.
The diagnosis of placenta accreta was confirmed either
intraoperatively when the placenta failed to be detached
with gentle controlled cord traction or pathologically
when the uterus and placenta were removed. For patients
with the placenta left in situ, serial ultrasound was
performed to monitor involution of the placenta in the
postoperative period.
Statistical Analysis
Basic patient demographics including the patients’ age,
gestational age, gravidity and parity, history of Caesarean
section, and other uterine surgery were recorded.
Obstetric outcomes, including estimated blood loss
during Caesarean section (as documented in the surgical record), requirements for blood product transfusion
(packed red blood cells [PRBCs], platelets, and fresh
frozen plasma [FFP]), hysterectomy rate (including
whether it was immediate or delayed), and complications
related to hysterectomy and balloon insertion were also
recorded.
Parametric variables are presented as mean and standard
deviation while non-parametric variables are presented
as median and range. Categorical variables are presented
as percentages. Statistical analysis was performed using
commercial software (SPSS Windows version 19.0;
IBM Corp, Armonk [NY], US).
RESULTS
Patient Cohort
From September 2009 to April 2016, 24 prophylactic
iliac artery balloon insertions were performed. One case
performed for uterine arteriovenous malformations was
excluded, leaving a total of 23 cases for analysis. The
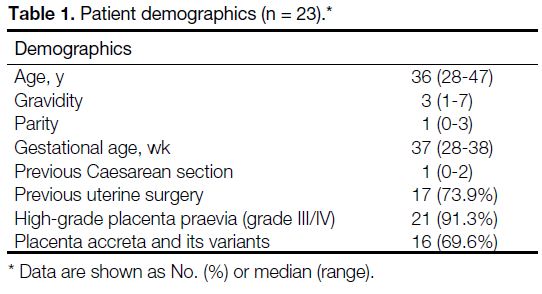
basic demographics of our patients are described in
Table 1. This group of patients demonstrated known
risk factors for abnormal placental implantation, such as
relatively advanced maternal age (median age=36 years)
and a history of Caesarean section or uterine surgery/curettage (74% of current patient cohort). Median
gravidity was 3 (range, 1-7) and parity was 1 (range,
0-3).
Table 1. Patient demographics (n = 23).
In these 23 cases, 69.6% (16/23) had the diagnosis
of placenta accreta or its variants confirmed either
intraoperatively or pathologically. A high percentage of
the cases had co-existing high-grade placenta praevia of
grade III (34.8%; 8/23) or IV (56.5%; 13/23) [Table 1].
Among the 23 cases, bleeding in two cases was deemed
not excessive and thus the obstetricians decided inflation
of the balloon catheters was not necessary. One of these
two cases had no accreta, as cord traction resulted in
complete separation of the placenta from the uterine wall
and the placenta was found to be complete. Inflation
of the balloons was not clearly documented in another
case, for a total percentage of confirmed actual balloon
inflation of 87% (20/23).
Obstetric Outcome
The estimated median blood loss during Caesarean
section was 1700 mL (range, 100-8000 mL). Blood
product transfusions thus were required by most patients,
with a mean of 2.5 (±3.7) units of PRBCs, 2.2 (±3.6) units
of platelet, and 2.0 (±3.4) units of FFP.
Immediate hysterectomy, primarily in order to control
blood loss, was performed in eight cases (35%)
while delayed hysterectomy was performed in three
cases, due to endometritis, placental infection, and
haemoperitoneum post-Caesarean section. Four cases
had retained placenta due to morbid adhesion and did not
require hysterectomy. Two of the four cases subsequently
developed infection of the retained placenta and one
required a delayed hysterectomy. No major structural
complications were reported. One case of loss of bladder
sensation was documented. There were no reported
injuries to the ureters or bowel during hysterectomy
or Caesarean section. Immediate postoperative uterine
artery embolisation was carried out in five patients
(21.7%; 5/23). Only one patient required repeated
uterine artery embolisation, which was performed to
control bleeding from a retained placenta 2 months after
the delivery. Postoperative sepsis was documented in
8.7% (2/23) of patients (Table 2).
Table 2. Summary of obstetric outcomes.
Technical Outcome
All the iliac balloon insertions were technically
successful. No balloon-related complications such as
vascular dissection or thromboembolism were reported
in these 23 cases. There was one case of balloon self-deflation
where reinflation was needed due to a loosened
3-way stopcock.
The mean radiation dose to the mother incurred in
the prophylactic Caesarean catheter insertion was
177 000±244 251 mGycm2, while mean fluoroscopic
screening time was 373±228 s. Estimated effective
dose was 46 mSv (conversion factor 0.26, adopted
from The National Council on Radiation Protection and
Measurements 2009).[9] Mean fetal dose was 11.2 mGy.
DISCUSSION
The basic demographics of our current cohort of
patients with relatively advanced maternal age, and
a high prevalence of previous Caesarean section/uterine surgery, show similarity to data published in the
literature.[4] [5] [6] The high percentage of intraoperatively/pathologically diagnosed placenta accreta or its variants
and co-existing high praevia are consistent with the
findings of Shrivastava et al[5] and Salim et al.[7]
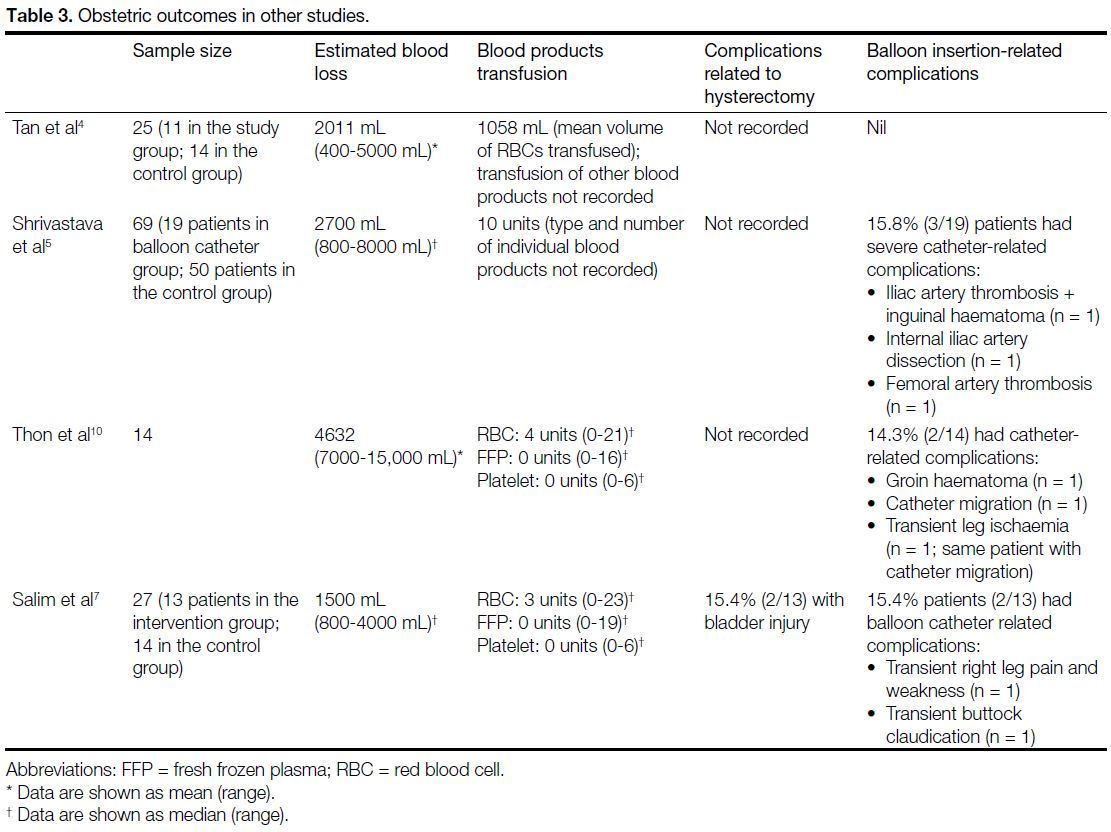
In the assessed obstetric outcomes, the estimated amount
of blood loss was comparable to published data on
patients undergoing balloon occlusion. The requirement
for blood product transfusion (PRBCs, platelets, and
FFP) varies significantly in the literature (Table 3[4] [5] [7] [10]),
with total units of blood product transfusion up to 10 in
a balloon catheter intervention group.[5] In contrast to this,
estimated blood loss and the need for blood products
was comparable/comparatively moderate in this cohort
of patients.
Table 3. Obstetric outcomes in other studies.
Conflicting evidence is observed in the literature
regarding the efficacy of the placement of internal iliac
artery balloons. In 2012, Dilauro et al[11] analysed 20 case
reports, including three case-control studies, seven
multiple-case series, and 10 single-case reports involving
a total of 132 patients who underwent this procedure,
and found that the reduction in postpartum haemorrhage
or transfusion requirements were inconsistent among
different investigators. Tan et al[4] reported a significant
decrease in intraoperative blood loss in patients with
occlusion balloon versus Caesarean section alone
(2011 mL vs. 3316 mL; p = 0.042) and reduction
in required blood transfusion volume (1058 mL vs.
2211 mL; p = 0.005). Angstmann et al[12] demonstrated
a significant decrease in intraoperative blood loss
(553 mL vs. 4517 mL; p < 0.001) and the need for
transfusion (p = 0.001). However, other studies such as
Levine et al,[13] Shrivastava et al,[5] and Salim et al[7] showed
no significant differences in estimated intraoperative
blood loss, need for transfused blood products, or length
of hospital stay. The available literature is mostly small-scale
retrospective studies and very few randomised controlled studies.[7] We believe that non-standardisation
of procedure techniques, operator experience, and
factors such as patient referral pattern and selection bias
are causing such inconclusive evidence and thus the
ambiguity about its efficacy. Also, there are potential
ethical limitations in performing randomised controlled
studies in this group of patients with high obstetric risks
leading to only very few randomised prospective studies
in this topic.
Reasons for failure to reduce intraoperative risks of
bleeding or obstetric outcomes may be due to the
presence of extensive vascular anastomoses in the gravid
uterus. The collateral arterial supply from the cervical,
ovarian, rectal, and even lumbar arteries can contribute
to overall blood loss[19] even the uterine arteries have been
occluded by balloon catheters. Kidney et al[14] have argued
that the presence of distended balloons reduces arterial
pressure to that of venous pressure, and by this means,
haemostasis and blood clot formation can be facilitated.
Therefore, investigators have also been trying to modify
the balloon occlusion technique by placing the balloon
at more proximal sites, such as at the common iliac
arteries or the infrarenal abdominal aorta. This was
performed with an aim to arrest the extensive collateral
flow arising proximal to the internal iliac arteries.
Omar et al[15] reviewed 19 studies, including 57 cases
of prophylactic arterial balloon occlusion performed at
multiple sites, including the abdominal aorta, common
iliac arteries, internal iliac arteries, the anterior division
of the internal iliac arteries and the uterine arteries. The
authors observed there is more conflicting evidence when
balloons were placed in the internal iliac arteries or the
uterine arteries, with one positive controlled study4 and
three negative controlled studies.[5] [6] [13] Angstmann et al[12]
placed balloon occlusion catheters in the common
iliac arteries and yielded positive result with reduction
in maternal morbidity. Authors of three case reports
using balloon occlusion in the abdominal aorta reported
favourable outcome as well.[16] [17] [18] A more recent study performing temporary occlusion of the infrarenal aorta
in 42 cases also gave favourable outcomes (estimated
blood loss 0.58 L).[19] Omar et al[15] postulated the clinical
success could be proportional to the ability of abolishing
the extensive collateral blood supply. To support or
validate this deduction, large-scale randomised and
prospective studies will be required. The safety profile of
more proximal balloon occlusion, such as in the common
iliac arteries or abdominal aorta, will have to be carefully
evaluated because potential complications include lower
limb ischaemia, lower limb embolism, reperfusion
injury, vessel thrombosis, and aortic rupture. Also,
aortic balloons have a much larger profile and are thus
more prone to migration during inflation. Its inflation
process will also likely increase the blood pressure due
to significant obstruction to circulation.
In our review of the procedural records of these 23
prophylactic internal iliac balloon catheter insertions,
it showed that this procedure could be performed
safely and was feasible. No major significant balloon
insertion–related complications were found in this
cohort of patients. However, arterial thrombosis, a major
complication requiring thrombectomy,[20] [21] and minor
complications such as transient leg pain, weakness,
and claudication have been reported.[7] [10] This aspect is
particularly important as major vascular complications
could have significant implications in these patients.
A well-defined safety profile will be essential in
establishing the efficacy of this prophylactic procedure,
assuming it is proven in randomised controlled trials to
be of benefit.
There were no hysterectomy-related complications in
terms of structural damage to the adjacent organs such
as the bowel, ureters or urinary bladder. One case of loss
of urinary bladder sensation was documented, however,
which may have been caused by inadvertent damage to
adjacent pelvic nerves.
It is always important to minimise radiation exposure
to both mothers and especially fetuses, so meticulous
techniques were employed, including low pulse rate
(≤6.25 frame/s), coning/shielding, and avoidance of
digital subtraction angiography (with roadmap only).
The mean fetal dose observed in the present study was
11.2 mGy. This represents a probability of birth without
malformation or childhood cancer of approximately
95.8% compared with 95.9% in children without
radiation exposure. This probability is 0.1% higher
than that in children born without radiation exposure during pregnancy.[22] This needs to be balanced against
the potential benefits of reduction of intraoperative blood
loss and need for transfusion,[4] [12] again necessitating a
randomised controlled multicentre trial.
The strength of current study was the consistency
of the balloon catheter procedures, as it was carried
out/supervised by single experienced interventional
radiologist, and the relatively large number of patients.
However, it is limited by its lack of a control group for
direct comparison and the fact that not all patients were
confirmed to have placenta accreta. Future studies with
standardised procedure protocol in prospective setting
will be helpful to evaluate and establish the clinical
efficacy of prophylactic internal iliac balloon insertion.
In conclusion, we have demonstrated prophylactic
internal iliac balloon insertion for patients with suspected
abnormal placental implantation is safe and feasible. The
obstetric outcomes were comparable to data in the major
literature.
REFERENCES
1. Miller DA, Chollet JA, Goodwin TM. Clinical risk factors for placenta previa-placenta accreta. Am J Obstet Gynecol.
1997;177:210-4. Crossref
2. Matthews TG, Crowley P, Chong A, McKenna P, McGarvey C, O’Regan M. Rising caesarean section rates: a cause for concern?
BJOG. 2003;110:346-9. Crossref
3. Dubois J, Garel L, Grignon A, Lemay M, Leduc L. Placenta percreta: balloon occlusion and embolization of the internal iliac
arteries to reduce intraoperative blood losses. Am J Obstet Gynecol.
1997;176:723-6. Crossref
4. Tan CH, Tay KH, Sheah K, Kwek K, Wong K, Tan HK, et al. Perioperative endovascular internal iliac artery occlusion balloon
placement in management of placenta accreta. AJR Am J
Roentgenol. 2007;189:1158-63. Crossref
5. Shrivastava V, Nageotte M, Major C, Haydon M, Wing D. Case-control comparison of cesarean hysterectomy with and without
prophylactic placement of intravascular balloon catheters for
placenta accreta. Am J Obstet Gynecol. 2007;197:402.e1-5. Crossref
6. Bodner LJ, Nosher JL, Gribbin C, Siegel RL, Beale S, Scorza W.
Balloon-assisted occlusion of the internal iliac arteries in patients
with placenta accreta/percreta. Cardiovasc Intervent Radiol.
2006;29:354-61. Crossref
7. Salim R, Chulski A, Romano S, Garmi G, Rudin M, Shalev E.
Precesarean prophylactic balloon catheters for suspected
placenta accreta: a randomized controlled trial. Obstet Gynecol.
2015;126:1022-8. Crossref
8. von Elm E, Altman DG, Egger M, Pocock SJ, Gøtzsche PC,
Vandenbroucke JP, et al. The Strengthening the Reporting of
Observational Studies in Epidemiology (STROBE) statement:
guidelines for reporting observational studies. Epidemiology.
2007;18:800-4. Crossref
9. National Council on Radiation Protection and Measurements.
Report 160 — Ionizing Radiation Exposure of the Population of the
United States. Available from: https://ncrponline.org/publications/reports/ncrp-report-160-2/. Accessed 11 Jun 2019.
10. Thon S, McLintic A, Wagner Y. Prophylactic endovascular
placement of internal iliac occlusion balloon catheters in parturients
with placenta accreta: a retrospective case series. Int J Obstet
Anesth. 2011;20:64-70. Crossref
11. Dilauro MD, Dason S, Athreya S. Prophylactic balloon occlusion
of internal iliac arteries in women with placenta accreta: literature
review and analysis. Clin Radiol. 2012;67:515-20. Crossref
12. Angstmann T, Gard G, Harrington T, Ward E, Thomson A,
Giles W. Surgical management of placenta accreta: a cohort series
and suggested approach. Am J Obstet Gynecol. 2010;202:38.e1-9. Crossref
13. Levine AB, Kuhlman K, Bonn J. Placenta accreta: comparison of
cases managed with and without pelvic artery balloon catheters. J
Matern Fetal Med. 1999;8:173-6. Crossref
14. Kidney DD, Nguyen AM, Ahdoot D, Bickmore D, Deutsch LS,
Majors C. Prophylactic perioperative hypogastric artery balloon
occlusion in abnormal placentation. AJR Am J Roentgenol.
2001;176:1521-4. Crossref
15. Omar HR, Karlnoski R, Mangar D, Mangar D, Patel R, Hoffman M,
Camporesi E. Staged endovascular balloon occlusion versus
conventional approach for patients with abnormal placentation: a
literature review. J Gynecol Surg. 2012;28:247-54. Crossref
16. Paull JD, Smith J, William L, Davison G, Devine T, Holt M. Balloon
occlusion of the abdominal aorta during caesarean hysterectomy for placenta percreta. Anaesth Intensive Care. 1995;23:731-4. Crossref
17. Bell-Thomas SM, Penketh RJ, Lord RH, Davies NJ, Collis R.
Emergency use of a transfemoral aortic occlusion catheter to
control massive haemorrhage at caesarean hysterectomy. BJOG.
2003;110:1120-2. Crossref
18. Masamoto H, Uehara H, Gibo M, Okubo E, Sakumoto K, Aoki Y.
Elective use of aortic balloon occlusion in caesarean hysterectomy
for placenta previa percreta. Gynecol Obstet Invest. 2009;67:92-5. Crossref
19. Duan XH, Wang YL, Han HW, Chen ZM, Chu QI, Wang L, et al.
Caesarean section combined with temporary aortic balloon
occlusion followed by uterine artery embolisation for the
management of placenta accreta. Clin Radiol. 2015;70:932-7. Crossref
20. Sewell MF, Rosenblum D, Ehrenberg H. Arterial embolus during common iliac balloon catheterization at cesarean hysterectomy. Obstet Gynecol. 2006;108:746-8. Crossref
21. Royal College of Obstetricians & Gynaecologists. Placenta
praevia, placenta praevia accreta and vasa praevia: diagnosis
and management (Green-top Guideline No. 27). 2011. Available
from: https://www.rcog.org.uk/en/guidelines-research-services/
guidelines/gtg27/. Accessed 17 Jun 2019.
22. McCollough CH, Schueler BA, Atwell TD, Braun NN, Regner DM, Brown DL, et al. Radiation exposure and pregnancy: when should
we be concerned? Radiographics. 2007;27:909-17. Crossref