Improving Breast Cancer Detection in Screening Mammography with Artificial Intelligence Assistance: A Multi-reader Retrospective Study
ORIGINAL ARTICLE
Hong Kong J Radiol 2026;29:Epub 26 February 2026
Improving Breast Cancer Detection in Screening Mammography with Artificial Intelligence Assistance: A Multi-reader Retrospective Study
PL Lam1, D Fenn1, EH Chan2, EWS Fok3, PH Lee1, KM Kwok2, LKM Wong1, WS Mak1, WP Cheung1, WI Sit1, WK Ng1, GCY Chan1, LW Lo1, EPY Fung1
1 Department of Diagnostic and Interventional Radiology, Kwong Wah Hospital, Hong Kong SAR, China
2 Department of Diagnostic and Interventional Radiology, Princess Margaret Hospital, Hong Kong SAR, China
3 Department of Radiology and Organ Imaging, United Christian Hospital, Hong Kong SAR, China
Correspondence: Dr PL Lam, Department of Diagnostic and Interventional Radiology, Kwong Wah Hospital, Hong Kong SAR, China. Email: lpl404@ha.org.hk
Submitted: 29 August 2024; Accepted: 9 December 2024. This version may differ from the final version when published in an issue.
Contributors: DF, EWSF and EPYF designed the study. DF, EWSF, PHL, KMK, LKMW, WSM, WPC, WIS, WKN, GCYC, LWL and EPYF
acquired the data. PLL, DF, EHC, EWSF and EPYF analysed the data. PLL drafted the manuscript. All authors critically revised the manuscript
for important intellectual content. All authors had full access to the data, contributed to the study, approved the final version for publication, and
take responsibility for its accuracy and integrity.
Conflicts of Interest: All authors have disclosed no conflicts of interest.
Funding/Support: This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Data Availability: All data generated or analysed during the present study are available from the corresponding author on reasonable request.
Ethics Approval: This research was approved by the Central Institutional Review Board of Hospital Authority, Hong Kong (Ref No.: CIRB-2024-074-5). The requirement for informed consent from patients was waived by the Board due to the retrospective nature of the research.
Acknowledgement: The authors thank the Well Women Clinic of Tung Wah Group of Hospitals and radiologists from the Department of
Diagnostic and Interventional Radiology of Kwong Wah Hospital for their support of this study.
Supplementary Material: The supplementary material was provided by the authors and some information may not have been peer reviewed. Any
opinions or recommendations discussed are solely those of the author(s) and are not endorsed by the Hong Kong College of Radiologists. The
Hong Kong College of Radiologists disclaims all liability and responsibility arising from any reliance placed on the content.
Abstract
Introduction
This study aimed to compare the performance of radiologists in screening mammography for breast
cancer detection, with and without artificial intelligence (AI) assistance, including subgroup comparison between
breast radiologists and general radiologists in Hong Kong.
Methods
This was a single-centre multi-reader retrospective study. A screening mammography test set was used
(the Hong Kong Personal Performance in Mammographic Screening Scheme), comprising 80 mammograms with
negative or benign findings and 36 mammograms with pathologically proven breast cancer acquired from December
2009 to December 2023. Radiologists’ performance with and without AI assistance from a commercially available
tool (Lunit INSIGHT MMG) was evaluated from December 2023 to April 2024. The two reading sessions were
separated by a 4-week washout period. Study endpoints included sensitivity and specificity in the mammographic
detection of breast cancer. The Obuchowski–Rockette model was used to estimate and compare diagnostic accuracy.
Results
A total of 16 radiologists completed the test set, including nine (56.3%) breast radiologists and seven
(43.8%) general radiologists. Without AI assistance, the overall sensitivity and specificity in breast cancer detection
were 73.3% and 89.9%, respectively. With AI assistance, both metrics improved significantly to 80.7% (p = 0.007)
and 94.3% (p < 0.001), respectively. Subgroup analysis showed that breast radiologists demonstrated improved
specificity from 87.6% to 92.6% (p < 0.001), while general radiologists acquired more sensitivity from 54.0% to
66.7% (p < 0.001) with the use of AI.
Conclusion
AI assistance significantly improved the diagnostic accuracy of breast radiologists and general
radiologists in screening mammography for breast cancer detection.
Key Words: Artificial intelligence; Breast neoplasms; Mammography; Mass screening
中文摘要
利用人工智能輔助乳房X光檢查提高乳癌篩檢檢出率:一項多位閱片者回顧性研究
林栢麟、范德信、陳恩灝、霍泳珊、李璧希、郭勁明、黃嘉敏、麥詠詩、張偉彬、薛詠妍、吳詠淇、陳頌恩、羅麗雲、馮寶恩
引言
本研究旨在比較香港放射科醫生在乳房X光檢查篩檢乳癌時應用和不應用人工智能輔助兩種情況下的表現,並對乳腺放射科醫生和一般放射科醫生進行亞組比較。
方法
本研究為單中心多位閱片者回顧性研究。研究採用篩檢乳房X 光攝影測試集(HKPERFORMS),此測試集包含於2009年12月至2023年12月期間採集的80例陰性或良性乳腺X光攝影影像及36例經病理證實為乳癌的乳腺X光攝影影像。研究於2023年12月至2024年4月期間評估了放射科醫生在應用和不應用商用人工智能輔助工具(Lunit INSIGHT MMG)兩種情況下的表現。兩次閱片之間相隔4週洗脫期。研究終點包括乳腺X光攝影檢測乳癌的敏感性和特異性。我們採用Obuchowski-Rockette模型評估及比較診斷準確性。
結果
共有16位放射科醫生完成了測試集,其中9名(56.3%)為乳腺放射科醫生,7名(43.8%)為一般放射科醫生。在未使用人工智能輔助的情況下,乳癌檢測的整體敏感性和特異性分別為73.3%和89.9%。使用人工智能輔助後,這兩項指標均顯著提高,分別達到80.7%(p = 0.007)和94.3%(p < 0.001)。亞組分析顯示,使用人工智能後,乳腺放射科醫生的特異性從87.6%提高到92.6%(p < 0.001),而一般放射科醫生的敏感性則從54.0%提高到66.7%(p < 0.001)。
結論
人工能輔助顯著提高了乳腺放射科醫生和一般放射科醫師在乳癌篩檢中應用乳房X光攝影的診斷準確率。
INTRODUCTION
In Hong Kong, breast cancer has been the most common
malignancy among the female population since the early
1990s, with increasing incidence every year. It accounted
for over a quarter (28.9%) of new cancer cases in 2023.[1]
It was also the third leading cause of cancer deaths in
women.[1] Fortunately, breast cancer can be curable in its
early stages, with over 95% 5-year survival for patients
with stage I disease.[2] Previous randomised controlled
trials and meta-analyses have demonstrated the efficacy
of screening mammography in detecting early-stage
tumours and reducing breast cancer–related deaths.[3] [4] [5] [6]
Breast screening programmes have been established in
multiple developed economies worldwide. In Western
countries, the American Cancer Society recommends
that women consider annual mammography screening
starting at the age of 40 years,[7] whereas in the United
Kingdom, the National Health Service offers breast
screening every 3 years for women aged between 50
and 71 years.[8] In Asian countries, such as Japan,[9] South Korea[10] and Singapore,[11] breast screening programmes
have been in place for over a decade. In Hong Kong, the
Centre for Health Protection recommends that women in
the general population aged 44 to 69 years with an average
risk of breast cancer consider mammography screening
every 2 years.[12] Together with increased advocacy from
non-profit organisations, which have heightened disease
awareness among the public, screening mammography
has become more popular.[13]
Like most tests, the diagnostic accuracy of screening
mammography is not absolute. Sensitivity and specificity
in breast cancer detection range between approximately
50% to 80% and about 80% to 90%, respectively, in the
literature.[14] [15] [16] [17] False-positive results lead to additional
workup and the associated anxiety in patients, while
false-negative results can delay treatment and worsen
prognosis.[14]
Recent advancements in machine learning have led to
the increased use of artificial intelligence (AI) in clinical radiology. Some studies, mainly conducted in Western
countries, have shown promising results in employing
AI-based tools to improve the diagnostic accuracy of
screening mammography.[18] [19] [20] [21]
AI-supported software has become more accessible
and commercially available. To the best of our
knowledge, there are no published studies evaluating
the diagnostic performance of screening mammography
with AI assistance in Hong Kong. The lack of
established evidence in our local population could
be a hurdle for radiologists to consider AI-assisted
screening mammography. The external validity of
previous research poses a major concern. Screening
mammography tests employed in studies performed
in Western countries were mainly selected from
Caucasian patients.[22] Asian women, on the other hand,
generally have different breast composition, with a
higher prevalence of dense breasts. This can obscure
abnormalities on mammograms, limiting the detection
of breast cancer and reducing diagnostic accuracy.[23] [24] [25]
Investigations on how AI-based tools could facilitate
screening mammography using test sets derived from a
local Asian population could bridge this data gap.
This study aimed to compare the performance of
radiologists in screening mammography to detect
breast cancer with and without AI assistance in the
local population. Subgroup comparisons between
breast radiologists and general radiologists were also
performed.
METHODS
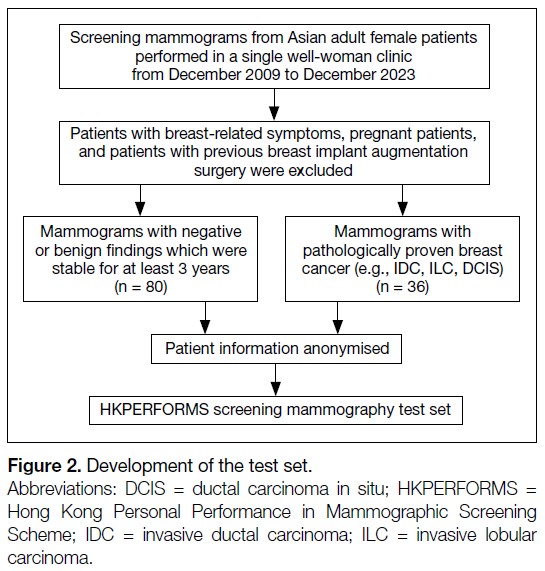
We developed a test set, the Hong Kong Personal
Performance in Mammographic Screening Scheme
(HKPERFORMS), to evaluate the diagnostic accuracy of
radiologists in detecting breast cancer in the local Asian
population with and without AI assistance. The test set
comprised mammograms retrospectively selected from
Asian adult female patients aged 40 years or above who
underwent breast screening in a single well-woman clinic
from December 2009 to December 2023. Exclusion
criteria included symptomatic patients (e.g., those with a
palpable breast mass), pregnant patients, and those with
a history of breast implant augmentation surgery.
All studies in HKPERFORMS were two-dimensional
(2D) screening full-field digital mammograms with
standard craniocaudal and mediolateral oblique views.
There were 80 mammograms showing negative or
benign findings, confirmed as stable on subsequent mammographic follow-up for at least 3 years as
assessed by breast radiologists recognised by the Hong
Kong College of Radiologists (HKCR). There were
36 mammograms with pathologically proven breast
cancer, including invasive ductal carcinoma, invasive
lobular carcinoma, and ductal carcinoma in situ. Their
mammographic appearances included mass (n = 21,
58.3%), calcifications (n = 6, 16.7%), architectural
distortion (n = 5, 13.9%), and asymmetry (n = 4, 11.1%).
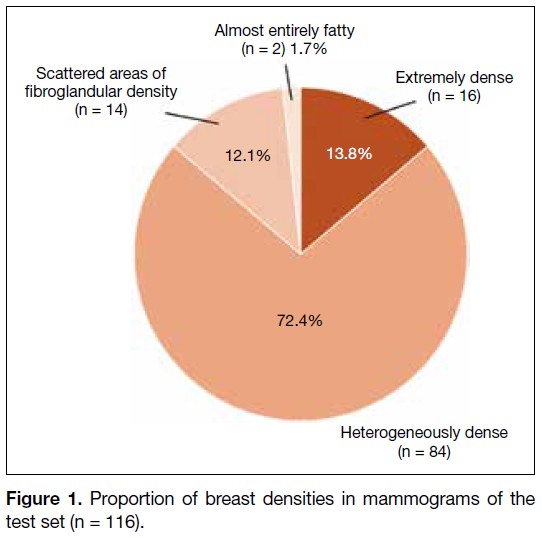
The mammograms in the test set (n = 116) included
breasts of varying densities: extremely dense (13.8%),
heterogeneously dense (72.4%), scattered areas of
fibroglandular density (12.1%), and almost entirely fatty
(1.7%) [Figure 1]. Patient information and identifiers,
such as name and age, were anonymised before compiled
into the HKPERFORMS test set (Figure 2).
Figure 1. Proportion of breast densities in mammograms of the test set (n = 116).
Figure 2. Development of the test set.
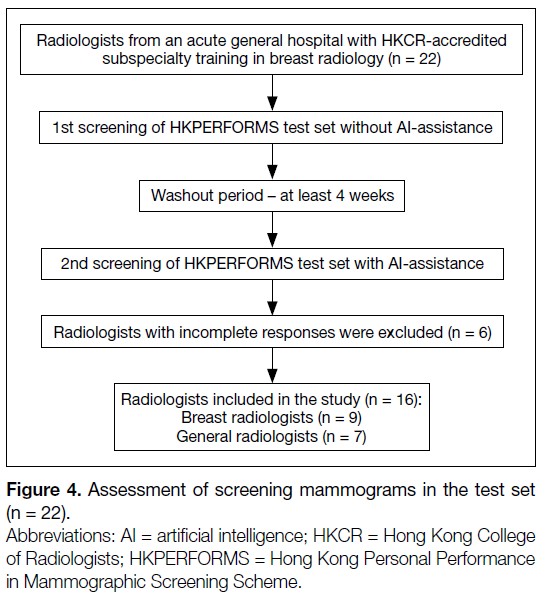
Reader Assessment
This was a single-centre study. Radiologists were
recruited from an acute general hospital with
subspecialty training in breast radiology accredited
by the HKCR. They included breast radiologists and
general radiologists. Breast radiologists were defined
as radiologists with at least 3 months of subspecialty
training recognised by the HKCR, or post-fellowship
breast radiology training, and at least 500 screening
mammograms read in the past year. General radiologists
were defined as HKCR members or fellows actively
practising in clinical radiology, but without dedicated
subspecialty training in breast radiology.
The recruited radiologists were blinded to all patient
information and identifiers in the HKPERFORMS
screening mammography test set. They assessed the
mammograms under standardised conditions using
dedicated software (Selenia Dimensions version 1.11;
Hologic, Bedford [MA], US) with diagnostic-quality
monitors (Coronis Uniti MDMC 12133; Barco, Kortrijk,
Belgium) in accordance with department standards.
Readers documented their screening results digitally
(SurveyMonkey; SurveyMonkey, San Mateo [CA], US).
Data to be entered included breast density, laterality,
quadrant, depth, and presence or absence of architectural
distortion if an abnormality was identified. Respondents
were required to classify each study as benign or
suspicious for malignancy.
All radiologists assessed the HKPERFORMS
test set twice. In the first reading, they read the
screening mammograms without AI assistance. In the
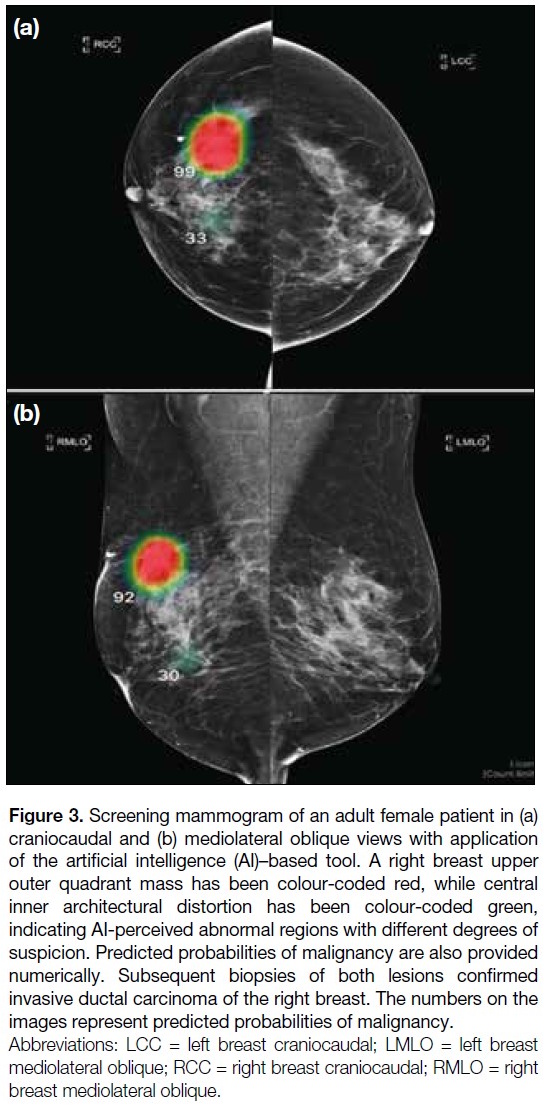
second reading, additional data were provided by a
commercially available AI-based tool (INSIGHT MMG
version 1.1.7.3; Lunit, Seoul, South Korea),[26] which
automatically highlighted regions perceived as abnormal
with a colour-coded heatmap indicating the degree of
suspicion. A predicted probability of malignancy was
also presented numerically (Figure 3). Both pre– and
post–AI-processed mammograms were available during
the second reading. Respondents were instructed to
record their screening results after reviewing all images. They were at liberty to follow or disregard the AI-based
assessment entirely. A washout period of at least 4 weeks
was observed between the two readings. The orders of
the screening mammograms in the test set were different
and randomised across the two sittings. Respondents
who did not complete either reading were excluded from
the study (Figure 4).
Figure 3. Screening mammogram of an adult female patient in (a)
craniocaudal and (b) mediolateral oblique views with application
of the artificial intelligence (AI)–based tool. A right breast upper
outer quadrant mass has been colour-coded red, while central
inner architectural distortion has been colour-coded green,
indicating AI-perceived abnormal regions with different degrees of
suspicion. Predicted probabilities of malignancy are also provided
numerically. Subsequent biopsies of both lesions confirmed
invasive ductal carcinoma of the right breast. The numbers on the
images represent predicted probabilities of malignancy.
Figure 4. Assessment of screening mammograms in the test set (n = 22).
Background information of the recruited radiologists,
including prior subspecialty training in breast radiology
and experience in reporting breast imaging, was
collected. All responses submitted electronically were anonymised and a random computer-generated number
was assigned to each radiologist. Researchers were
blinded to the identity of the respondents.
Statistical Analysis
Statistical analysis was performed using R (macOS
version 4.4.1; R Core Team, Vienna, Austria).[27] Study
endpoints of diagnostic accuracy included sensitivity
and specificity in the mammographic detection of breast
cancer. The Obuchowski–Rockette model was used to
estimate and compare diagnostic accuracy.[28] A p value
of < 0.05 was considered statistically significant.
This manuscript was prepared in accordance with the
STROBE (Strengthening the Reporting of Observational
Studies in Epidemiology) guidelines.
RESULTS
Overall Performance
A total of 22 radiologists were invited to participate
in this study; six respondents who did not complete
the HKPERFORMS screening mammography test set
were excluded, resulting in 16 radiologists completing
the test set (Figure 4). Without AI assistance, the
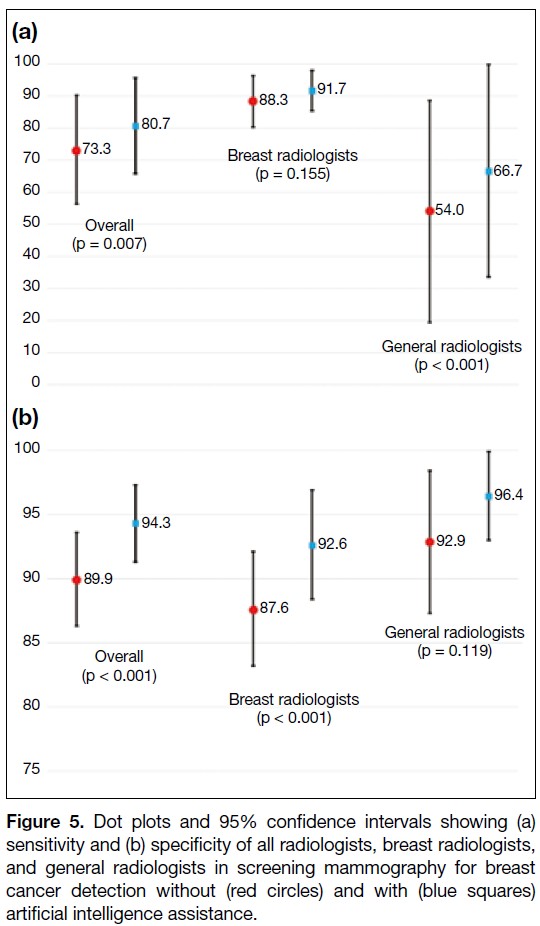
mean sensitivity and specificity for detecting breast
cancer were 73.3% and 89.9%, respectively. With
AI assistance, there was significant improvement
in diagnostic accuracy, with the mean sensitivity
and specificity increasing to 80.7% (p = 0.007) and 94.3% (p < 0.001), respectively (Figure 5 and online supplementary Table).
Figure 5. Dot plots and 95% confidence intervals showing (a)
sensitivity and (b) specificity of all radiologists, breast radiologists,
and general radiologists in screening mammography for breast
cancer detection without (red circles) and with (blue squares)
artificial intelligence assistance.
Subgroup Analysis
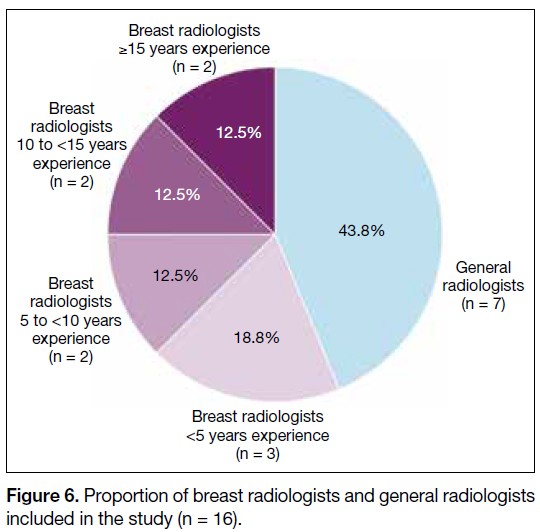
Among the respondents, nine (56.3%) were breast
radiologists and seven (43.8%) were general radiologists.
The experience of the breast radiologists is shown in
Figure 6. Without AI assistance, the mean sensitivity
of the breast radiologists (88.3%) was significantly
higher than that of the general radiologists (54.0%) in
identifying breast cancer (p = 0.017). There was no
significant difference in the mean specificity between
the two groups (breast radiologists: 87.6% vs. general
radiologists: 92.9%; p = 0.051). Using the AI-based tool,
there was significant improvement in the specificity of
the breast radiologists (from 87.6% to 92.6%; p < 0.001)
and the sensitivity of the general radiologists (from
54.0% to 66.7%; p < 0.001). No significant changes in the sensitivity of breast radiologists and the specificity
of general radiologists were observed after using the AI-based
tool (Figure 5 and online supplementary Table).
Figure 6. Proportion of breast radiologists and general radiologists
included in the study (n = 16).
DISCUSSION
Diagnostic Accuracy Without Artificial
Intelligence Assistance
Without assistance from the AI-based tool, the diagnostic
accuracy of the breast radiologists included in this study
was comparable to figures reported in the literature, with
both sensitivity and specificity exceeding 85%.[15] [16] [17] In
contrast, general radiologists were less likely to detect
breast malignancy, with a sensitivity of about 54%.
Screening tests with low sensitivity lead to a higher
proportion of false-negative results, potentially leading
to false reassurance and missed opportunities for early
diagnosis and treatment.[14] These findings highlight the
importance of dedicated training in breast radiology.[29] [30]
The HKCR Mammography Statement outlines the
standards for radiologists involved in screening. These
include a minimum of 3 months of subspecialty training
in breast radiology, interpretation of at least 500 screening
mammograms annually, and ongoing participation in
continuing medical education and multidisciplinary
meetings.[31]
Improved Performance with Artificial
Intelligence Assistance
There were significant improvements in overall
sensitivity and specificity in breast cancer detection
when radiologists in this study performed AI-assisted screening mammography. This echoed previous studies
which demonstrated improved diagnostic accuracy in AI-assisted
mammography readings.[18] [19] [20] [21] Subgroup analysis
further showed that the benefits of AI assistance differed
between general radiologists and breast radiologists.
For general radiologists, there was significant
improvement in sensitivity, from approximately 50%
when screening unaided to over 65% with the use of AI-based
tool. A previous study also demonstrated reduced
variability in screening results and increased inter-reader
reliability with AI assistance.[32] This indicates
that utilising AI could yield more expertise-independent
results. AI could act as an extra pair of eyes. Radiologists
could refer to colour-coded heatmaps generated by AI-based
software after initial mammography assessment to
reduce the probability of missing breast cancer.[26]
Among the breast radiologists, there was improvement
in specificity, while sensitivity in detecting breast cancer
remained similar with and without AI assistance. The
crux of screening lies in striking a balance between
sensitivity and specificity. Tests with high sensitivity but
low specificity may lead to over-investigation, resulting
in unnecessary stress and interventions for patients.[14]
While the specificity of the breast radiologists in breast
cancer detection was satisfactory without AI assistance,
it improved from over 85% to over 90% with the use
of the AI-based tool without compromising sensitivity.
Increased specificity in screening mammography would
reduce call-back rates, avoid unwarranted workups for
patients, and decrease the workload for radiologists.[20] [33]
A study by Raya-Povedano et al[34] revealed a reduction
of over 70% in radiologists’ workload following the
implementation of AI-based strategies. Additionally,
AI tools could be helpful to prioritise screening
mammograms with suspected malignancy. Such
abnormal studies could be flagged for earlier reporting
by radiologists, expediting subsequent workup and
treatment. Furthermore, placing flagged studies at the
beginning of a screening session could minimise the
risk of missed breast cancers due to reader fatigue. With
the burgeoning demand for screening mammography in
Hong Kong, AI-based tools could potentially alleviate
the stress faced by radiologists.
Limitations
The HKPERFORMS test set was enriched with
abnormal mammograms, and the proportion of cases
with biopsy-proven breast cancer was not representative
of routine screening practice or the general population.[1] [2] Although respondents were instructed to interpret each
individual mammogram as an independent screening
case, their diagnostic accuracy might have been
negatively influenced by the study design. Second,
test sets used in the sittings with and without AI
assistance were identical. Despite a washout period
of at least 4 weeks with randomisation of the image
order, radiologists might have recalled the proportion
of normal to abnormal cases, potentially introducing
bias in the second sitting. Third, all mammograms in
the test set were 2D full-field digital mammograms. In
recent years, three-dimensional mammography or digital
breast tomosynthesis (DBT) has become more popular,
with evidence showing improved diagnostic accuracy
compared with traditional 2D mammography. Studies on
AI-assisted DBT have shown non-inferior or improved
sensitivity and specificity in detecting breast cancer.[35] [36]
Our study did not investigate DBT performance, which
remains a potential direction for further research. Finally,
this was a single-centre study with limited sample size.
The performance and influence of AI may vary among
radiologists with differing levels of experience across
diverse clinical settings. Further large-scale multi-centre
investigations would provide a more comprehensive
assessment.
CONCLUSION
This multi-reader study evaluated the potential of AI to
aid breast cancer detection using HKPERFORMS, an
original screening mammography test set developed from
a local Asian female population with a high incidence of
dense breasts. The results demonstrated that diagnostic
accuracy in screening mammography was improved
across radiologists with varying levels of experience in
breast radiology when supported by AI-based tools.
REFERENCES
1. Centre for Health Protection, Department of Health, Hong Kong SAR Government. Breast Cancer. 23 Jan 2026. Available from: https://www.chp.gov.hk/en/healthtopics/content/25/53.html. Accessed 2 Feb 2026.
2. Kwong A, Mang OW, Wong CH, Chau WW, Law SC; Hong Kong Breast Cancer Research Group. Breast cancer in Hong Kong, Southern China: the first population-based analysis of epidemiological characteristics, stage-specific, cancer-specific, and disease-free survival in breast cancer patients: 1997–2001. Ann Surg Oncol. 2011;18:3072–8.
Crossref
3. Moss SM, Cuckle H, Evans A, Johns L, Waller M, Bobrow L, et al. Effect of mammographic screening from age 40 years on breast cancer mortality at 10 years’ follow-up: a randomised controlled trial. Lancet. 2006;368:2053–60.
Crossref
4. Duffy SW, Tabár L, Chen HH, Holmqvist M, Yen MF, Abdsalah S, et al. The impact of organized mammography service screening on breast carcinoma mortality in seven Swedish counties. Cancer. 2002;95:458–69.
Crossref
5. Tabár L, Vitak B, Chen HH, Yen MF, Duffy SW, Smith RA. Beyond randomized controlled trials: organized mammographic screening substantially reduces breast carcinoma mortality. Cancer. 2001;91:1724–31.
Crossref
6. Kerlikowske K, Grady D, Rubin SM, Sandrock C, Ernster VL. Efficacy of screening mammography. A meta-analysis. JAMA. 1995;273:149–54.
Crossref
7. American Cancer Society. American Cancer Society recommendations for the early detection of breast cancer. Available from: https://www.cancer.org/cancer/types/breast-cancer/screening-tests-and-early-detection/american-cancer-society-recommendations-for-the-early-detection-of-breast-cancer.html. Accessed 20 Aug 2024.
8. National Health Service, Department of Health and Social Care, United Kingdom Government. Breast screening (mammogram). Available from: https://www.nhs.uk/conditions/breast-screening-mammogram/. Accessed 20 Aug 2024.
9. Hamashima CC, Hattori M, Honjo S, Kasahara Y, Katayama T, Nakai M, et al. The Japanese guidelines for breast cancer screening. Jpn J Clin Oncol. 2016;46:482–92.
Crossref
10. Shin DW, Yu J, Cho J, Lee SK, Jung JH, Han K, et al. Breast cancer screening disparities between women with and without disabilities: a national database study in South Korea. Cancer. 2020;126:1522–9.
Crossref
11. Loy EY, Molinar D, Chow KY, Fock C. National Breast Cancer Screening Programme, Singapore: evaluation of participation and performance indicators. J Med Screen. 2015;22:194–200.
Crossref
12. Cancer Expert Working Group on Cancer Prevention and Screening, Centre for Health Protection, Department of Health, Hong Kong SAR Government. Recommendations on Prevention and Screening for Breast Cancer for Health Professionals. June 2020. Available from: https://www.chp.gov.hk/files/pdf/breast_cancer_professional_hp.pdf. Accessed 20 Aug 2024.
13. Hong Kong Breast Cancer Foundation. What is breast cancer. Available from: https://www.hkbcf.org/en/breast_cancer/main/422/. Accessed 20 Aug 2024.
Crossref
14. Marmot MG, Altman DG, Cameron DA, Dewar JA, Thompson SG, Wilcox M. The benefits and harms of breast cancer screening: an independent review. Br J Cancer. 2013;108:2205–40.
Crossref
15. Hollingsworth AB. Redefining the sensitivity of screening mammography: a review. Am J Surg. 2019;218:411–8.
Crossref
16. Kerlikowske K, Grady D, Barclay J, Sickles EA, Ernster V. Likelihood ratios for modern screening mammography. Risk of breast cancer based on age and mammographic interpretation. JAMA. 1996;276:39–43.
Crossref
17. Lehman CD, Wellman RD, Buist DS, Kerlikowske K, Tosteson AN, Miglioretti DL, et al. Diagnostic accuracy of digital screening mammography with and without computer-aided detection. JAMA Intern Med. 2015;175:1828–37.
Crossref
18. Dembrower K, Crippa A, Colón E, Eklund M, Strand F; ScreenTrustCAD Trial Consortium. Artificial intelligence for breast cancer detection in screening mammography in Sweden: a prospective, population-based, paired-reader, non-inferiority study. Lancet Digit Health. 2023;5:e703–11.
Crossref
19. Lång K, Josefsson V, Larsson AM, Larsson S, Högberg C, Sartor H,
et al. Artificial intelligence–supported screen reading versus
standard double reading in the Mammography Screening with
Artificial Intelligence trial (MASAI): a clinical safety analysis of
a randomised, controlled, non-inferiority, single-blinded, screening
accuracy study. Lancet Oncol. 2023;24:936-44. Crossref
20. Lauritzen AD, Lillholm M, Lynge E, Nielsen M, Karssemeijer N,
Vejborg I. Early indicators of the impact of using AI in mammography
screening for breast cancer. Radiology. 2024;311:e232479.
Crossref
21. Ng AY, Oberije CJ, Ambrózay E, Szabó E, Serfozó O, Karpati E, et al. Prospective implementation of AI-assisted screen reading to improve early detection of breast cancer. Nat Med. 2023;29:3044–9.
Crossref
22. Chen Y, Gale A. Performance assessment using standardized
data sets: the PERFORMS scheme in breast screening and other
domains. In: Samei E, Krupinski EA, editors. The Handbook of
Medical Image Perception and Techniques. 2nd ed. Cambridge,
England: Cambridge University Press; 2018: 328-42. Crossref
23. Bao C, Shen J, Zhang Y, Zhang Y, Wei W, Wang Z, et al.
Evaluation of an artificial intelligence support system for breast
cancer screening in Chinese people based on mammogram. Cancer
Med. 2023;12:3718-26.
Crossref
24. Yan H, Ren W, Jia M, Xue P, Li Z, Zhang S, et al. Breast cancer
risk factors and mammographic density among 12518 average-risk
women in rural China. BMC Cancer. 2023;23:952.
Crossref
25. Jackson VP, Hendrick RE, Feig SA, Kopans DB. Imaging of the
radiographically dense breast. Radiology. 1993;188:297-301.
Crossref
26. Kim HE, Kim HH, Han BK, Kim KH, Han K, Nam H, et al. Changes
in cancer detection and false-positive recall in mammography using
artificial intelligence: a retrospective, multireader study. Lancet
Digit Health. 2020;2:e138-48. Crossref
27. R Core Team. R: a language and environment for statistical
computing. Vienna: R Foundation for Statistical Computing; 2020.
28. Hillis SL, Obuchowski NA, Berbaum KS. Power estimation for
multireader ROC methods: an updated and unified approach. Acad
Radiol. 2011;18:129-42. Crossref
29. Trieu PD, Lewis SJ, Li T, Ho K, Wong DJ, Tran OT, et al.
Improving radiologist’s ability in identifying particular abnormal
lesions on mammograms through training test set with immediate feedback. Sci Rep. 2021;11:9899. Crossref
30. Miglioretti DL, Gard CC, Carney PA, Onega TL, Buist DS,
Sickles EA, et al. When radiologists perform best: the learning
curve in screening mammogram interpretation. Radiology.
2009;253:632-40. Crossref
Hong Kong College of Radiologists. Hong Kong College of
Radiologists Mammography Statement. Revised 25 August 2015.
Available from: https://www.hkcr.org/templates/OS03C00336/case/lop/HKCR%20Mammography%20Statement_rev20150825.
pdf. Accessed 20 Aug 2024.
32. Pacilè S, Lopez J, Chone P, Bertinotti T, Grouin JM, Fillard P.
Improving breast cancer detection accuracy of mammography with
the concurrent use of an artificial intelligence tool. Radiol Artif
Intell. 2020;2:e190208. Crossref
33. Kim YS, Jang MJ, Lee SH, Kim SY, Ha SM, Kwon BR, et al.
Use of artificial intelligence for reducing unnecessary recalls at
screening mammography: a simulation study. Korean J Radiol.
2022;23:1241-50. Crossref
34. Raya-Povedano JL, Romero-Martín S, Elías-Cabot E, Gubern-
Mérida A, Rodríguez-Ruiz A, Álvarez-Benito M. AI-based
strategies to reduce workload in breast cancer screening with
mammography and tomosynthesis: a retrospective evaluation.
Radiology. 2021;300:57-65. Crossref
35. Goldberg JE, Reig B, Lewin AA, Gao Y, Heacock L, Heller SL,
et al. New horizons: artificial intelligence for digital breast
tomosynthesis. Radiographics. 2022;43:e220060. Crossref
36. Park EK, Kwak S, Lee W, Choi JS, Kooi T, Kim EK. Impact of
AI for digital breast tomosynthesis on breast cancer detection and
interpretation time. Radiol Artif Intell. 2024;6:e230318.
Crossref