Urachal Adenocarcinoma in a Young Adult: A Rare Case Report
CASE REPORT
Hong Kong J Radiol 2026;29:Epub 26 February 2026
Urachal Adenocarcinoma in a Young Adult: A Rare Case Report
LLA Chan, IS Bandong
Institute of Radiology, St. Luke’s Medical Center–Quezon City, Quezon City, The Philippines
Correspondence: Dr LLA Chan, Institute of Radiology, St Luke’s Medical Center–Quezon City, Quezon City, The Philippines. Email: llachan@stlukes.com.ph
Submitted: 12 February 2025; Accepted: 28 April 2025. This version may differ from the final version when published in an issue.
Contributors: LLAC designed the study, acquired and analysed the data, and drafted the manuscript. ISB critically revised the manuscript for
important intellectual content. Both authors had full access to the data, contributed to the study, approved the final version for publication, and
take responsibility for its accuracy and integrity.
Conflicts of Interest: Both authors have disclosed no conflicts of interest.
Funding/Support: This study received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Data Availability: All data generated or analysed during the present study are available from the corresponding author on reasonable request.
Ethics Approval: This study was approved by the Institutional Ethics Review Committee of St Luke’s Medical Center–Quezon City, The
Philippines (Ref No.: SL-21346). The patient was treated in accordance with the Declaration of Helsinki. Informed consent for publication of
this case report and the accompanying images was obtained from the patient’s mother, as the patient is deceased.
CASE PRESENTATION
A 19-year-old female presented to our institution in
February 2023 with intermittent gross haematuria
and dysuria for 2 months without seeking medical
consultation. She then experienced a syncopal attack,
prompting consultation and eventual admission. Her
medical history included recurrent untreated urinary
tract infections since childhood. No family history of
malignancy or prior abdominal surgery was noted.
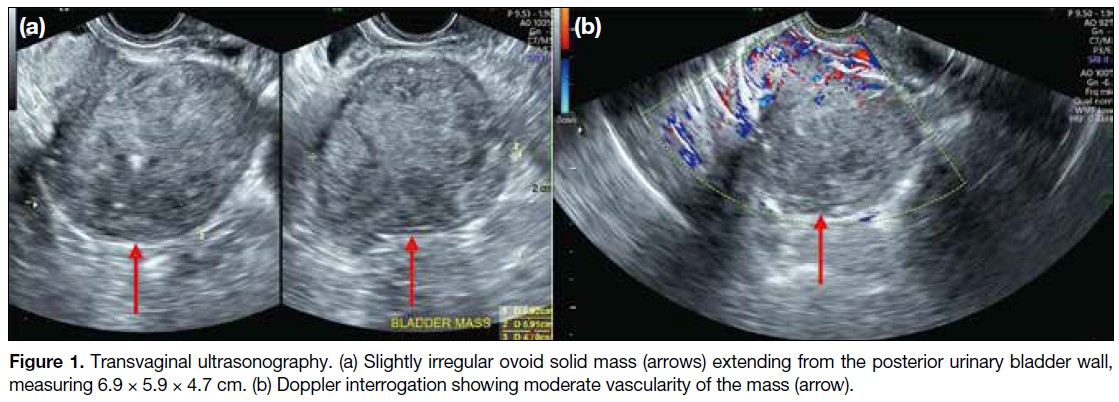
Initial transvaginal ultrasound revealed a solid, slightly
irregular ovoid mass measuring 6.9 × 5.9 × 4.7 cm,
located in the posterior bladder wall (Figure 1a). The
mass exhibited heterogeneous echogenicity with
punctate calcifications. Doppler ultrasound revealed
moderate vascularity (Figure 1b). The ovaries, adnexa,
and uterus appeared unremarkable.
Figure 1. Transvaginal ultrasonography. (a) Slightly irregular ovoid solid mass (arrows) extending from the posterior urinary bladder wall,
measuring 6.9 × 5.9 × 4.7 cm. (b) Doppler interrogation showing moderate vascularity of the mass (arrow).
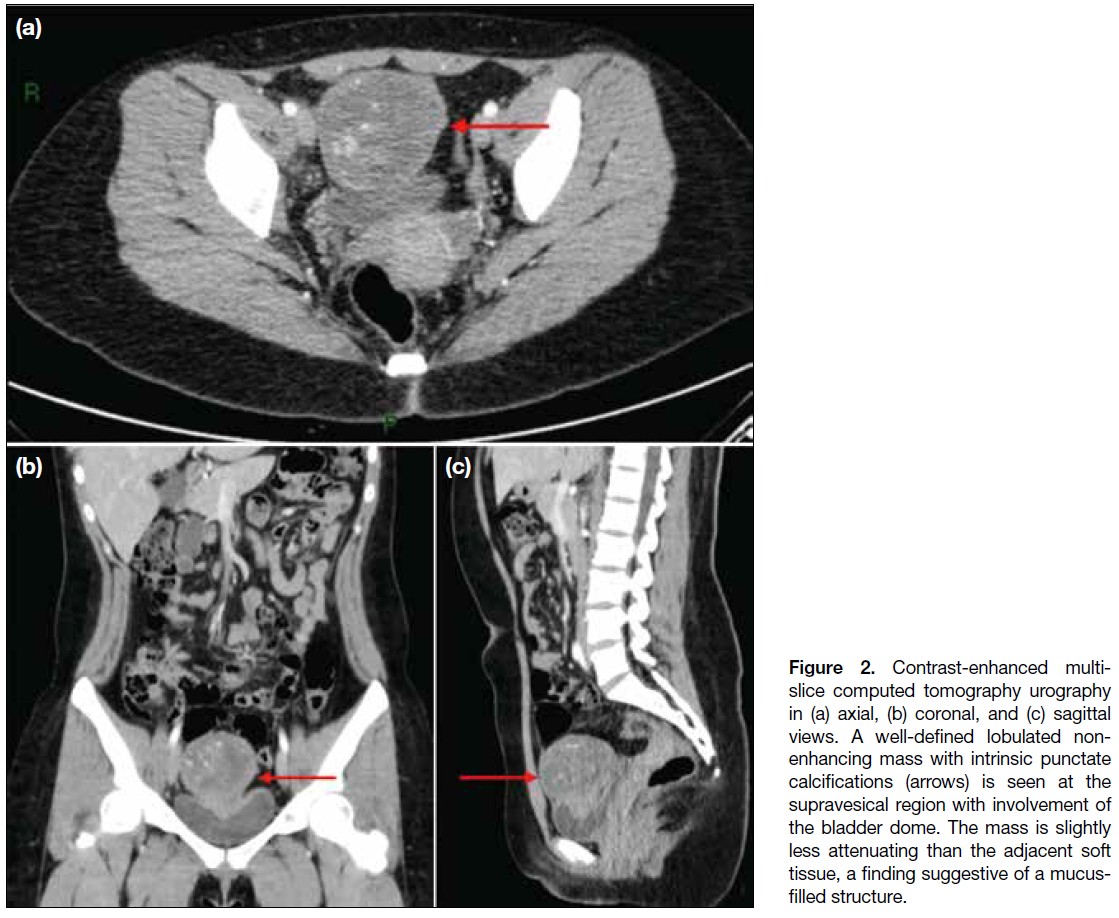
A subsequent computed tomography (CT) urography
(Figure 2) revealed a lobulated, heterogeneously
enhancing mass in the supravesical region with associated
calcifications. The mass abutted the bladder dome with
obliteration of the fat plane, suggesting infiltration. A
1.8-cm enlarged lymph node was also noted in the right
paravesical region. A urachal neoplasm was considered.
Figure 2. Contrast-enhanced multi-slice
computed tomography urography
in (a) axial, (b) coronal, and (c) sagittal
views. A well-defined lobulated non-enhancing
mass with intrinsic punctate
calcifications (arrows) is seen at the
supravesical region with involvement of
the bladder dome. The mass is slightly
less attenuating than the adjacent soft
tissue, a finding suggestive of a mucus-filled
structure.
The patient underwent radical cystectomy and total
abdominal hysterectomy with bilateral salpingectomy,
all of which were well tolerated without complications.
Histopathological examination of the excised mass
revealed a moderately differentiated mucinous
adenocarcinoma, consistent with urachal carcinoma.
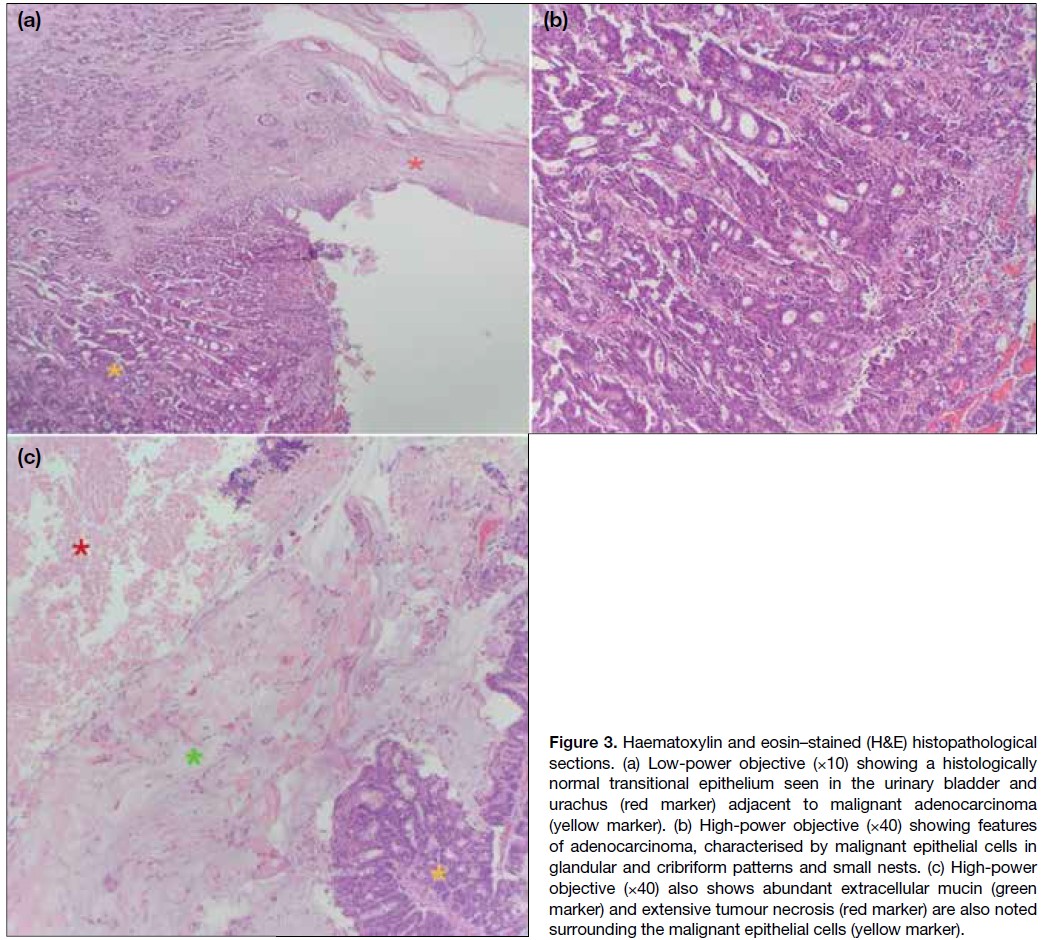
Histopathological Findings
The mass was located approximately 6 cm from the
umbilicus, with smooth external surfaces and yellow-tan
friable content. Histological analysis showed malignant
epithelial cells arranged in glandular and cribriform
patterns, with extensive extracellular mucin and areas
of tumour necrosis (Figure 3). The tumour infiltrated
the bladder’s lamina propria, muscularis propria, and
perivesical fat. These findings were consistent with
mucinous adenocarcinoma, a type of urachal carcinoma.
Figure 3. Haematoxylin and eosin–stained (H&E) histopathological
sections. (a) Low-power objective (×10) showing a histologically
normal transitional epithelium seen in the urinary bladder and
urachus (red marker) adjacent to malignant adenocarcinoma
(yellow marker). (b) High-power objective (×40) showing features
of adenocarcinoma, characterised by malignant epithelial cells in
glandular and cribriform patterns and small nests. (c) High-power
objective (×40) also shows abundant extracellular mucin (green
marker) and extensive tumour necrosis (red marker) are also noted
surrounding the malignant epithelial cells (yellow marker).
Postoperative Course and Outcome
Following surgery, the patient’s recovery was uneventful.
She was eventually discharged and underwent
three cycles of chemotherapy comprising FOLFOX
(leucovorin, 5-fluorouracil, and oxaliplatin). Eighteen
months after surgery, she was frequently admitted with
recurrent urinary tract infections that were found to be caused by a newly discovered metastatic growth on the
anterior pelvic wall, compressing the urinary collecting
system. The patient underwent palliative care and
eventually deceased within a year.
DISCUSSION
Urachal adenocarcinoma is a very rare primary bladder
neoplasm, accounting for only 0.35% to 0.7% of all
primary bladder cancers.[1] This malignancy tends to have a male predilection and typically occurs in adults
between 40 and 70 years old. The most common clinical
feature is haematuria, as seen in the index patient. Other
signs and symptoms include dysuria, abdominal pain, a
suprapubic mass, and discharge of blood, pus, or mucus
from the umbilicus.[2] Only six adult cases of urachal
adenocarcinoma diagnosed before the age of 30 years
have been reported in the English literature, with the
youngest diagnosed at age 26 years.[1] [3] [4] [5] [6]
Ultrasonography is often performed as the initial
imaging modality and can provide a general impression
of the lesion, including its location and characteristics.[7]
Sonographic imaging features of urachal adenocarcinoma include: (1) a solid mass extending between the dome
of the bladder and the abdominal wall, with an irregular
shape and bladder wall invasion; (2) a hypoechoic,
heterogeneous echo pattern with a small amount of
calcification; and (3) patchy, short-line blood flow signals
within the mass.[8] These characteristic features were
analogous to those seen on the initial ultrasonography
performed in our patient.
CT imaging can be used to confirm the ultrasonographic
findings or serve as the first-line imaging to evaluate
local disease, tumour extension, and the presence of
pelvic lymph node involvement or distant metastases.[7]
A key diagnostic feature of urachal adenocarcinoma on CT is its supravesical midline location. The mass
often demonstrates predominantly low attenuation,
attributable to its mucinous content found on pathological
examination. Calcifications are also commonly seen in
mucinous tumours.[9] These findings closely correspond
to the appearance and location of the tumour in the index
patient’s CT urography.
Although urachal remnants are lined by urothelial
epithelium, 80% of urachal cancers are adenocarcinomas,
including mucin-producing (69%) and mucin-negative
(15%) subtypes.7 The reason why adenocarcinoma is the
predominant malignant epithelial type in urachal cancers
remains unclear, but it has been hypothesised that
chronic irritation may induce malignant transformation
of transitional epithelium into columnar epithelium.[7]
Another theory proposes that intestinal metaplasia
in the urinary bladder is associated with cytogenetic
abnormalities and significant telomere shortening
relative to telomere length in adjacent normal urothelial
cells.[10] These theories may help explain how urachal
adenocarcinoma can, albeit rarely, present in a younger
demographic, such as in the case of the index patient
who experienced recurrent urinary tract infections and
was therefore subject to d from childhood.
Differential diagnoses for urachal adenocarcinoma
include ovarian malignancies and other types of urinary
bladder cancer. Sonographic and CT findings of these
malignancies may reveal large, complex masses similar to
the radiographic findings of urachal adenocarcinoma.[11] [12] [13]
Nonetheless, the key feature that supports a diagnosis of
urachal adenocarcinoma over other possibilities is the
supravesical midline location of the mass.
Surgery remains the mainstay of treatment for urachal
adenocarcinoma. For muscle-invasive disease, radical
cystectomy with en bloc resection of the urachal
ligament may be the only curative option. Nonetheless,
survival still strongly correlates with the stage and grade
of the disease. A study reported a 5-year survival rate of
50% for stage I to III tumours, while no stage IV patients
survived beyond 2 years.[11] Urachal adenocarcinoma has
also been found to be resistant to chemotherapy and radiotherapy; therefore, early definitive diagnosis and
radical resection are essential for a better outcome.[11]
CONCLUSION
Urachal carcinoma is a rare and aggressive malignancy
that should be considered in the differential diagnosis
of pelvic masses, even in young patients. The rarity of
this condition highlights the importance of radiological
imaging in early detection. Ultrasonography and CT are
essential for identifying the tumour and assessing its
extent. Although surgical resection remains the treatment
of choice, the prognosis is generally poor, underscoring
the need for further research into effective therapies for
this rare and challenging type of cancer.
REFERENCES
1. Gopalan A, Sharp DS, Fine SW, Tickoo SK, Herr HW, Reuter VE, et al. Urachal carcinoma: a clinicopathologic analysis of 24 cases with outcome correlation. Am J Surg Pathol. 2009;33:659–68.
Crossref
2. Chen X, Kang C, Zhang M. Imaging features of urachal cancer: a case report. Front Oncol. 2019;9:1274.
Crossref
3. Henly DR, Farrow GM, Zincke H. Urachal cancer: role of conservative surgery. Urology. 1993;42:635–9.
Crossref
4. Lee SR, Kang H, Kang MH, Yu YD, Choi CI, Choi KH, et al. The youngest Korean case of urachal carcinoma. Case Rep Urol. 2015;2015:707456.
Crossref
5. Pinthus JH, Haddad R, Trachtenberg J, Holowaty E, Bowler J, Herzenberg AM, et al. Population based survival data on urachal tumors. J Urol. 2006;175:2042–7.
Crossref
6. Machida H, Ueno E, Nakazawa H, Fujimura M, Kihara T.
Computed tomographic appearance of urachal carcinoma associated
with urachal diverticulum misdiagnosed by cystoscopy. Abdom
Imaging. 2008;33:363-6.
Crossref
7. Parada Villavicencio C, Adam SZ, Nikolaidis P, Yaghmai V,
Miller FH. Imaging of the urachus: anomalies, complications, and
mimics. Radiographics. 2016;36:2049-63.
Crossref
8. Koster IM, Cleyndert P, Giard RW. Best cases from the AFIP:
urachal carcinoma. Radiographics. 2009;29:939-42.
Crossref
9. Brick SH, Friedman AC, Pollack HM, Fishman EK, Radecki PD,
Siegelbaum MH, et al. Urachal carcinoma: CT findings. Radiology.
1988;169:377-81.
Crossref
10. Lim H, Lusaya D. Urachal mucinous adenocarcinoma of the
bladder. Philipp J Urol. 2020;28:115-7.
11. Marko J, Marko KI, Pachigolla SL, Crothers BA, Mattu R,
Wolfman DJ. Mucinous neoplasms of the ovary: radiologic-pathologic
correlation. Radiographics. 2019;39:982-97.
Crossref
12. Wong-You-Cheong JJ, Woodward PJ, Manning MA, Sesterhenn IA.
From the Archives of the AFIP: neoplasms of the urinary bladder:
radiologic-pathologic correlation. Radiographics. 2006;26:553-80.
Crossref
13. Varma V, Myers DT. Urachal adenocarcinoma. Appl Radiol.
2019;48:44-5.
Crossref