Pathologies and Postoperative Features of Posterior Tibial Tendon Dysfunction: A Pictorial Essay
PICTORIAL ESSAY
Hong Kong J Radiol 2024 Mar;27(1):e51-64 | Epub 22 March 2024
Pathologies and Postoperative Features of Posterior Tibial Tendon Dysfunction: A Pictorial Essay
RYS Mak, JHM Cheng, KH Chin, CY Chu
Department of Radiology, Pamela Youde Nethersole Eastern Hospital, Hong Kong SAR, China
Correspondence: Dr RYS Mak, Department of Radiology, Pamela Youde Nethersole Eastern Hospital, Hong Kong SAR, China. Email: mys877@ha.org.hk
Submitted: 18 November 2022; Accepted: 13 March 2023.
Contributors: All authors designed the study. RYSM and JHMC acquired and analysed the data. RYSM, JHMC and KHC drafted the
manuscript. JHMC, KHC and CYC critically revised the manuscript for important intellectual content. All authors had full access to the data,
contributed to the study, approved the final version for publication, and take responsibility for its accuracy and integrity.
Conflicts of Interest: All authors have disclosed no conflicts of interest.
Funding/Support: The study received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Data Availability: All data generated or analysed during the present study are available from the corresponding author on reasonable request.
Declaration: Part of the study was previously presented as a poster in the 30th Annual Scientific Meeting of Hong Kong College of Radiologists (12-13 November 2022, virtual).
Ethics Approval: The study was approved by the Hong Kong East Cluster Research Ethics Committee of Hospital Authority, Hong Kong (Ref No.: HKECREC-2022-043). The patients were treated in accordance with the tenets of the Declaration of Helsinki. The requirement for patient
consent was waived by the Committee due to the retrospective and descriptive nature of the study.
INTRODUCTION
The posterior tibial tendon (PTT) is the largest tendon
in the medial compartment of the ankle and the most
important dynamic stabiliser of the longitudinal
foot arch.[1] It is also the most common site of tendon
abnormalities in the medial ankle, where dysfunction
would result in a cascade of failures of other secondary
supporting structures, eventually leading to collapse of
the longitudinal arch and acquired pes planus deformity.[1]
Knowledge of the imaging appearance of posterior tibial
tendon dysfunction (PTTD) enables early diagnosis and
treatment, hence preventing progression to fixed pes
planus deformity.
ANATOMY AND PATHOPHYSIOLOGY
The PTT runs posterior to the medial malleolus and
inserts mainly on the medial aspect of the navicular, with
minor slips inserting into the cuneiforms and the first to
fourth metatarsals.[1] As the tendon passes posterior to the axis of the ankle joint and medial to the subtalar joint, it
acts as a plantar flexor and invertor of the foot, as well as
the adductor of the forefoot at the midtarsal joint.[2] [3] [4]
PTTD is a spectrum of pathologies ranging from
tenosynovitis, tendonitis, and partial tear to complete
rupture.[5] It is most prevalent in middle- to old-aged
females,[5] commonly resulting from chronic degeneration
but may also be caused by acute trauma or by conditions
with alterations of anatomy, mechanical forces, and
tendon vascularity. Risk factors include pre-existing pes
planus, obesity, hypertension, chronic steroid use, gout,
and inflammatory arthropathies such as rheumatoid
arthritis.[3] [6] Ischaemia and mechanical stress are the
main underlying pathophysiologies of most PTTD.[6]
In particular, the PTT around the level of the medial
malleolus is the most susceptible site due to a number
of factors.[6] First, the mid tendon has relatively poor
vascular supply; second, the synovial sheath ends at the
midportion of the talus, distal to which the mesotendon is absent; third, the PTT curves around the medial
malleolus, creating a focal point of mechanical strain.[6]
The second most common location for tears is at the
distal portion of the tendon.[6]
In PTTD, the tendon’s normal antagonist, the peroneus
brevis, acts as a deforming factor in foot eversion,
leading to hindfoot valgus and forefoot varus
deformities. Progression to arch collapse is associated
with tension on the spring and deltoid ligaments and the
talonavicular capsule. Hindfoot valgus also leads to an
eversion force of the Achilles tendon on the calcaneus,
leading to equinus deformity due to shortening of this
tendon.[2] [4] [7] [8] [9]
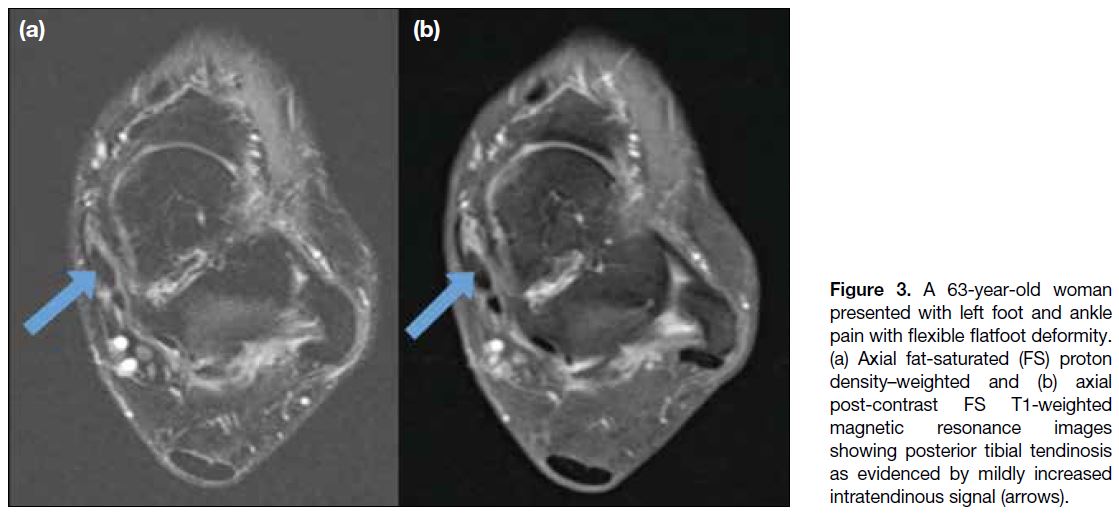
CLASSIFICATION
Johnson and Strom’s four-stage classification is a widely adopted clinical staging system for PTTD, which serves
as a guide to management.[10] The clinical findings and
corresponding magnetic resonance imaging (MRI)
features of PTTD are described in the Table.[10] [11] [12]
Table. Clinical staging and corresponding magnetic resonance imaging findings for posterior tibial tendon dysfunction.[10] [11] [12]
PRIMARY IMAGING FINDINGS
Tenosynovitis
Tenosynovitis can be caused by mechanical trauma
or inflammatory arthropathy such as seronegative
spondyloarthropathies, rheumatoid arthritis, systemic
lupus erythematosus, and gout.[3] [6] [13] Tenosynovitis
manifests radiologically as an abnormal amount of fluid in the tendon sheath on both MRI and ultrasound
(Figures 1 and 2). The tendon itself appears normal at
this early stage with normal size and signal intensity on
MRI.[5] On ultrasound, increased flow in the peritendon
area and hypoechoic tissue around the tendon may be
detected.[14]
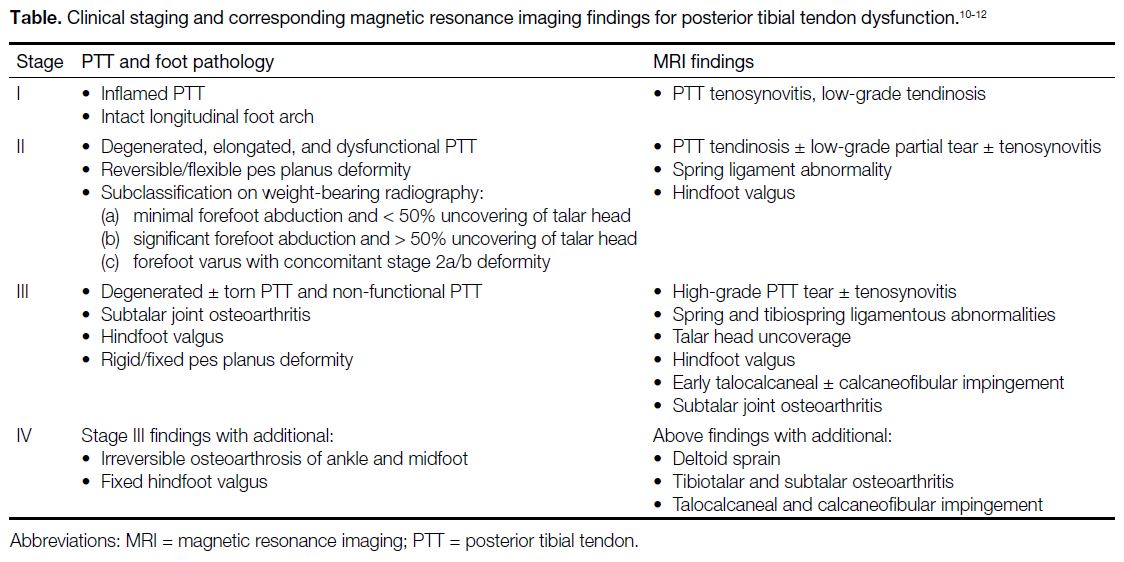
Figure 1. A 25-year-old man
with known seronegative
spondyloarthropathy complained
of right ankle pain. (a, b) Axial
fat-saturated proton density–weighted magnetic resonance
images showing mild tenosynovial
effusion along the posterior tibial
tendon (arrows), suggestive of
mild tenosynovitis. There was also
Achilles tendinosis (not shown).
His symptoms improved after
starting on golimumab.
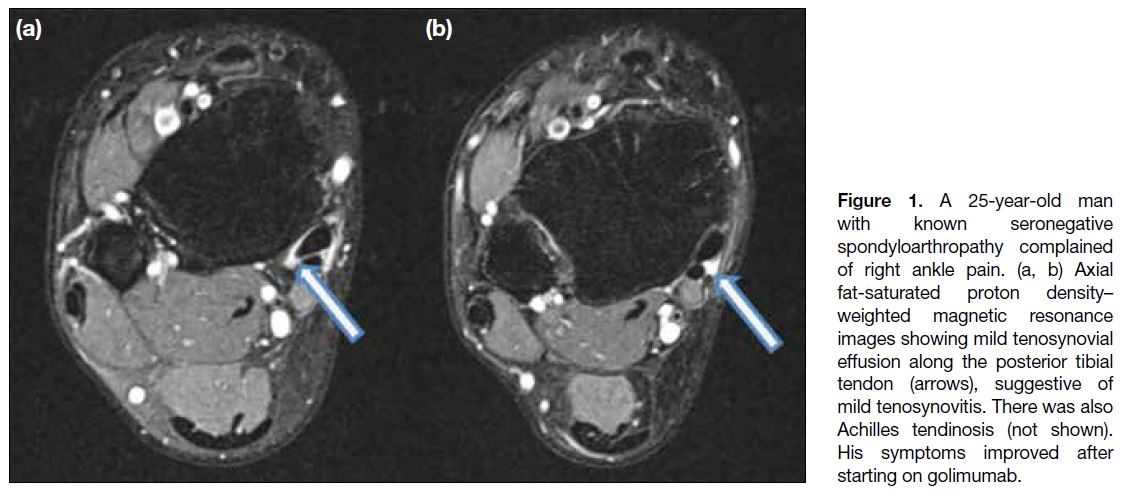
Figure 2. A 10-year-old girl with enthesitis-related arthritis (HLA-B27–positive) presented with right medial ankle pain. (a, b) Short-axis
ultrasound (US) images of the posterior tibial tendon (PTT) showing effusion (blue and white arrow) around the tendon, which is thickened
with a moderate increase in vascularity within the tendon (green arrow), in the tendon sheath (arrowheads), and adjacent soft tissue (curved
arrow). (c) Long-axis US image of the PTT showing similar findings, with effusion (blue and white arrow) and increased vascularity in the
tendon (green arrow) and the adjacent soft tissue (curved arrow). Findings are suggestive of tenosynovitis and tendinosis.
Tendinosis
The next phase of the disease is tendinosis, with
collagen degeneration, local necrosis, calcification,
and hypocellularity.[6] On MRI, the normal PTT should
appear black on all spin-echo images[5] and is about twice
the size of the adjacent round flexor digitorum longus
(FDL) and flexor hallucis longus tendons.[1] In posterior
tibial tendinosis, the tendon appears thickened with
normal or increased signal intensity on both T1- and
proton density–weighted images (Figure 3); these
intrasubstance signals represent severe degeneration or
intrasubstance tears not reaching the tendon surface, thus
there is a certain degree of overlap of the appearance of
tendinosis and partial tear.[5] Contrast enhancement of the
tendon is also a common finding in tendinosis.[14]
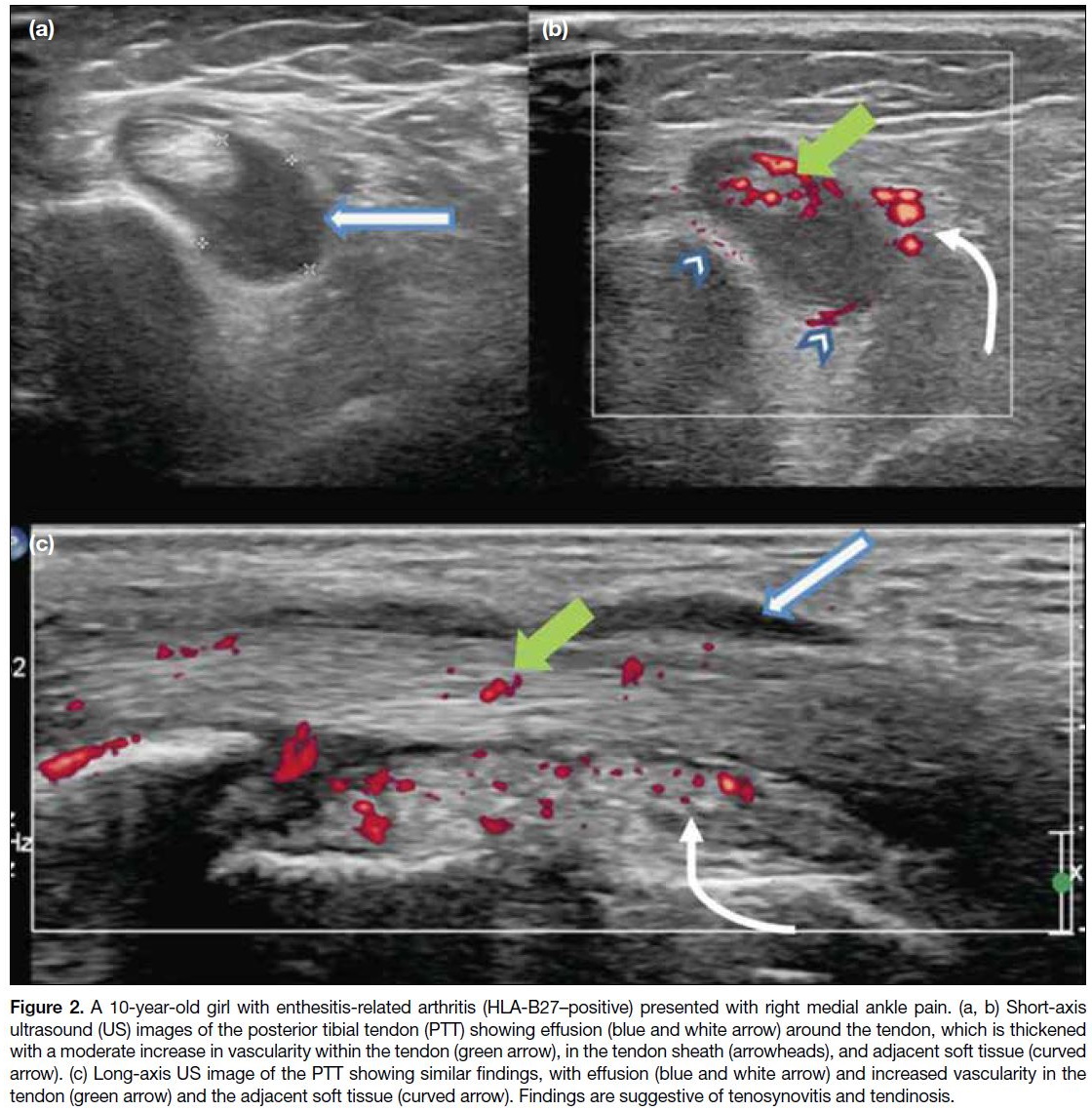
Figure 3. A 63-year-old woman
presented with left foot and ankle
pain with flexible flatfoot deformity.
(a) Axial fat-saturated (FS) proton
density–weighted and (b) axial
post-contrast FS T1-weighted
magnetic resonance images
showing posterior tibial tendinosis
as evidenced by mildly increased
intratendinous signal (arrows).
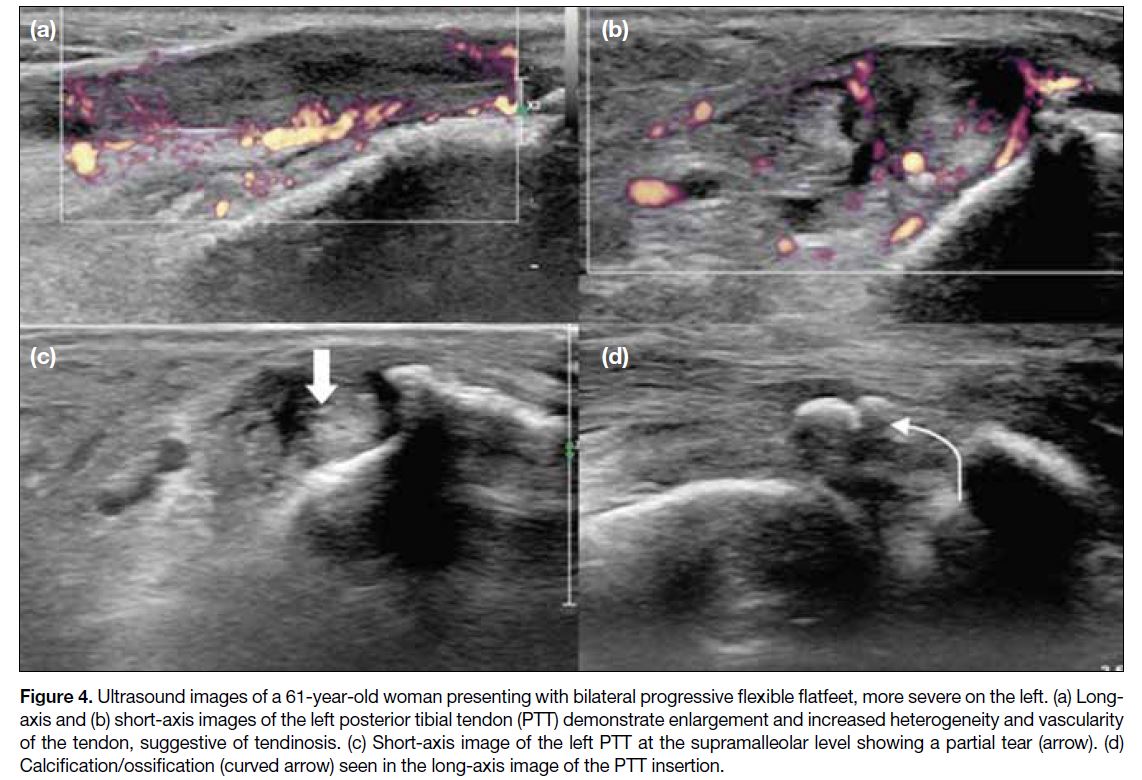
On sonography, the PTT normally shows homogeneous
echogenic longitudinal fibres with no tendinous
or peritendinous vascularity.[14] In tendinosis, the
tendon is enlarged and appears inhomogeneous, with
internal vascular flow sometimes detected on Doppler
examination (Figure 4).[14]
Figure 4. Ultrasound images of a 61-year-old woman presenting with bilateral progressive flexible flatfeet, more severe on the left. (a) Long-axis
and (b) short-axis images of the left posterior tibial tendon (PTT) demonstrate enlargement and increased heterogeneity and vascularity
of the tendon, suggestive of tendinosis. (c) Short-axis image of the left PTT at the supramalleolar level showing a partial tear (arrow). (d)
Calcification/ossification (curved arrow) seen in the long-axis image of the PTT insertion.
Partial and Complete Tears
In chronic tenosynovitis or tendinosis, the PTT is
weakened, and tears of the tendon can occur. They can
be classified into three types according to Rosenberg
et al.[15] Type 1 is a partial tear with associated tendon
hypertrophy. On MRI, the tendon is thickened, appearing
rounded with loss of its normal oval shape and shows
increased linear or heterogenous intrasubstance signal in
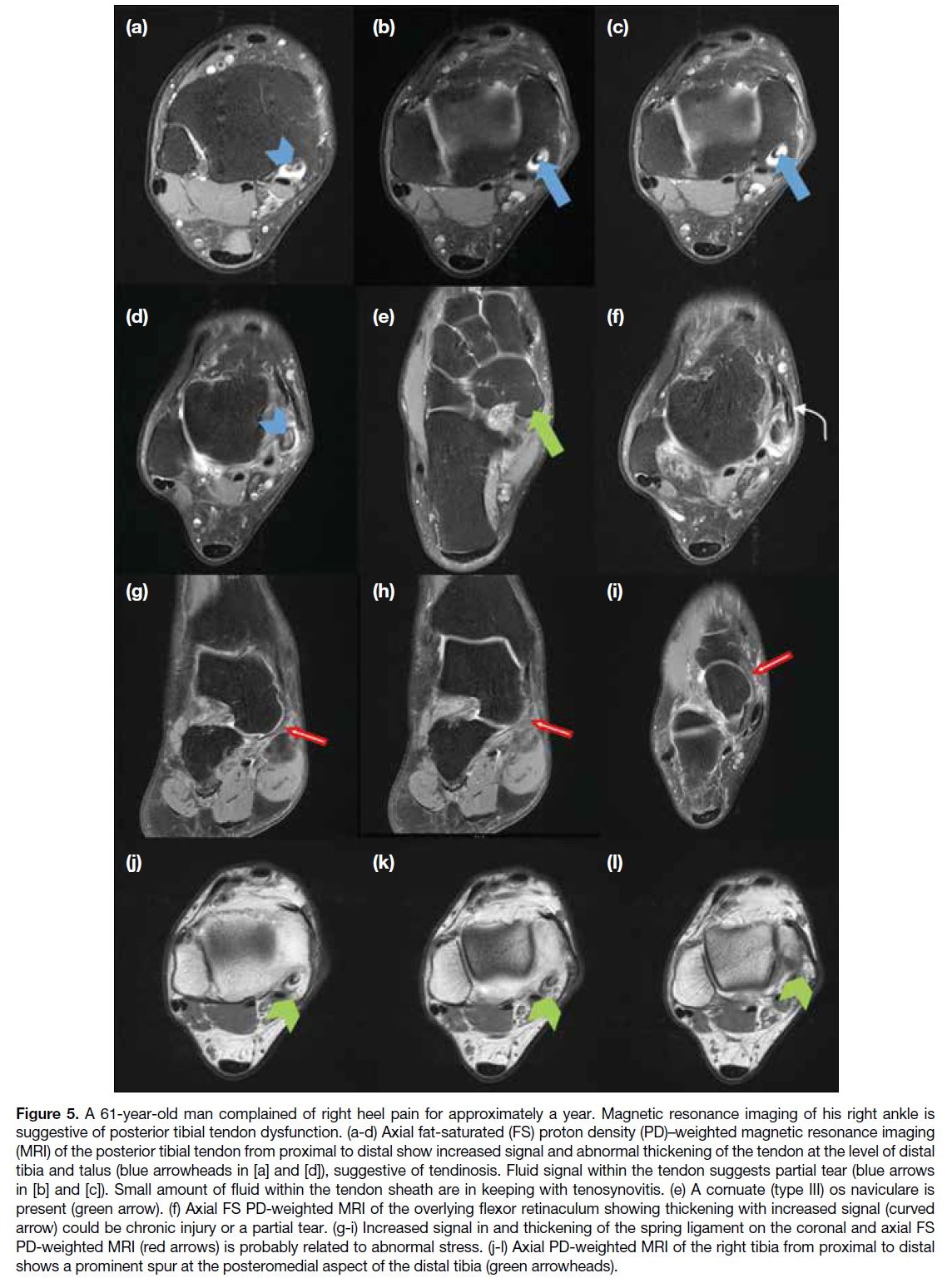
all sequences, which represents internal tears (Figure 5).[5]
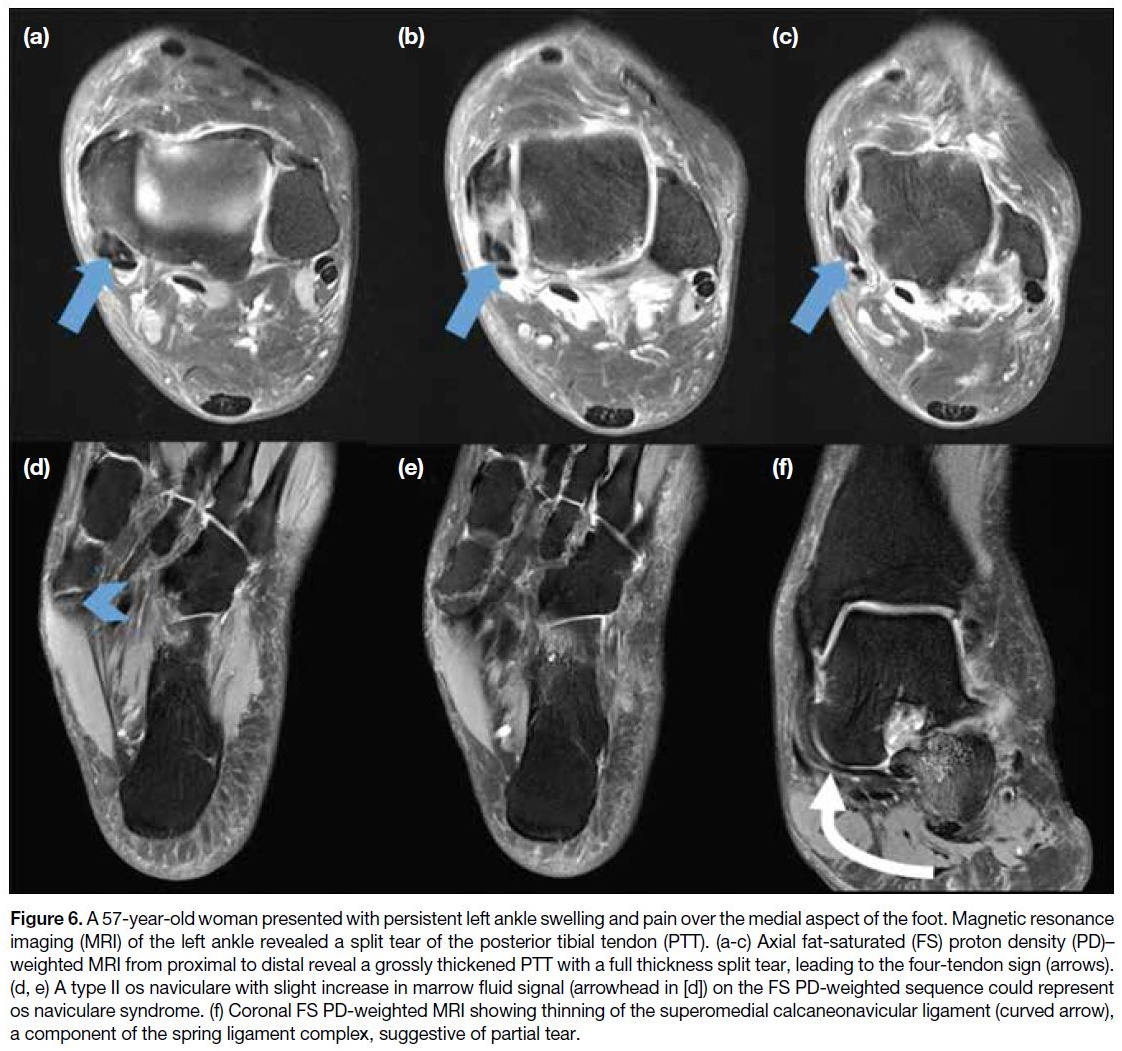
A severe form of this type of injury is a longitudinal split
of the PTT into two separate parts, which together with
the adjacent FDL and flexor hallucis longus tendons,
give rise to the appearance of four medial ankle tendons,
known as the four-tendon sign (Figure 6a-c).[13] Type 2 is
a more severe partial tear with reduced tendon calibre
and further increase in internal signal.[5] The segment
proximal and distal to the segment of thinned tendon
is frequently hypertrophied, which may be due to the
background chronic type 1 injury.[5] [13] Type 3 injury is a
complete tear characterised by a visible gap in the tendon
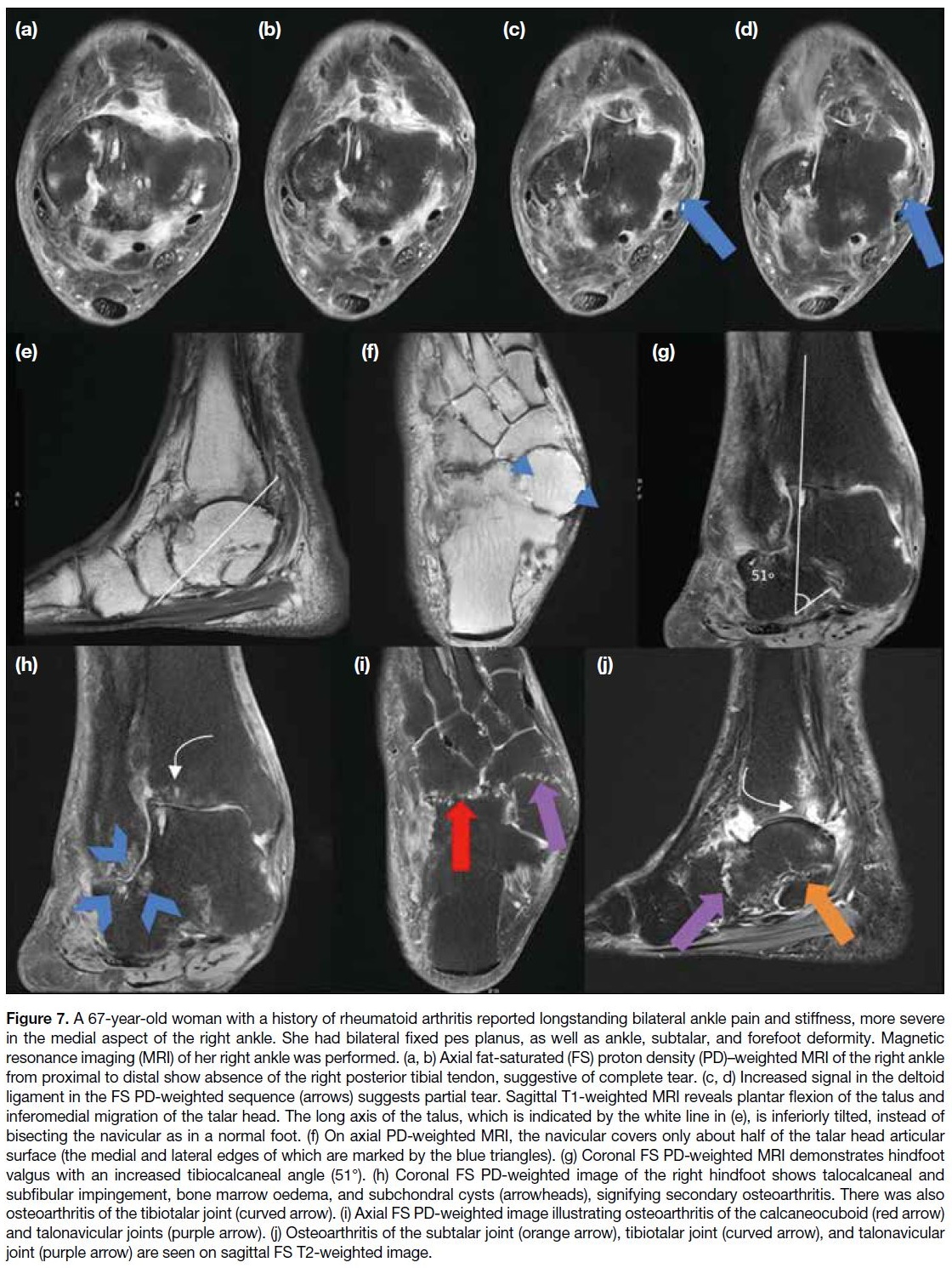
within the tendon sheath (Figure 7).[5]
Figure 5. A 61-year-old man complained of right heel pain for approximately a year. Magnetic resonance imaging of his right ankle is
suggestive of posterior tibial tendon dysfunction. (a-d) Axial fat-saturated (FS) proton density (PD)–weighted magnetic resonance imaging
(MRI) of the posterior tibial tendon from proximal to distal show increased signal and abnormal thickening of the tendon at the level of distal
tibia and talus (blue arrowheads in [a] and [d]), suggestive of tendinosis. Fluid signal within the tendon suggests partial tear (blue arrows
in [b] and [c]). Small amount of fluid within the tendon sheath are in keeping with tenosynovitis. (e) A cornuate (type III) os naviculare is
present (green arrow). (f) Axial FS PD-weighted MRI of the overlying flexor retinaculum showing thickening with increased signal (curved
arrow) could be chronic injury or a partial tear. (g-i) Increased signal in and thickening of the spring ligament on the coronal and axial FS
PD-weighted MRI (red arrows) is probably related to abnormal stress. (j-l) Axial PD-weighted MRI of the right tibia from proximal to distal
shows a prominent spur at the posteromedial aspect of the distal tibia (green arrowheads).
Figure 6. A 57-year-old woman presented with persistent left ankle swelling and pain over the medial aspect of the foot. Magnetic resonance
imaging (MRI) of the left ankle revealed a split tear of the posterior tibial tendon (PTT). (a-c) Axial fat-saturated (FS) proton density (PD)–weighted MRI from proximal to distal reveal a grossly thickened PTT with a full thickness split tear, leading to the four-tendon sign (arrows).
(d, e) A type II os naviculare with slight increase in marrow fluid signal (arrowhead in [d]) on the FS PD-weighted sequence could represent
os naviculare syndrome. (f) Coronal FS PD-weighted MRI showing thinning of the superomedial calcaneonavicular ligament (curved arrow),
a component of the spring ligament complex, suggestive of partial tear.
Figure 7. A 67-year-old woman with a history of rheumatoid arthritis reported longstanding bilateral ankle pain and stiffness, more severe
in the medial aspect of the right ankle. She had bilateral fixed pes planus, as well as ankle, subtalar, and forefoot deformity. Magnetic
resonance imaging (MRI) of her right ankle was performed. (a, b) Axial fat-saturated (FS) proton density (PD)–weighted MRI of the right ankle
from proximal to distal show absence of the right posterior tibial tendon, suggestive of complete tear. (c, d) Increased signal in the deltoid
ligament in the FS PD-weighted sequence (arrows) suggests partial tear. Sagittal T1-weighted MRI reveals plantar flexion of the talus and
inferomedial migration of the talar head. The long axis of the talus, which is indicated by the white line in (e), is inferiorly tilted, instead of
bisecting the navicular as in a normal foot. (f) On axial PD-weighted MRI, the navicular covers only about half of the talar head articular
surface (the medial and lateral edges of which are marked by the blue triangles). (g) Coronal FS PD-weighted MRI demonstrates hindfoot
valgus with an increased tibiocalcaneal angle (51°). (h) Coronal FS PD-weighted image of the right hindfoot shows talocalcaneal and
subfibular impingement, bone marrow oedema, and subchondral cysts (arrowheads), signifying secondary osteoarthritis. There was also
osteoarthritis of the tibiotalar joint (curved arrow). (i) Axial FS PD-weighted image illustrating osteoarthritis of the calcaneocuboid (red arrow)
and talonavicular joints (purple arrow). (j) Osteoarthritis of the subtalar joint (orange arrow), tibiotalar joint (curved arrow), and talonavicular
joint (purple arrow) are seen on sagittal FS T2-weighted image.
SECONDARY AND ASSOCIATED IMAGING FINDINGS
Accessory Navicular Bones
There are three types of accessory navicular bones. Type
I is a 2- to 3-mm sesamoid bone in the PTT separated
from the navicular bone; type II is connected to the
navicular bone by a thin layer of cartilage (Figures 6d-e and 8a); type III is a prominent protuberance fused with
the navicular tuberosity (Figure 5e).[3] Types II and III may be associated with a younger onset of midfoot pain
and planovalgus foot,[3] which are associated with a more
proximal insertion of the PTT, straightening the distal
tendon curve.[6] [16] As a result, there is more focal frictional
wear and tear on the tendon at the medial malleolus.[6] [16]
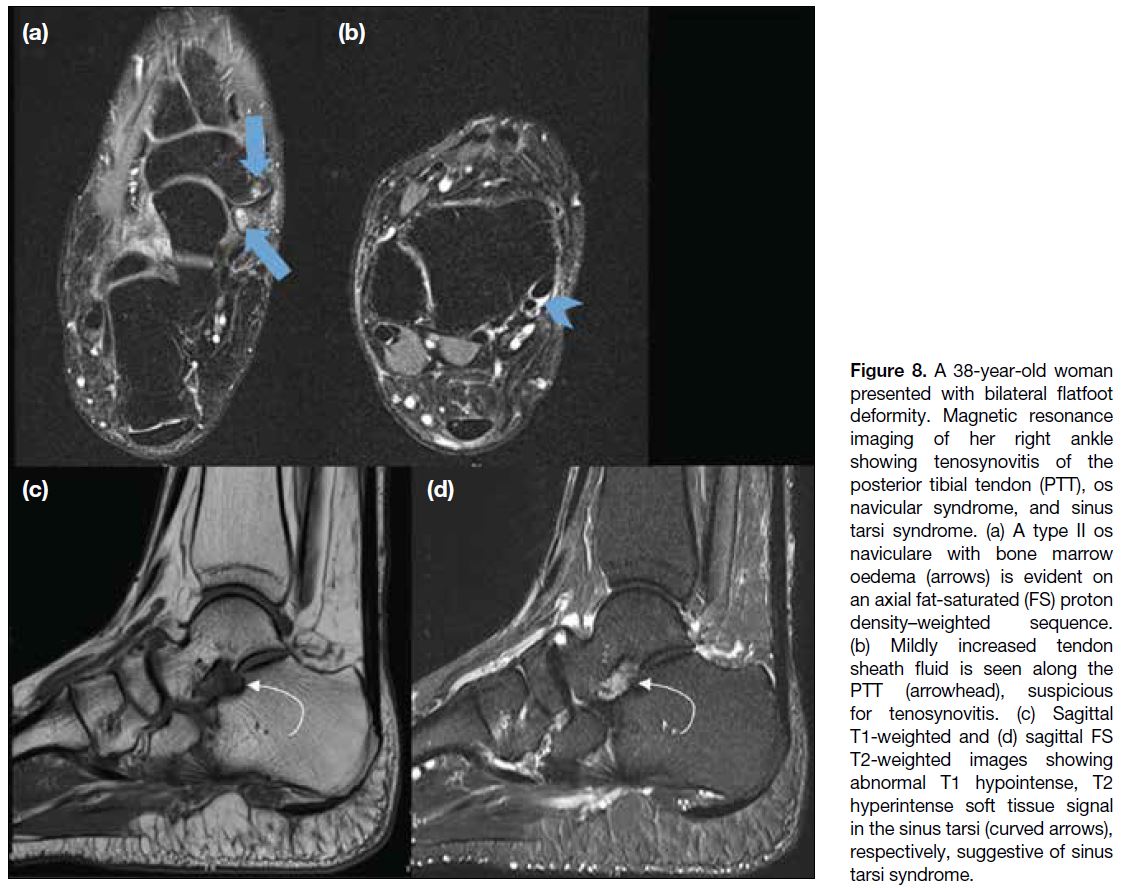
Figure 8. A 38-year-old woman
presented with bilateral flatfoot
deformity. Magnetic resonance
imaging of her right ankle
showing tenosynovitis of the
posterior tibial tendon (PTT), os
navicular syndrome, and sinus
tarsi syndrome. (a) A type II os
naviculare with bone marrow
oedema (arrows) is evident on
an axial fat-saturated (FS) proton
density–weighted sequence.
(b) Mildly increased tendon
sheath fluid is seen along the
PTT (arrowhead), suspicious
for tenosynovitis. (c) Sagittal
T1-weighted and (d) sagittal FS
T2-weighted images showing
abnormal T1 hypointense, T2
hyperintense soft tissue signal
in the sinus tarsi (curved arrows),
respectively, suggestive of sinus
tarsi syndrome.
MRI features include bone marrow oedema in the
accessory navicular bone and the adjacent tuberosity
(Figures 6d-e and 8a), fluid in the synchondrosis, soft
tissue swelling, adventitial bursa formation, and fracture
of the synchondrosis from the chronic pulling of the
PTT.[3] [6]
Tibial Spur
Osteophytes may develop at the posteromedial aspect of
the medial malleolus adjacent to the tendon, presumably
due to reactive periostitis in chronic tears (Figure 5j-l).[5] [15]
Spring Ligament Injury
As the PTT weakens, it fails to invert the hindfoot and
lock the transverse tarsal joints, causing the force of
the gastrocnemius-soleus muscle complex to act at the
talonavicular joint rather than the metatarsal joints.[17]
This leads to increased stress on the talar head and injury
to the spring ligament.[17] On MRI, insufficiency of the
spring ligament is characterised by thickening (> 6 mm)
and increased signal heterogeneity on proton density–weighted images (Figures 5g-i and 6f).[18]
Sinus Tarsi Syndrome
Increased plantar flexion of the talus and hindfoot valgus due to PTTD also increases the stress on the
interosseous talocalcaneal ligaments and cervical
ligaments within the sinus tarsi, causing insufficiency
of these ligaments over time.[3] [18] On MRI, the normal fat
signal around the ligament in the sinus tarsi is replaced
by abnormal tissue that shows T1 hypointensity and T2
hyperintensity (Figure 8). When fibrotic change within
the abnormal tissue predominates, the signal may show
T1 and T2 hypointensity.[3] [18] Other imaging features
include partial or complete tears of the tarsal sinus ligaments.[3]
Plantar Fasciitis
Plantar fasciitis has been found to have a low association
with advanced PTT injury, due to increased strain on the
plantar fascia which is responsible for supporting the
longitudinal arch.[3] [18] MRI shows thickening (> 4 mm),
irregularity or increased heterogenous signal within the
fascia, associated with perifascial and bone marrow
oedema.[3]
Deltoid Ligament Complex Injury
The deltoid ligament complex consists of deep and
superficial layers. The deep layer opposes ankle valgus and stabilises the tibiotalar articulation, while
the superficial ligaments limit hindfoot eversion and
inward displacement of the talar head to stabilise the talonavicular joint.[3] Insufficiency of the deltoid ligament
complex is seen in late stage of PTTD.[18] On MRI, low-grade
sprain causes amorphous signal with loss of the
normal striated appearance (Figure 7c and 7d), while
high-grade injuries manifest as fluid-filled gaps or even
discontinuity of the ligament.[3]
Bony Malalignment
Dysfunction of the PTT causes tension and eventual
failure of the secondary supporting structures including
the spring ligament, tarsal sinus ligaments, and deltoid
ligaments. The biomechanics of the foot are altered, causing a cascade of foot deformities.
Pes Planus
Collapse of the medial longitudinal arch results in pes
planus, which can be demonstrated by a reduction
in the calcaneal inclination angle on lateral weight-bearing
radiographs.[19] The calcaneal inclination angle is
drawn between the plane of support and the calcaneal
inclination axis (i.e., the line connecting the most inferior
point of the calcaneal tuberosity with the most distal and
inferior point of the calcaneus along the calcaneocuboid
joint) [Figure 9].[19] [20] The normal range is 20° to 30°.[19] [20]
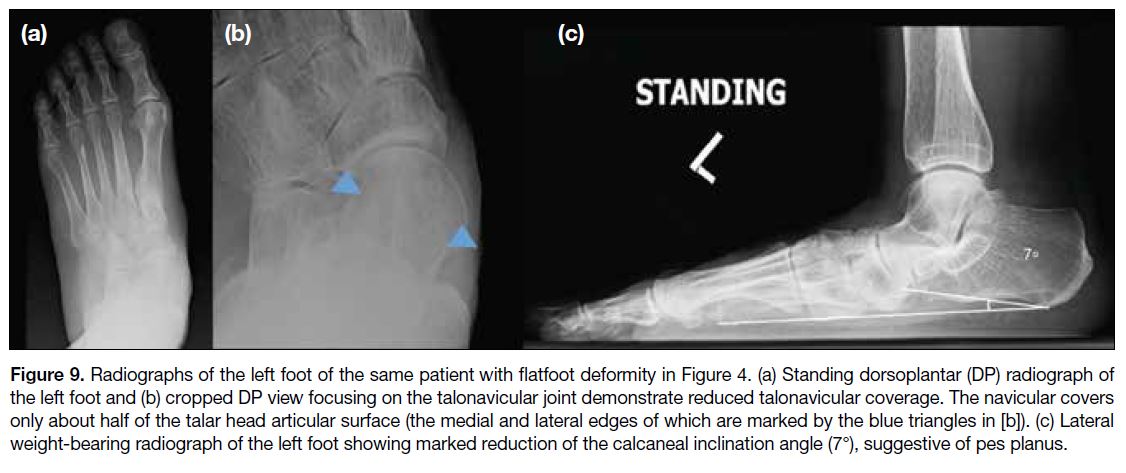
Figure 9. Radiographs of the left foot of the same patient with flatfoot deformity in Figure 4. (a) Standing dorsoplantar (DP) radiograph of
the left foot and (b) cropped DP view focusing on the talonavicular joint demonstrate reduced talonavicular coverage. The navicular covers
only about half of the talar head articular surface (the medial and lateral edges of which are marked by the blue triangles in [b]). (c) Lateral
weight-bearing radiograph of the left foot showing marked reduction of the calcaneal inclination angle (7°), suggestive of pes planus.
Talonavicular Fault
As pes planus progresses, there is excessive plantar
flexion of the talus, resulting in talonavicular fault
which can be demonstrated on weight-bearing lateral
radiographs of the foot or sagittal MRI.[6] [19] On sagittal
images including the base of the first metatarsal, a long
axis drawn along the talus should normally divide the
navicular into equal superior and inferior halves. If the
line is inferiorly positioned, it suggests talonavicular
fault (Figure 7e).[6]
Talar Head Uncoverage
The unopposed pull of the peroneus brevis abducts
the forefoot and causes lateral subluxation of the
talonavicular joint and uncoverage of the talus.[6] [16] [19] In
a normal foot, most (75%-100%) of the talar head is
covered by the navicular; in flatfoot deformity, the talar
head becomes more uncovered (Figures 7f and 9b).[21]
Hindfoot Valgus
The degree of hindfoot valgus can be measured on
radiographs or on the most posterior coronal image that
includes the tibia and calcaneus on MRI, by measuring
the angle between the long axis of the tibia and a line
along the medial calcaneal wall (Figure 7g), with the
normal range measuring 0° to 6°.[22]
Lateral Hindfoot Impingement
As hindfoot valgus progresses, lateral hindfoot
impingement, including talocalcaneal impingement
and/or subfibular impingement, may occur (Figure 7h).[18] On MRI, talocalcaneal impingement causes bone
marrow oedema, cysts, and sclerosis at the site where
the lateral talar process impinges on the lateral cortex
of the calcaneus.[18] Common findings in subfibular
impingement include low signal on T1-weighted imaging
and predominantly low signal on T2-weighted imaging
from soft tissue entrapment between the distal fibula and
calcaneus, direct osseous contact between calcaneus and
fibula, and distal fibular oedema.[18] [22]
Secondary Osteoarthritis
In late-stage disease, chronic uneven stress and bony
malalignment result in secondary osteoarthritis of the
subtalar, talonavicular, calcaneocuboid, and tibiotalar joints.[10]
MANAGEMENT
Conservative treatment is the first-line treatment for
PTTD and is indicated before operative treatment
is considered. It involves treatment of the acute inflammation in the early stages, physiotherapy and
accommodation of the deformity in late stages, including
appropriate footwear (e.g., flat lace-up shoes) and
orthosis.[7] [23] [24]
Operative treatment is indicated if conservative
treatment fails, typically in Stages II to IV diseases
and less frequently in Stage I disease, with reported
good to excellent outcome in > 80% of the patients at
5 years’ follow-up. A wide variety of surgical options
have been reported; in general, they consist of different
combinations of soft tissue procedures addressing
the PTTD and osseous procedures addressing the
malalignment. A few commonly performed procedures
are discussed below.
In early PTTD (Stages I and II diseases), surgical release, simple tenosynovectomy and tendon debridement
are indicated in cases of mild tendon inflammation,
which have been reported to be helpful in pain relief.[25]
However, combined procedures including soft tissue
reconstructions (e.g., tendon transfer and side-to-side
anastomosis) and bony procedures are often performed
to address the PTTD and osseous deformities.[23]
In more advanced Stage II disease, tendon transfer is
often the choice of treatment (Figures 10 and 11).[3] The
FDL tendon is the most commonly transferred tendon,
which is transferred to replicate the function of the PTT
by augmenting hindfoot inversion strength and reducing
adduction across the tarsal joint.[23] A calcaneal osteotomy
is often performed at the same time to correct hindfoot
valgus and medialise the pulling force of the Achilles
tendon.[3] [23] [25]
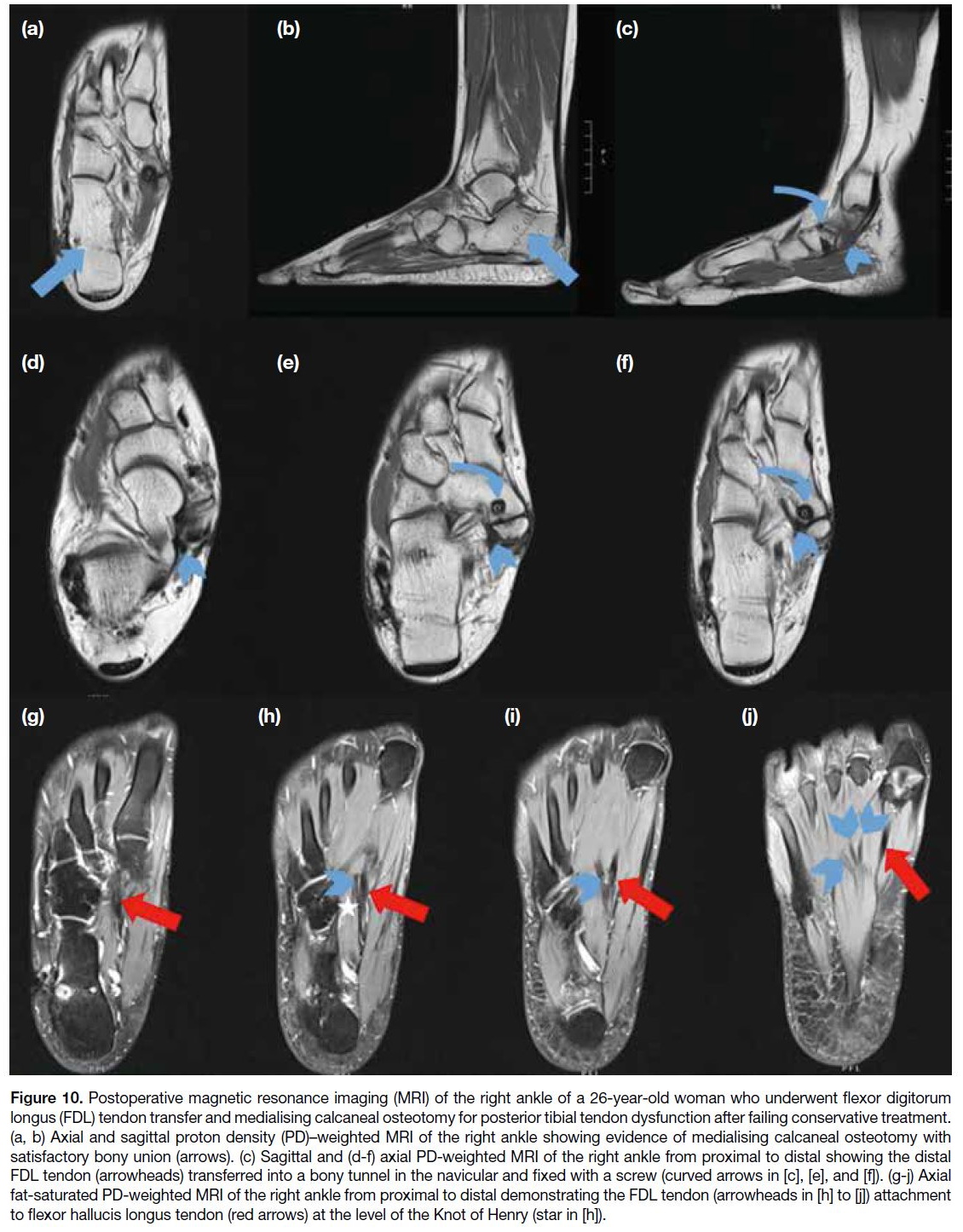
Figure 10. Postoperative magnetic resonance imaging (MRI) of the right ankle of a 26-year-old woman who underwent flexor digitorum
longus (FDL) tendon transfer and medialising calcaneal osteotomy for posterior tibial tendon dysfunction after failing conservative treatment.
(a, b) Axial and sagittal proton density (PD)–weighted MRI of the right ankle showing evidence of medialising calcaneal osteotomy with
satisfactory bony union (arrows). (c) Sagittal and (d-f) axial PD-weighted MRI of the right ankle from proximal to distal showing the distal
FDL tendon (arrowheads) transferred into a bony tunnel in the navicular and fixed with a screw (curved arrows in [c], [e], and [f]). (g-j) Axial
fat-saturated PD-weighted MRI of the right ankle from proximal to distal demonstrating the FDL tendon (arrowheads in [h] to [j]) attachment
to flexor hallucis longus tendon (red arrows) at the level of the Knot of Henry (star in [h]).
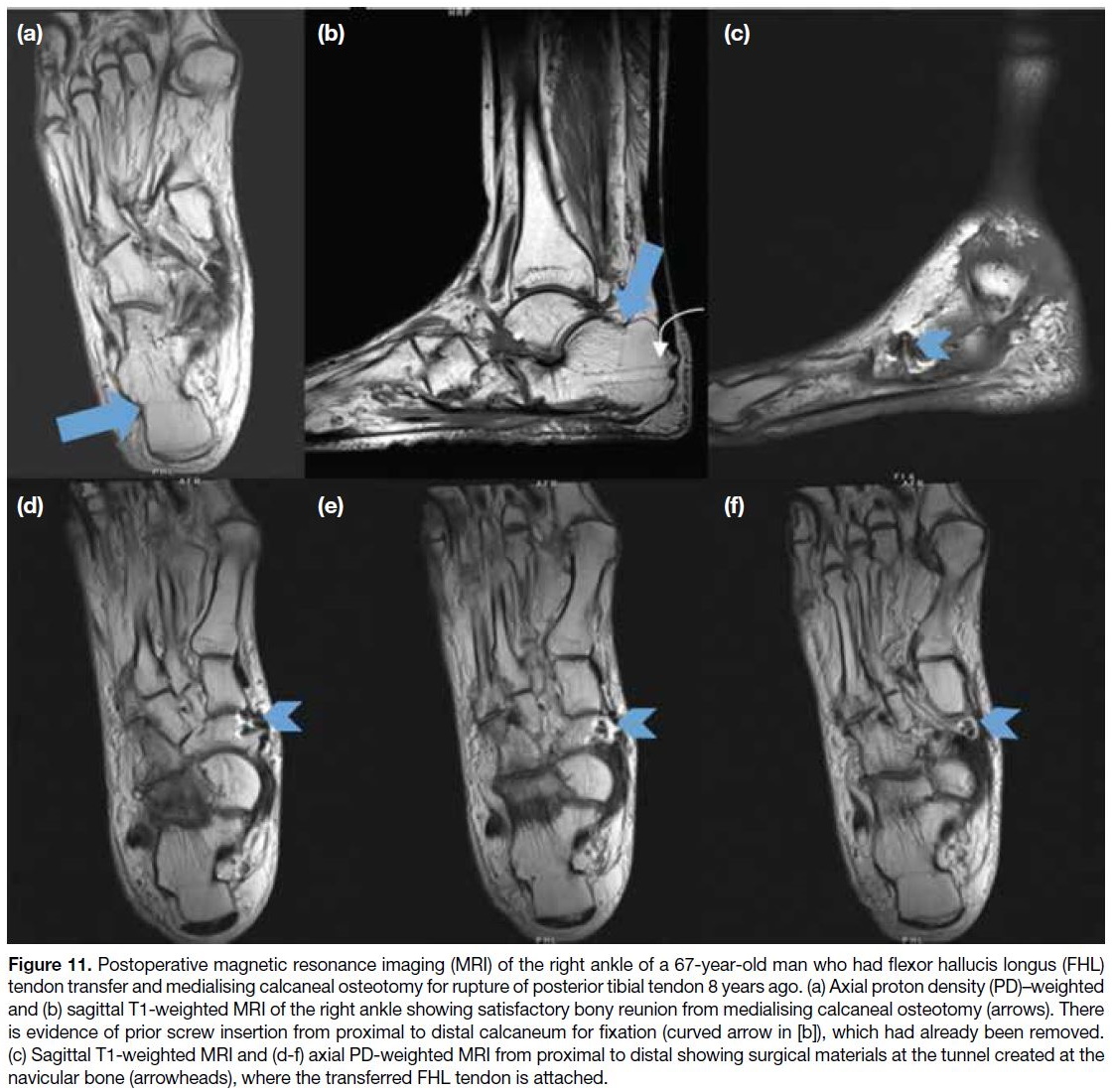
Figure 11. Postoperative magnetic resonance imaging (MRI) of the right ankle of a 67-year-old man who had flexor hallucis longus (FHL)
tendon transfer and medialising calcaneal osteotomy for rupture of posterior tibial tendon 8 years ago. (a) Axial proton density (PD)–weighted
and (b) sagittal T1-weighted MRI of the right ankle showing satisfactory bony reunion from medialising calcaneal osteotomy (arrows). There
is evidence of prior screw insertion from proximal to distal calcaneum for fixation (curved arrow in [b]), which had already been removed.
(c) Sagittal T1-weighted MRI and (d-f) axial PD-weighted MRI from proximal to distal showing surgical materials at the tunnel created at the
navicular bone (arrowheads), where the transferred FHL tendon is attached.
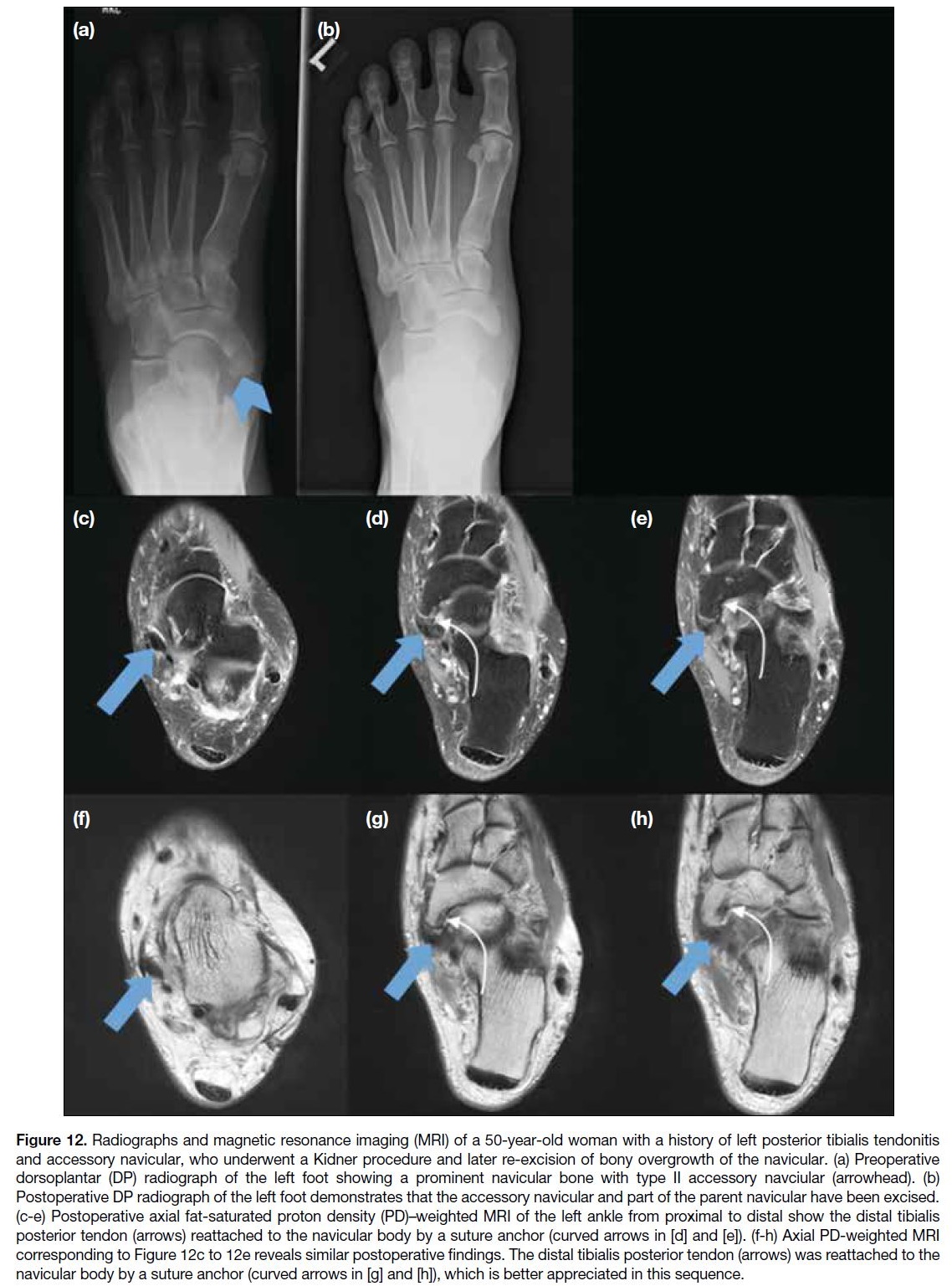
Patients with an accessory navicular bone causing
PTTD can benefit from the Kidner procedure, in which
the accessory navicular bone is excised and the PTT is
rerouted to a more plantar position at the undersurface of
the remaining navicular (Figure 12).[26] Modifications of
the Kidner procedure have been reported, which involve
advancement of the PTT insertion using suture anchors,
biotenodesis screws, or osseous tunnels to reattach the
PTT.[23] The Kidner procedure and its modifications
remain the current standard of care, with reported success
rates of up to 96%.[27] [28]
Figure 12. Radiographs and magnetic resonance imaging (MRI) of a 50-year-old woman with a history of left posterior tibialis tendonitis
and accessory navicular, who underwent a Kidner procedure and later re-excision of bony overgrowth of the navicular. (a) Preoperative
dorsoplantar (DP) radiograph of the left foot showing a prominent navicular bone with type II accessory navicular (arrowhead). (b)
Postoperative DP radiograph of the left foot demonstrates that the accessory navicular and part of the parent navicular have been excised.
(c-e) Postoperative axial fat-saturated proton density (PD)–weighted MRI of the left ankle from proximal to distal show the distal tibialis
posterior tendon (arrows) reattached to the navicular body by a suture anchor (curved arrows in [d] and [e]). (f-h) Axial PD-weighted MRI
corresponding to Figure 12c to 12e reveals similar postoperative findings. The distal tibialis posterior tendon (arrows) was reattached to the
navicular body by a suture anchor (curved arrows in [g] and [h]), which is better appreciated in this sequence.
In the late stages of PTTD (Stages III and IV),
surgical management often involves different forms
of arthrodesis, with or without concomitant soft tissue
reconstruction and osteotomy procedures.[3] [23] [24]
CONCLUSION
Imaging findings of PTTD include a spectrum of
changes ranging from tenosynovitis, tendinosis, and
partial to complete tear. A myriad of related secondary
findings may be seen as PTTD increases strain on other
supporting structures, causing progressive malalignment
and deformity of the foot. Familiarisation with the
imaging features of PTTD allows early diagnosis, guides
appropriate treatment, and aids surgical planning in
advanced cases.
REFERENCES
1. Major NM, Anderson MW. Musculoskeletal MRI E-Book. Elsevier Health Sciences; 2019.
2. Den Hartog BD. Flexor digitorum longus transfer with medial displacement calcaneal osteotomy. Biomechanical rationale. Foot Ankle Clin. 2001;6:67-76. Crossref
3. Flores DV, Mejía Gómez C, Fernández Hernando M, Davis MA,
Pathria MN. Adult acquired flatfoot deformity: anatomy,
biomechanics, staging, and imaging findings. Radiographics.
2019;39:1437-60. Crossref
4. Funk DA, Cass J, Johnson K. Acquired adult flat foot secondary to posterior tibial tendon pathology. J Bone Joint Surg Am. 1986;68:95-102. Crossref
5. Khoury NJ, el-Khoury GY, Saltzman C, Brandser E. MR imaging of posterior tibial tendon dysfunction. AJR Am J Roentgenol. 1996;167:675-82. Crossref
6. Schweitzer ME, Karasick D. MR imaging of disorders of the posterior tibialis tendon. AJR Am J Roentgenol. 2000;175:627-35. Crossref
7. Giza E, Cush G, Schon LC. The flexible flatfoot in the adult. Foot Ankle Clin. 2007;12:251-71. Crossref
8. Jesse MK, Hunt KJ, Strickland C. Postoperative imaging of the ankle. AJR Am J Roentgenol. 2018;211:496-505. Crossref
9. Mann RA, Thompson F. Rupture of the posterior tibial tendon causing flat foot. Surgical treatment. J Bone Joint Surg Am. 1985;67:556-61. Crossref
10. Johnson KA, Strom DE. Tibialis posterior tendon dysfunction. Clin Orthop Relat Res. 1989;(239):196-206. Crossref
11. Chhabra A, Soldatos T, Chalian M, Faridian-Aragh N, Fritz J, Fayad LM, et al. 3-Tesla magnetic resonance imaging evaluation of posterior tibial tendon dysfunction with relevance to clinical staging. J Foot Ankle Surg. 2011;50:320-8. Crossref
12. Mankey MG. A classification of severity with an analysis of causative problems related to the type of treatment. Foot Ankle Clin. 2003;8:461-71. Crossref
13. Petersen B, Fitzgerald J, Schreibman K. Musculotendinous magnetic resonance imaging of the ankle. Semin Roentgenol. 2010;45:250-76. Crossref
14. Premkumar A, Perry MB, Dwyer AJ, Gerber LH, Johnson D, Venzon D, et al. Sonography and MR imaging of posterior tibial tendinopathy. AJR Am J Roentgenol. 2002;178:223-32. Crossref
15. Rosenberg ZS, Cheung Y, Jahss MH, Noto AM, Norman A, Leeds NE. Rupture of posterior tibial tendon: CT and MR imaging with surgical correlation. Radiology. 1988;169:229-35. Crossref
16. Schweitzer ME, Caccese R, Karasick D, Wapner KL, Mitchell DG. Posterior tibial tendon tears: utility of secondary signs for MR imaging diagnosis. Radiology. 1993;188:655-9. Crossref
17. Gazdag AR, Cracchiolo A 3rd. Rupture of the posterior tibial
tendon. Evaluation of injury of the spring ligament and clinical
assessment of tendon transfer and ligament repair. J Bone Joint
Surg Am. 1997;79:675-81. Crossref
18. Mengiardi B, Pinto C, Zanetti M. Spring ligament complex and
posterior tibial tendon: MR anatomy and findings in acquired adult
flatfoot deformity. Semin Musculoskelet Radiol. 2016;20:104-15. Crossref
19. Soliman SB, Spicer PJ, van Holsbeeck MT. Sonographic and
radiographic findings of posterior tibial tendon dysfunction: a
practical step forward. Skeletal Radiol. 2019;48:11-27. Crossref
20. Gentili A, Masih S, Yao L, Seeger LL. Pictorial review: foot axes
and angles. Br J Radiol. 1996;69:968-74. Crossref
21. Meyr AJ, Sansosti LE, Ali S. A pictorial review of reconstructive
foot and ankle surgery: evaluation and intervention of the flatfoot
deformity. J Radiol Case Rep. 2017;11:26-36. Crossref
22. Donovan A, Rosenberg ZS. Extraarticular lateral hindfoot
impingement with posterior tibial tendon tear: MRI correlation.
AJR Am J Roentgenol. 2009;193:672-8. Crossref
23. Dimmick S, Chhabra A, Grujic L, Linklater JM. Acquired flat foot
deformity: postoperative imaging. Semin Musculoskelet Radiol.
2012;16:217-32. Crossref
24. Pomeroy GC, Pike RH, Beals TC, Manoli A 2nd. Acquired flatfoot
in adults due to dysfunction of the posterior tibial tendon. J Bone
Joint Surg Am. 1999;81:1173-82. Crossref
25. Zaw H, Calder JD. Operative management options for symptomatic
flexible adult acquired flatfoot deformity: a review. Knee Surg
Sports Traumatol Arthrosc. 2010;18:135-42. Crossref
26. Kidner FC. The prehallux in relation to flatfoot. JAMA. 1933;101:1539-42. Crossref
27. Kopp FJ, Marcus RE. Clinical outcome of surgical treatment of the symptomatic accessory navicular. Foot Ankle Int. 2004;25:27-30. Crossref
28. Prichasuk S, Sinphurmsukskul O. Kidner procedure for symptomatic accessory navicular and its relation to pes planus.
Foot Ankle Int. 1995;16:500-3. Crossref