First-Line Therapy for Metastatic Castration-sensitive Prostate Cancer: a Network Meta-analysis
REVIEW ARTICLE CME
First-Line Therapy for Metastatic Castration-sensitive Prostate Cancer: a Network Meta-analysis
KYC Zheng1, AKH Fong1, SK Chan2, TH So2
1 Li Ka Shing Faculty of Medicine, The University of Hong Kong, Hong Kong
2 Department of Clinical Oncology, The University of Hong Kong, Hong Kong
Correspondence: Dr TH So, Department of Clinical Oncology, The University of Hong Kong, Hong Kong. Email: sotszhim2@gmail.com
Submitted: 11 May 2021; Accepted: 5 Oct 2021.
Contributors: THS designed the study. KYCZ and AKHF acquired the data. KYCZ, SKC and THS analysed the data. All authors drafted the
manuscript. THS critically revised the manuscript for important intellectual content.
Conflicts of Interest: The authors have no conflicts of interest to declare.
Funding/Support: The authors received no financial support for the research or publication of this article.
Data Availability: All data generated or analysed during the present study are available from the corresponding author on reasonable request.
Ethics Approval: Approval was not required as no patients were involved in setting the research question or the outcome measures, nor were
they involved in developing plans for design and implementation of the study.
Declaration: The results/methods from this study were presented in part at ESMO Asia Congress 22-24 November 2019, Singapore.
Abstract
Objective
The treatment landscape of metastatic castration-sensitive prostate cancer (mCSPC) has been transforming
in the past decade. Abiraterone acetate plus prednisolone (AAP), apalutamide (APA), enzalutamide (ENZA), and
docetaxel (Doce) added to androgen deprivation therapy (ADT) were shown to outperform ADT alone. However,
data on direct comparison of the different regimens are sparse. We sought to review current evidence on first-line
therapies in mCSPC and compare their results in terms of overall survival (OS) and progression-free survival (PFS)
in a network meta-analysis.
Methods
We performed a systematic search of PubMed, MEDLINE, Web of Science, EMBASE, ClinicalTrials.gov,
and Cochrane Library databases in September 2020. ADT was the reference category. Treatments were grouped into
four categories: Doce+ADT, AAP+ADT, APA+ADT, and ENZA+ADT. The primary endpoint of our study was OS.
Results
We analysed eight trials with 7790 total patients, using frequentist network meta-analysis and P-score to
rank the treatments. AAP+ADT showed the highest P-score of 86% with a hazard ratio (HR) of 0.63 (95% confidence
interval [CI]=0.56-0.71) in OS while ENZA+ADT performed best in PFS (HR=0.40, 95% CI=0.34-0.46) with a
P-score of 98%.
Conclusion
We found that AAP+ADT treatment was most effective in prolonging OS. ENZA+ADT might provide
better PFS in mCSPC. Analysis of OS and PFS provides guidance on selecting the best choice of first-line treatments.
Key Words: Prostatic neoplasms; Meta-analysis; Therapeutics
中文摘要
轉移性去勢敏感性前列腺癌的一線治療:網絡薈萃分析
鄭裕誠、房嘉希、陳錫坤、蘇子謙
目的
在過去十年,轉移性去勢敏感性前列腺癌(mCSPC)的治療前景出現轉變。將阿比特龍併潑尼松龍(AAP)、阿帕魯胺(APA)、恩扎盧胺(ENZA)和多西紫杉醇(Doce)加入雄激素剝奪療法(ADT)的效果優於單純ADT。然而,有關直接比較不同方案的數據很少。本文回顧目前mCSPC一線治療的證據,並在網絡薈萃分析中比較治療方案的總生存期和疾病無進展生存期。
方法
研究於2020年 9月對PubMed、MEDLINE、Web of Science、EMBASE、ClinicalTrials.gov和 Cochrane Library 數據庫進行系統搜索。以ADT作為參考類別。治療分為四類:Doce+ADT、AAP+ADT、APA+ADT 和 ENZA+ADT。我們研究的主要終點指標是總生存期。
結果
我們分析 8項試驗研究,涉及 7790名患者,使用頻率學網絡薈萃分析和 P 評分對治療進行排名。 AAP+ADT 在整體存活期中的 P值最高,達 86%,風險比為 0.63(95%置信區間 = 0.56-0.71),而ENZA+ADT在疾病無進展生存期中表現最佳,風險比為0.40(95%置信區間 = 0.34-0.46),P 值為98%。
結論
AAP+ADT治療對延長mCSPC總生存期最為有效,而ENZA+ADT則有助改善mCSPC疾病無進展生存期。總生存期和疾病無進展生存期的分析可為選擇最佳一線治療方案提供指引。
INTRODUCTION
Prostate cancer (PC) has the second highest incidence
and is the third leading cause of cancer death among
men in the world. In 2018, more than 1.2 million new
cases were diagnosed and 359,000 deaths were reported
worldwide.[1] Approximately 89% of newly diagnosed
cases are locoregional, with a 5-year survival rate of
nearly 100%; 6% are diagnosed at the metastatic stage,
which has a significantly worse 5-year relative survival
of 30.2%.[2]
Androgen deprivation therapy (ADT) has been the
standard of care for patients with metastatic PC
since Huggins and Hodges discovered the hormone
dependency of PC in the 1940s.[3] ADT was proven to
be effective in producing improved radiological and
biochemical profiles and prolonging overall survival
(OS).[4] Multiple randomised controlled trials (RCTs)
have been performed to evaluate the benefits of other
therapies added to ADT for metastatic castration-sensitive
PC (mCSPC). Following the STAMPEDE
Arm C,[5] [6] CHAARTED,[7] [8] and GETUG-AFU 15 trials,[9] [10]
docetaxel (Doce) plus ADT was recommended as first-line
treatment (especially for high-volume disease) for
mCSPC in 2015.[11] Treatment protocols have further evolved over the past 5 years. Positive results from the
LATITUDE,[12] [13] STAMPEDE Arm G,[14] [15] TITAN,[16]
ARCHES[17] and ENZAMET[18] studies have shown
that addition of abiraterone acetate plus prednisolone
(AAP), apalutamide (APA), or enzalutamide (ENZA)
to ADT showed superior results compared to ADT
alone in mCSPC and are now considered standard
treatment protocols.[19] However, no head-to-head RCT
has been conducted to compare the survival benefits
of these regimens. We therefore conducted a network
meta-analysis to guide the selection of the best first-line
combination therapy for mCSPC.
METHODS
Inclusion Criteria
This study was conducted in accordance with the PRISMA
(Preferred Reporting Items for Systematic Review and
Meta-analysis) guidelines.[20] We included RCTs only
if they included treatment of mCSPC. We specifically
included trials of treatment regimens containing ADT
plus AAP, APA, Doce, or ENZA to assess the efficacy
in systemic treatments. Included clinical trials needed to
contain a control arm with ADT alone or with placebo
for carrying out indirect comparison by network meta-analysis.
Literature Search and Data Collection
We performed a comprehensive systematic literature
search for full-length journal publications, which
included mCSPC ADT. The PubMed/MEDLINE
Ovid, Embase, Cochrane Library, CINAHL Databases,
trial registries, and other sources were employed and
searched from 1990 to the present day, with the most
recent search carried out on 30 September 2020. Key
words and medical sub-heading (MeSH) terms included:
‘prostate cancer’, ‘metastatic’, ‘castration-sensitive’,
and ‘treatment’. Key words used for searching included:
“prostate” OR “prostatic”; “neoplasm” OR “cancer”
OR “cancers” OR “cancerous” “tumor” OR “tumour”;
“metastatic” OR “metastasis”; “treatment” OR
“therapy”; and “castration-sensitive” OR “castration”.
We aimed at identifying any journal articles or abstract
proceedings that published the efficacy of first-line
therapy in mCSPC. The key words and MeSH terms
within each concept were then separated by the Boolean operator ‘AND’. Only articles written in English were
included. The details and results of the literature search
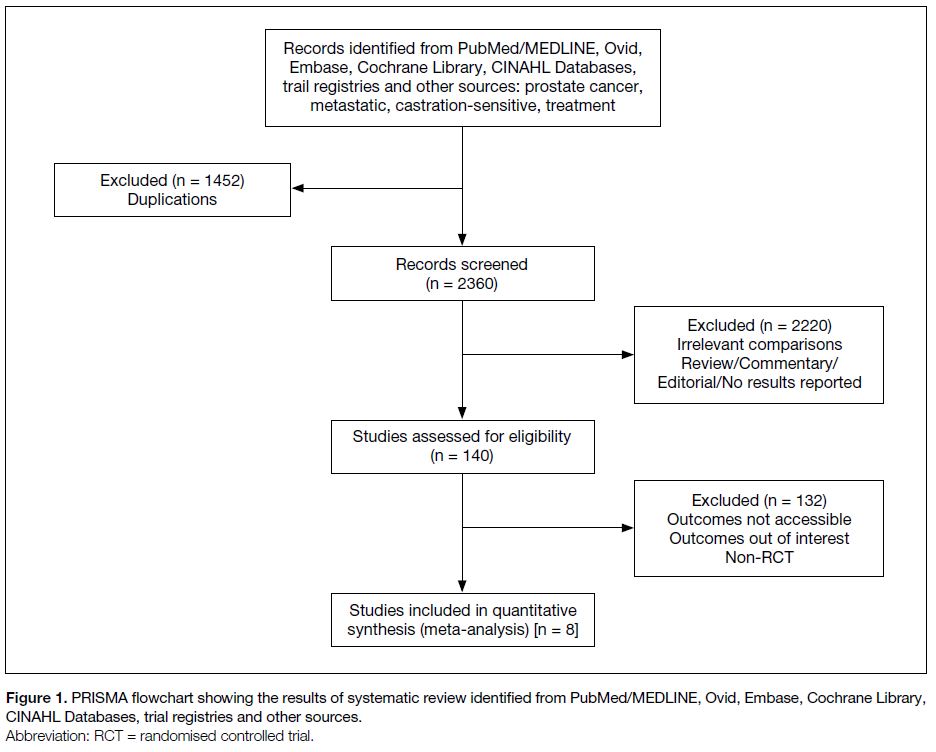
are provided in Figure 1 and Table 1.
Figure 1. PRISMA flowchart showing the results of systematic review identified from PubMed/MEDLINE, Ovid, Embase, Cochrane Library, CINAHL Databases, trial registries and other sources.
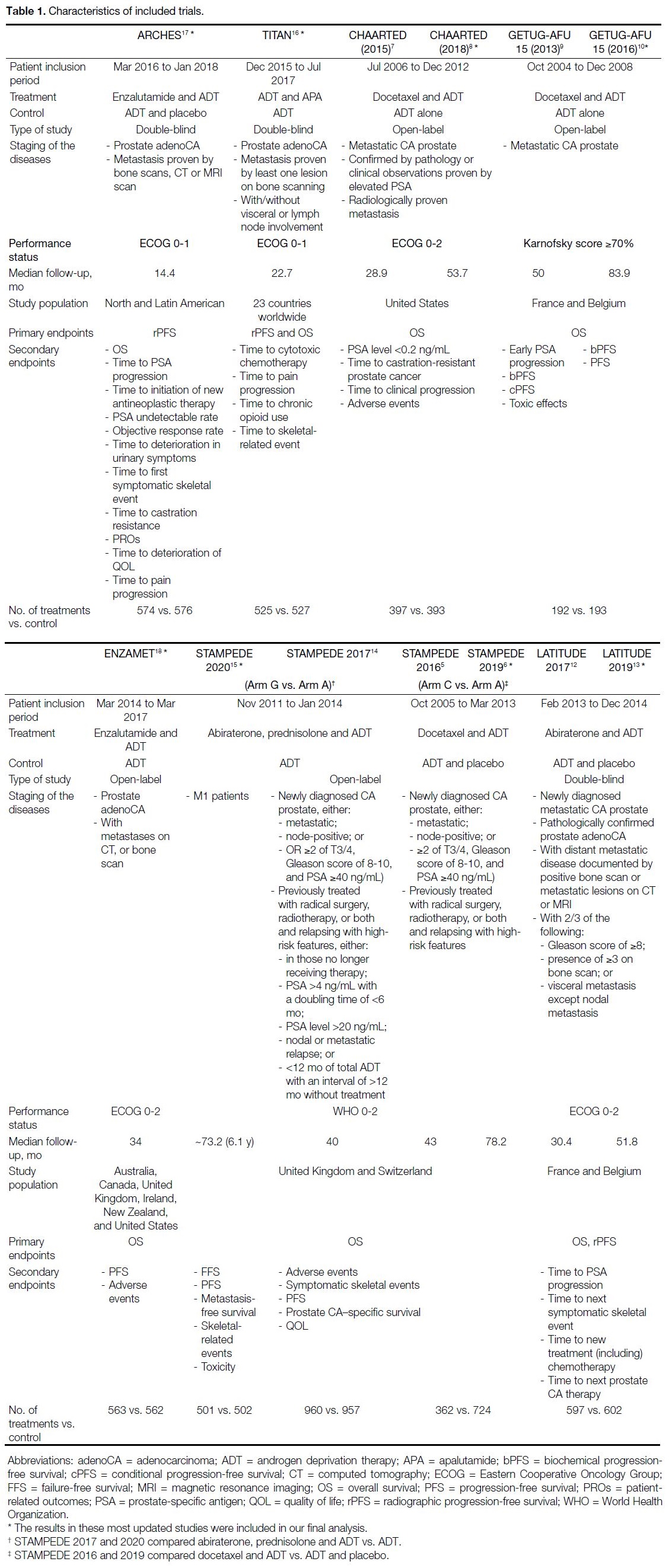
Table 1. Characteristics of included trials.
Data Extraction
The results of the findings were first screened by title
and abstract for appropriateness by two independent
reviewers (KYCZ and AKHF), while the third reviewer
(THS) was consulted if there were any disputes on
selection of relevant literature. For relevant abstracts,
the full papers would be reviewed for inclusion. More
updated versions of the publications were adopted for
meta-analysis when more than one version of the same
studies were found. Two authors (KYCZ and AKHF)
were responsible for independent data extraction based
on the same criteria.
Endpoint Definitions
The primary endpoint of our study was OS, defined as the time from the date of randomisation until the date
of death from any cause. The secondary endpoint was
progression-free survival (PFS), the time from the date
of randomisation to the date of first disease progression
(locoregional or distant) or death from any cause,
whichever occurred earlier.
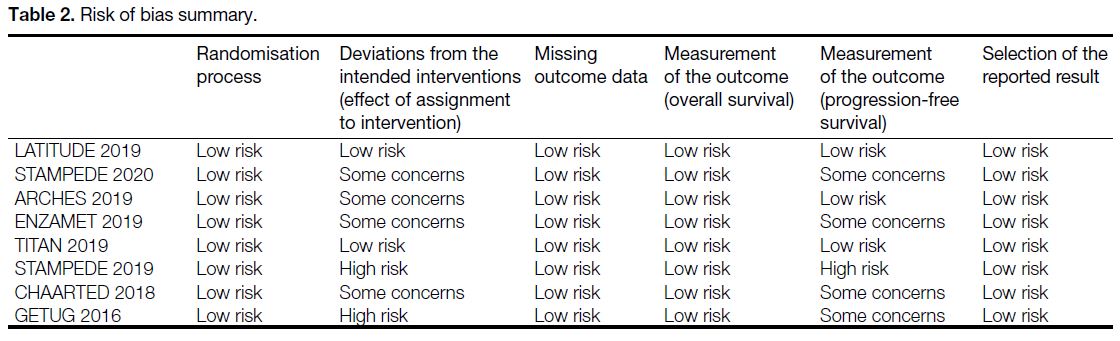
Risk of Bias Assessment
The assessment of study quality for all RCTs was
carried out independently by two authors (KYCZ
and AKHF) using the Cochrane Collaboration tool.[21]
Domains for the risk of bias assessment included:
(1) the randomisation process; (2) deviations from the intended interventions; (3) missing data; (4)
measurements of the outcomes (OS, PFS); and (5)
selection of reported results. Risk of bias was judged as ‘low risk’, ‘some concerns’, or ‘high risk’ for
individual domains based on the information provided
by the authors of the included trials Table 2.
Table 2. Risk of bias summary
Statistical Analysis
Our primary analysis in comparing the efficacy of
different therapies on mCSPC was performed with both
random- and fixed-effects models under a frequentist
framework. Reported hazard ratios (HRs) for OS and
PFS in eight included trials were incorporated into the
mathematical model. Since PFS was displayed in various
forms among different studies, we adopted radiological
PFS as our primary interest. For the studies with no
radiological PFS provided, clinical or biochemical PFS
was adopted as a surrogate in our analysis.
Relative effects of each treatment were evaluated in
indirect comparison by network meta-analysis. The I2
and Q statistics were used to quantify the heterogeneity
among different trials; an I2 value of >50% and/or
significant Q statistic at p < 0.1 was regarded as significant
heterogeneity. P-scores were further employed to
evaluate the mean extent of probability that one treatment
outperforms the others.[22] In other words, the higher the
P-score, the higher the certainty that a treatment is better
than the others. Efficacy of treatments on OS and PFS
was ranked by the relative P-score in our analysis.
Publication bias could not be formally assessed in this
network meta-analysis because of the small number of
trials included. Despite the real potential for this bias,
given the relatively small number of trials, we judged the
certainty in the evidence was unlikely to be decreased by
this concern.
All statistical analyses were performed using opensource
software, (R version 4.0.2, R Foundation for
Statistical Computing, Vienna, Austria, 2018). A p value
of <0.05 was considered statistically significant.
RESULTS
The network meta-analysis consisted of eight trials
and 7790 patients, including CHAARTED, GETUG,
LATITUDE, TITAN, ARCHES, and ENZAMET.
Two studies from STAMPEDE (with Doce+ADT
and AAP+ADT as their experimental regimens,
respectively).[5] [6] [7] [8] [9] [10] [12] [13] [14] [15] [16] [17] [18] We also included the most updated
results of STAMPEDE, from the 2020 European Society
for Medical Oncology Congress.[15]
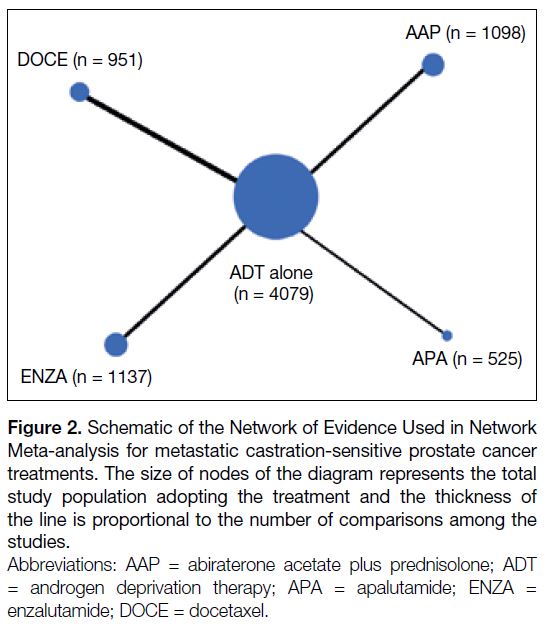
ADT was the reference category. Treatments were
grouped into four categories: Doce+ADT, AAP+ADT,
APA+ADT and ENZA+ADT. The network is displayed
in Figure 2.
Figure 2. Schematic of the Network of Evidence Used in Network
Meta-analysis for metastatic castration-sensitive prostate cancer
treatments. The size of nodes of the diagram represents the total
study population adopting the treatment and the thickness of
the line is proportional to the number of comparisons among the
studies.
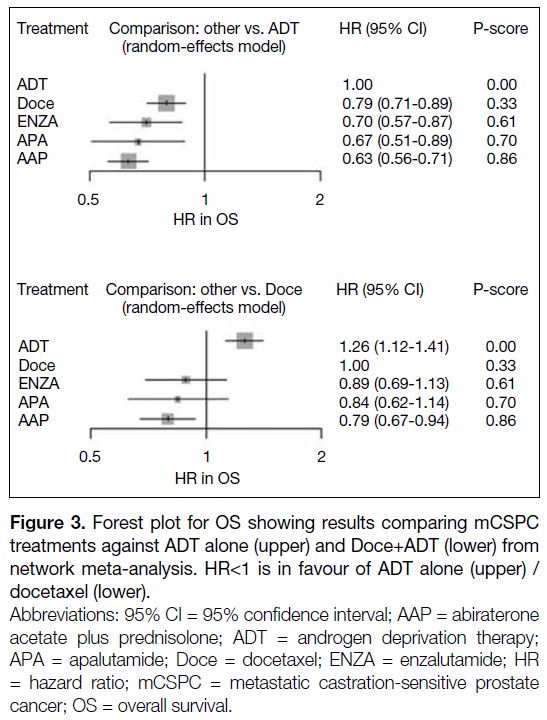
Overall Survival
AAP+ADT had the longest OS, followed by APA+ADT,
ENZA+ADT and Doce+ADT with respective P-scores
of 86%, 70%, 61% and 33% where a higher score
means a higher probability of being the best treatment
(Figure 3). Their corresponding HRs (95% confidence
interval; 95% CI) compared to ADT alone were 0.63
(0.56-0.71), 0.67 (0.51-0.89), 0.70 (0.57-0.87), and 0.79
(0.71-0.89). There was no significant heterogeneity
observed (I2 = 0%; p = 0.610 for the Q statistic).
Figure 3. Forest plot for OS showing results comparing mCSPC
treatments against ADT alone (upper) and Doce+ADT (lower) from
network meta-analysis. HR<1 is in favour of ADT alone (upper) /
docetaxel (lower).
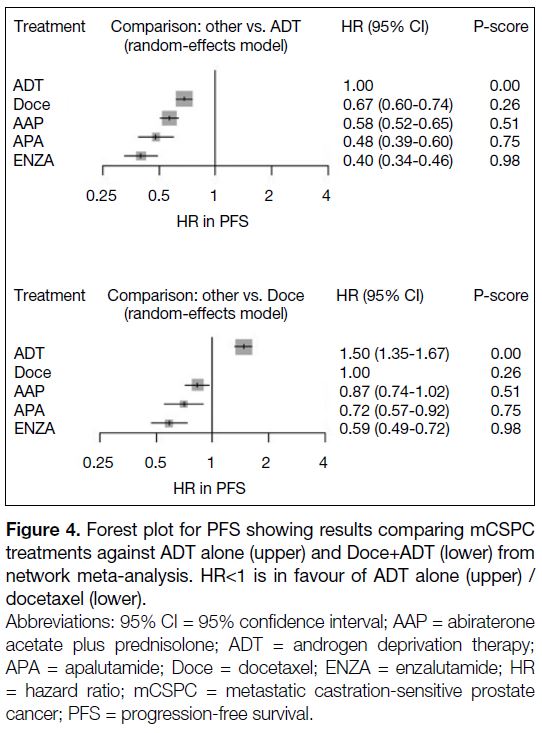
Progression-Free Survival
Results of our study in PFS did not match with our
analysis in OS. Figure 4 shows that ENZA+ADT had
the highest P-score of 98%, implying a high certainty
that ENZA+ADT was the best treatment in terms of PFS
(HR=0.40, 95% CI=0.34-0.46). APA+ADT, AAP+ADT
and Doce+ADT had P-scores of 75%, 51% and 26%,
respectively. Their corresponding HRs (95% CI) were
0.48 (0.39-0.60), 0.58 (0.52-0.65) and 0.67 (0.60-0.74).
Figure 4. Forest plot for PFS showing results comparing mCSPC
treatments against ADT alone (upper) and Doce+ADT (lower) from
network meta-analysis. HR<1 is in favour of ADT alone (upper) / docetaxel (lower).
The same results were obtained in fixed- and randomeffects
models in both OS and PFS analyses.
Exploratory Analysis
Figure 3 shows our explorative analysis in OS comparing different treatment combinations against Doce+ADT.
Our study revealed that only AAP+ADT showed a
significant benefit when compared with Doce+ADT
(HR=0.79, 95% CI=0.67-0.94). A P-score of 86%
was recorded when compared with Doce+ADT. Other
modalities, including APA+ADT and ENZA+ADT, had
no significant OS benefit compared to Doce+ADT.
In the explorative analysis against Doce+ADT, ENZA+ADT (HR=0.59, 95% CI=0.49-0.72) and APA+ADT (HR=0.72, 95% CI=0.57-0.92) had significant benefit in PFS. Although all four treatment options showed superiority over ADT alone (HR=1.50, 95% CI=1.35-1.67), AAP+ADT did not show a significant difference in improving the PFS of mCSPC patients, as compared to Doce+ADT (HR=0.87, 95% CI=0.74-1.02).
Similarly, the results of exploratory analysis were
consistent in fixed- and random-effects models.
DISCUSSION
With updated evidence in 2020, the primary and
secondary endpoints were reached and revealed that
the addition of AAP, ENZA, APA, or Doce to ADT
prolonged the OS and PFS of mCSPC patients. Among
the four treatment modalities, AAP+ADT provided the
best effect in prolonging OS. This result differs from
that of another network meta-analysis published in early
2020, which suggested that ENZA+ADT is the most
effective in prolonging OS.[22] This discrepancy may be
because the ARCHES trial, which compared
ENZA+ADT versus ADT alone in mCSPC (median
OS HR = 0.81, 95% CI = 0.53-1.25, p = 0.3361) was
not included in their analysis. Furthermore, the most
updated OS data in 2020 from the STAMPEDE trial
for AAP+ADT versus ADT alone is included in our
analysis.[15] One must also notice that some RCTs had
a rather short median follow-up and the OS can be
considered premature; for instance, the median followups
for ARCHES and TITAN were 14.4 months and
22.7 months, respectively.[16] [17]
In contrast to the OS results, our network meta-analysis
showed that ENZA+ADT is the most favourable in
terms of prolonging PFS. Despite the differences in PFS
definition in each trial — be it radiographic, biochemical,
or clinical—the PFS HRs were rather consistent across
the trials concerning the same drug. We therefore
postulate that the prevention of all forms of progression
may be best achieved by ENZA+ADT as compared to
other treatment modalities.
With the difference in OS and PFS results in mind, it is
however important to note that there is no established
evidence towards PFS being a surrogate for OS, while
the latter is considered a golden endpoint. Moreover, OS
is also affected by subsequent salvage treatment beyond
progression and therefore the sequence of drugs may
contribute to the difference in PFS and OS results. Cross-resistance
exists between androgen receptor-targeted
therapies, for instance, AAP+ADT and ENZA+ADT.[23]
The sequence of agent initiation may affect the OS. There
is some evidence suggesting that the use of AAP+ADT
before ENZA+ADT gives rise to better outcomes. A
phase 2 randomised crossover trial conducted by Khalaf
et al24 has compared the prostate-specific antigen (PSA)
decline >50% (PSA50) on second-line therapy and the
median time to second PSA progression for AAP+ADT
followed by ENZA+ADT (arm A) versus ENZA+ADT
followed by AAP+ADT (arm B). It was shown that the
sequence of AAP+ADT followed by ENZA+ADT (arm
A) achieved superior PSA50 and time to second PSA
progression (HR=0.75, 95% CI=0.53-1.06). Although
this trial was designed for metastatic castration-resistant PC, it is still a direct
comparison of the biochemical effectiveness between
the two sequences on PC. We believe this might explain
the different results in PFS and OS.
In addition, the follow-up period of ARCHES (14.4 mo)
and ENZAMET (34 mo) for ENZA+ADT were
relatively short compared with that of LATITUDE
(51.8 mo) and STAMPEDE (73.2 mo) for AAP+ADT. It
remains unclear whether extended follow-up of studies
of ENZA+ADT would lead to a better OS outcome.
Thus, further analysis may be required in the future.
The above results on OS, PFS and time to PSA
progression do not provide a clear answer to which agent
is more preferable. Several other factors including the potential toxicity profile and quality of life (QOL) should
be taken into account. Apart from the general adverse
effects shared among androgen receptor targeting agents,
there are some adverse events unique to each specific
agent that may influence the treatment choice. For
example, ENZA is commonly associated with fatigue
(24.1%) and musculoskeletal events (26.4%) including
falls and fractures[17]; abiraterone acetate exerts excessive
mineralocorticoid activity; thus, it must be given
together with prednisolone, leading to some steroid-specific
adverse events which are not common in other
agents[13]; APA causes particularly high incidence of rash
(27.1%) as compared to ADT alone (8.5%).[16] Patients’
QOL with different agents is a considerable factor. A
QOL sub-study extended from the STAMPEDE trial
suggested a significantly higher global QOL score in the
first 2 years of treatment with AAP+ADT as compared
with Doce+ADT.[25] The health-related QOL, Patient
Health Questionnaire-9 depression score, and Montreal
Cognitive Assessment score of AAP+ADT and
ENZA+ADT were assessed in another trial.[26] Minimal
difference was seen in terms of Montreal Cognitive
Assessment score; while AAP+ADT was shown to be
associated with better health-related QOL and Patient
Health Questionnaire-9 scores over time, with greater
significance in the elderly group.[26]
The choice of treatment should always include
careful discussion and shared decision making among
physicians, the patient, and caregivers. It is observed that
AAP+ADT is superior to Doce+ADT, ENZA+ADT,
and APA+ADT in terms of OS, QOL, and being the
first agent initiated in mCSPC patients. However, the
abovementioned evidence on OS, PFS, sequence of
treatments, adverse events, and QOL may only serve as
a guide; no single factor should be used to pursue one
treatment over another.
Our results are in line with a recently published study by
Wang et al[27] suggesting that AAP+ADT treatment were
more effective in prolonging OS, while ENZA+ADT
might provide better PFS in mCSPC patients. The
strength of our study is that we have included the most
up-to-date data from all these major trials, including
the STAMPEDE with AAP+ADT as the experimental
regimen.[15] Also, the comprehensive inclusion of all
the most updated studies in this network meta-analysis
covering more than 7500 patients provides a clearer
comparison of survival data among different treatment
modalities, in addition to our previously published
preliminary results.[28]
However, several limitations exist in our comparison
and the findings should be interpreted within this
context. Similar to the other network meta-analyses
in the field, this analysis only provided indirect
comparisons and rankings among the named systematic
treatments by comparing the efficacy in the RCTs,
which cannot replace head-to-head clinical trials for
comparison. As shown in Table 1, the study population
and inclusion criteria varied among the different RCTs.
The variations among different studies may contribute
to heterogeneity in treatment effect, which limits our
power in comparing the efficacy among the systematic
treatments. Furthermore, our study did not account for
other outcomes, such as QOL and cost-effectiveness.
Given that the relevant data in either prospective or
retrospective clinical trials are not adequate among our
study populations, current clinical trials provide limited
insights on the effects on QOL and cost-effectiveness
among different patients.
Another possible weakness of our study is that we have
not performed subgroup analysis according to low/high
risk (according to LATITUDE study) and low/high
volume (according to CHAARTED study). However,
recent studies have disproven the importance of risk and
volume classification in selecting treatment regimens.[29]
Also, our analysis only included drug regimens and
did not include radiotherapy to the primary site as a
comparison arm.
CONCLUSION
We found that AAP+ADT is the most effective first-line
treatment for mCSPC in terms of OS, while ENZA+AA
may provide better PFS. Clinicians should take these
findings into consideration when planning treatment of
mCSPC.
REFERENCES
1. Ferlay J, Colombet M, Soerjomataram I, Mathers C, Parkin DM,
Piñeros M, et al. Estimating the global cancer incidence and
mortality in 2018: GLOBOCAN sources and methods. Int J Cancer.
2019;144:1941-53. Crossref
2. National Cancer Institute, National Institutes of Health, US
Department of Health and Human Services. SEER, Surveillance,
Epidemiology, and End Results Program [electronic resource].
Bethesda, MD. 2005. Available from: https://seer.cancer.gov/.
Accessed 22 Oct 2021.
3. Huggins C, Hodges CV. Studies on prostatic cancer: I. The effect of castration, of estrogen and of androgen injection on serum
phosphatases in metastatic carcinoma of the prostate. 1941. J Urol.
2002;168:9-12. Crossref
4. Harris WP, Mostaghel EA, Nelson PS, Montgomery B. Androgen
deprivation therapy: progress in understanding mechanisms of
resistance and optimizing androgen depletion. Nat Clin Pract Urol.
2009;6:76-85. Crossref
5. James ND, Sydes MR, Clarke NW, Mason MD, Dearnaley DP,
Spears MR, et al. Addition of docetaxel, zoledronic acid, or
both to first-line long-term hormone therapy in prostate cancer
(STAMPEDE): survival results from an adaptive, multiarm,
multistage, platform randomised controlled trial. Lancet.
2016;387:1163-77. Crossref
6. Clarke NW, Ali A, Ingleby FC, Hoyle A, Amos CL, Attard G, et al.
Addition of docetaxel to hormonal therapy in low- and high-burden
metastatic hormone sensitive prostate cancer: long-term survival
results from the STAMPEDE trial. Ann Oncol. 2019;30:1992-2003. Crossref
7. Sweeney CJ, Chen YH, Carducci M, Liu G, Jarrard DF,
Eisenberger M, et al. Chemohormonal therapy in metastatic
hormone-sensitive prostate cancer. N Engl J Med. 2015;373:737-46. Crossref
8. Kyriakopoulos CE, Chen YH, Carducci MA, Liu G, Jarrard DF,
Hahn NM, et al. Chemohormonal therapy in metastatic hormone-sensitive
prostate cancer: long-term survival analysis of the
randomized phase III E3805 CHAARTED trial. J Clin Oncol.
2018;36:1080-7. Crossref
9. Gravis G, Fizazi K, Joly F, Oudard S, Priou F, Esterni B, et al.
Androgen-deprivation therapy alone or with docetaxel in
non-castrate metastatic prostate cancer (GETUG-AFU 15): a
randomised, open-label, phase 3 trial. Lancet Oncol. 2013;14:149-58. Crossref
10. Gravis G, Boher JM, Joly F, Soulié M, Albiges L, Priou F, et al.
Androgen deprivation therapy (ADT) plus docetaxel versus ADT
alone in metastatic non castrate prostate cancer: impact of metastatic
burden and long-term survival analysis of the randomized phase 3
GETUG-AFU15 trial. Eur Urol. 2016;70:256-62. Crossref
11. Parker C, Gillessen S, Heidenreich A, Horwich A, ESMO
Guidelines Committee. Cancer of the prostate: ESMO Clinical
Practice Guidelines for diagnosis, treatment and follow-up. Ann
Oncol. 2015;26 Suppl 5:v69-77. Crossref
12. Fizazi K, Tran N, Fein L, Matsubara N, Rodriguez-Antolin A,
Alekseev BY, et al. Abiraterone plus prednisone in metastatic,
castration-sensitive prostate cancer. N Engl J Med. 2017;377:352-60. Crossref
13. Fizazi K, Tran N, Fein L, Matsubara N, Rodriguez-Antolin A,
Alekseev BY, et al. Abiraterone acetate plus prednisone in patients
with newly diagnosed high-risk metastatic castration-sensitive
prostate cancer (LATITUDE): final overall survival analysis
of a randomised, double-blind, phase 3 trial. Lancet Oncol.
2019;20:686-700. Crossref
14. James ND, de Bono JS, Spears MR, Clarke NW, Mason MD, Dearnaley DP, et al. Abiraterone for prostate cancer not previously treated with hormone therapy. N Engl J Med. 2017;377:338-51. Crossref
15. James N, Rush H, Clarke N, Attard G, Cook A, Dearnaley D, et al.
611O abiraterone acetate plus prednisolone for hormone-naïve
prostate cancer (PCa): long-term results from metastatic (M1)
patients in the STAMPEDE randomised trial (NCT00268476).
Ann Oncology. 2020;31 Suppl 4:S509. Crossref
16. Chi KN, Agarwal N, Bjartell A, Chung BH, Pereira de Santana
Gomes AJ, Given R, et al. Apalutamide for metastatic, castration-sensitive
prostate cancer. N Engl J Med. 2019;381:13-24. Crossref
17. Armstrong AJ, Szmulewitz RZ, Petrylak DP, Holzbeierlein J,
Villers A, Azad A, et al. ARCHES: A randomized, phase III study
of androgen deprivation therapy with enzalutamide or placebo in
men with metastatic hormone-sensitive prostate cancer. J Clin
Oncol. 2019;37:2974-86. Crossref
18. Davis ID, Martin AJ, Stockler MR, Begbie S, Chi KN, Chowdhury S, et al. Enzalutamide with standard first-line therapy
in metastatic prostate cancer. N Engl J Med. 2019;381:121-31. Crossref
19. Parker C, Castro E, Fizazi K, Heidenreich A, Ost P, Procopio G,
et al. Prostate cancer: ESMO Clinical Practice Guidelines for
diagnosis, treatment and follow-up. Ann Oncol. 2020;31:1119-34. Crossref
20. Hutton B, Salanti G, Caldwell DM, et al. The PRISMA extension
statement for reporting of systematic reviews incorporating
network meta-analyses of health care interventions: checklist and
explanations. Ann Intern Med. 2015;162:777-84. Crossref
21. Cochrane Training. Cochrane Handbook for Systematic Reviews
of Interventions version 6.1 (updated September 2020). Cochrane.
2020. Available from: http://www.training.cochrane.org/handbook.
Accessed 22 Oct 2021.
22. Rücker G, Schwarzer G. Ranking treatments in frequentist network
meta-analysis works without resampling methods. BMC Med Res Methodol. 2015;15:58. Crossref
23. Lombard AP, Liu L, Cucchiara V, Liu C, Armstrong CM, Zhao R,
et al. Intra versus inter cross-resistance determines treatment
sequence between taxane and AR-targeting therapies in advanced
prostate cancer. Mol Cancer Ther. 2018;17:2197-205. Crossref
24. Khalaf D, Annala M, Finch DL, Oja CD, Vergidis J, Zulfiqar M,
et al. Phase 2 randomized cross-over trial of abiraterone +
prednisone (ABI+P) vs enzalutamide (ENZ) for patients (pts) with
metastatic castration resistant prostate cancer (mCPRC): Results for
2nd-line therapy. J Clin Oncol. 2018;36(15 Suppl):5015. Crossref
25. Rush HL, Cook AD, Brawley CD, Murphy L, Macnair A,
Millman R, et al. Comparative quality of life in patients randomized
contemporaneously to docetaxel or abiraterone in the STAMPEDE
trial. J Clin Oncol. 2020;38(6 Suppl):14. Crossref
26. Khalaf DJ, Sunderland K, Eigl BJ, Kollmannsberger CK,
Ivanov N, Finch DL, et al. Health-related quality of life for
abiraterone plus prednisone versus enzalutamide in patients with
metastatic castration-resistant prostate cancer: results from a phase
ii randomized trial. Eur Urol. 2019;75:940-7. Crossref
27. Wang L, Paller CJ, Hong H, De Felice A, Alexander GC,
Brawley O. Comparison of systemic treatments for metastatic
castration-sensitive prostate cancer: a systematic review and
network meta-analysis. JAMA Oncol. 2021;7:412-20. Crossref
28. So TH, Chiang CL, Lam TC, Chan FT, Choi HH. What is the best
first-line therapy for metastatic castration-sensitive prostate cancer
in 2019? A network meta-analysis. Ann Oncol. 2019;30 (Suppl
9):ix69. Crossref
29. Sathianathen NJ, Koschel S, Thangasamy IA, Teh J, Alghazo O,
Butcher G, et al. Indirect comparisons of efficacy between
combination approaches in metastatic hormone-sensitive prostate
cancer: a systematic review and network meta-analysis. Eur Urol.
2020;77:365-72. Crossref