Treatment Outcomes of Nasopharyngeal Carcinoma in Patients Aged 80 Years or Above
ORIGINAL ARTICLE
Hong Kong J Radiol 2026;29:Epub 9 March 2026
Treatment Outcomes of Nasopharyngeal Carcinoma in Patients Aged 80 Years or Above
PW Kwok, I Yeung, WWY Tin, SY Tung
Department of Clinical Oncology, Tuen Mun Hospital, Hong Kong SAR, China
Correspondence: Dr PW Kwok, Department of Clinical Oncology, Tuen Mun Hospital, Hong Kong SAR, China. Email: kwokpw@ha.org.hk
Submitted: 1 July 2025; Accepted: 18 December 2025. This version may differ from the final version when published in an issue.
Contributors: All authors designed the study. PWK and IY acquired and analysed the data. PWK drafted the manuscript. PWK, WWYT and
SYT critically revised the manuscript for important intellectual content. All authors had full access to the data, contributed to the study, approved
the final version for publication, and take responsibility for its accuracy and integrity.
Conflicts of Interest: All authors have disclosed no conflicts of interest.
Funding/Support: This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Data Availability: All data generated or analysed during the present study are available from the corresponding author on reasonable request.
Ethics Approval: This study was approved by the Central Institutional Review Board of Hospital Authority, Hong Kong (Ref No.: CIRB-2024-421-1). The requirement for informed consent was waived by the Board due to the retrospective nature of the study.
Supplementary Material: The supplementary material was provided by the authors and some information may not have been peer reviewed. Any opinions or recommendations discussed are solely those of the author(s) and are not endorsed by the Hong Kong College of Radiologists. The
Hong Kong College of Radiologists disclaims all liability and responsibility arising from any reliance placed on the content.
Abstract
Introduction
Optimal treatment for nasopharyngeal carcinoma (NPC) in patients aged 80 years or above remains
controversial due to concerns about the tolerability of radical radiotherapy (RT). This study evaluated treatment
outcomes and toxicities in octogenarians with NPC in Hong Kong.
Methods
This retrospective analysis included patients aged 80 years or above with NPC treated at a single institution
in Hong Kong between January 2009 and December 2023. Patients with distant metastases at diagnosis were
excluded. Patient characteristics, treatment outcomes, and toxicities were analysed.
Results
A total of 42 patients (median age, 83 years; range, 80-94) were included. The median follow-up duration
was 20.3 months. In the entire cohort, the median overall survival (OS) was 22.8 months (95% confidence interval
[95% CI] = 14.6-30.9) and the 5-year OS rate was 23.8%. Twenty-seven patients (64.3%) received radical RT
using intensity-modulated radiotherapy (IMRT); none received chemotherapy. Among these patients (Cohort A),
the median OS was 41.3 months (95% CI = 27.7-55.0), while the 5-year OS and cancer-specific survival rates were
38.1% and 74.2%, respectively. Grade ≥3 acute toxicities occurred in 22.2% of patients; one patient (3.7%) died
due to treatment-related toxicity. Treatment failure occurred in five patients (18.5%), all due to distant metastases.
Among patients who received non-radical RT (Cohort B), the median OS was 12.8 months (95% CI = 10.9-14.7),
and none survived beyond 5 years. Most deaths in Cohort A (57.9%) were unrelated to NPC, whereas the majority
in Cohort B (66.7%) were NPC-related.
Conclusion
In appropriately selected patients aged 80 years or above with NPC, radical RT using modern IMRT
techniques is a viable treatment option, offering reasonable survival outcomes and an acceptable toxicity profile.
Chronological age alone should not be regarded as a barrier to radical treatment in NPC.
Key Words: Nasopharyngeal carcinoma; Octogenarians; Radiotherapy
中文摘要
80歲或以上鼻咽癌患者的治療結果
郭伯維、楊溢、佃穎恩、董煜
引言
對於年滿80歲或以上的鼻咽癌患者,其最佳治療方案仍具爭議,主要源於對根治性放射治療耐受性的顧慮。本研究旨在評估本港80歲或以上鼻咽癌患者的治療成效及相關毒性。
方法
本回顧性研究分析一所醫院於2009年1月至2023年12月期間治療的80歲或以上鼻咽癌患者資料。診斷時已出現遠處轉移者予以排除。研究分析內容包括患者特徵、治療成效及毒性情況。
結果
本研究共納入42名患者(年齡中位數83歲,介乎80至94歲),中位隨訪時間為20.3個月。整體患者的中位總生存期為22.8個月(95%置信區間:14.6-30.9個月),5年總生存率為23.8%。其中27名患者(64.3%)接受以調強放射治療進行的根治性放療,無人接受化療。在該組患者(A組)中,中位總生存期為41.3個月(95%置信區間:27.7-55.0個月),5年總生存率及癌症特異性生存率分別為38.1%及74.2%。3級或以上急性毒性發生率為22.2%;1名患者(3.7%)因治療相關毒性死亡。共有5名患者(18.5%)出現治療失敗,均為遠處轉移所致。接受非根治性放療的患者(B組)其中位總生存期為12.8個月(95%置信區間:10.9-14.7個月),且無人存活超過5年。A組多數死亡個案(57.9%)與鼻咽癌無關,而B組大多數死亡個案(66.7%)則與鼻咽癌相關。
結論
對於經審慎篩選的80歲或以上鼻咽癌患者,採用現代調強放射治療技術進行根治性放療屬可行治療選項,可帶來合理的生存成效及可接受的毒性水平。年齡本身不應被視為接受根治性治療的障礙。
INTRODUCTION
Nasopharyngeal carcinoma (NPC) is an epithelial
carcinoma originating from the nasopharyngeal mucosa.
This malignancy is most prevalent in Asia, accounting
for over 80% of global incident cases in 2022.[1] In
endemic regions, NPC incidence peaks in the 45-59
years age-group and declines thereafter.[2] Data from
the Hong Kong Cancer Registry indicate that in 2023,
approximately 4.9% of new NPC cases occurred in
patients aged 80 years or above.[3]
Standard treatment for NPC involves high-dose radical
radiotherapy (RT) of 66 to 70 Gy, often combined with
concurrent, induction, and/or adjuvant chemotherapy for
locally advanced disease.[4] [5] However, these treatment
guidelines are largely based on clinical studies that have
underrepresented or excluded older adult populations.
For instance, in a meta-analysis of chemotherapy in NPC,
only 13% of the cohort was aged 60 years or above.[6]
Older adults with NPC have worse survival outcomes
compared to their younger counterparts.[5] Previous
studies have reported 5-year overall survival (OS) rates ranging from 44% to 60% among patients aged
70 years or above with NPC,[7] [8] [9] whereas those aged 80
years or above exhibit a considerably lower survival
rate of approximately 30%.[10] Treating older adults with
NPC presents particular challenges due to increased co-morbidities,
nutritional issues, organ dysfunction, and
greater susceptibility to treatment-related toxicities.[11]
Despite these clinical challenges, studies specifically
addressing treatment outcomes and strategies in
older adults with NPC remain limited. Furthermore,
inconsistencies exist regarding the definition of ‘older
adults’ or ‘elderly’ across published studies, with age
thresholds typically ranging from 65 to 70 years.[7] [8] [9] [10] [12] [13] Notably, outcomes for the oldest patients with NPC,
specifically those aged 80 years or above, are rarely
reported. These much older patients may represent a
distinct subgroup, even within the broader geriatric
population. Huang et al[10] reported that patients aged
80 years or above with NPC had significantly worse
survival than those aged 65 to 69 years. This study aimed
to investigate treatment patterns and survival outcomes
in older adults aged 80 years or above with NPC in Hong
Kong.
METHODS
Patient Characteristics
We conducted a retrospective review of the medical
records of patients with NPC who received care at Tuen
Mun Hospital between 1 January 2009 and 31 December
2023. Patients aged 80 years or above at diagnosis with
histologically confirmed NPC were included. Those with
distant metastasis at initial diagnosis were excluded.
Data on demographics, disease status, co-morbidities,
and treatment outcomes were retrieved from electronic
patient records and analysed. Patients were categorised
into those who received radical RT to the nasopharynx
(Cohort A) and those who did not (Cohort B).
Staging and Evaluation
Patients underwent clinical evaluation, including history
taking and physical examination. Local and regional
staging was performed using magnetic resonance
imaging of the nasopharynx and neck and/or computed
tomography. Between 2009 and 2017, positron emission
tomography–computed tomography (PET-CT) was
selectively performed in patients with symptoms,
laboratory abnormalities, or chest radiograph findings
suggestive of distant metastasis. From 2018 onwards,
PET-CT has been routinely performed for all patients
with tumour (T) stage T4, nodal (N) stage N3, or T3N2
disease, as well as those with clinical suspicion of
metastatic disease, in accordance with Hospital Authority
(HA) standard indications.
NPC staging was performed according to the 8th
edition of the American Joint Committee on Cancer
(AJCC) staging manual.[14] Patients diagnosed prior to the
introduction of the AJCC 8th edition were retrospectively
re-staged. Patient performance status was assessed using
the Karnofsky Performance Status (KPS) Scale.[15] Co-morbidities
and overall health status were retrospectively
evaluated using the Adult Comorbidity Evaluation–27
(ACE-27),[16] the Charlson Comorbidity Index (CCI),[17]
and the modified Frailty Index-11 (mFI-11).[18]
Radiotherapy
All patients who received radical RT underwent
intensity-modulated radiotherapy (IMRT). Patients were
immobilised in the supine position using a thermoplastic
cast applied to the head and shoulders. A non-contrast
simulation computed tomography scan was acquired and
fused with the diagnostic magnetic resonance imaging
scan. Target volumes were contoured according to
international guidelines.[19] [20] The gross tumour volume
encompassed the primary tumour and enlarged lymph nodes. Clinical target volumes (CTVs) were defined
as high-risk, intermediate-risk, and low-risk CTVs.
The high-risk CTV included the gross tumour volume
plus a 5-mm margin and the whole nasopharynx. The
intermediate-risk CTV included the high-risk CTV
plus a 5-mm margin and was expanded to cover sites at
risk of microscopic extension, as well as the involved
nodal levels. The low-risk CTV included uninvolved
but potentially at-risk nodal levels. Prescribed doses to
the high-, intermediate-, and low-risk CTVs were 70
Gy, 60 Gy, and 54 Gy, respectively, delivered in 33
daily fractions using the simultaneous integrated boost
technique. A 3-mm margin from CTV to planning target
volume was added to account for setup uncertainty. The
planning target volume was subsequently cropped 3 mm
from the external body contour, and midline avoidance
structures were created to minimise skin and mucosal
toxicities.
Treatment Evaluation and Follow-up
Patients undergoing radical RT were monitored
weekly during treatment. RT-related toxicities were
prospectively recorded and graded according to the
National Cancer Institute Common Terminology
Criteria for Adverse Events, version 5.0.[21] Treatment
response evaluations were conducted 8 to 12 weeks after
completion of RT and included physical examination
and nasopharyngoscopy. For patients treated after 2021,
routine magnetic resonance imaging of the nasopharynx
and neck was also performed in addition to physical
examination and nasopharyngoscopy. Patients were
subsequently followed up at regular 3- to 6-month
intervals by oncologists and otolaryngologists. Each visit
included a clinical examination and nasopharyngoscopy.
Further investigations (e.g., imaging and blood tests)
were performed when recurrence was suspected.
Cause-of-Death Analysis
Causes of death were determined from electronic
medical records and classified into four categories: (1)
NPC-related death, defined as death resulting from the
primary NPC or its metastases; (2) treatment-related
death, defined as death due to complications arising from
NPC treatment; (3) non-NPC death, defined as death
from causes unrelated to the cancer or its treatment; and
(4) unknown, defined as death for which a definitive
cause could not be established based on the available
clinical information. Classification as NPC-related
death required the terminal event to be attributable to
metastatic disease or to a documented complication
of symptomatic or progressive local disease. When competing causes were present, the primary cause was
determined based on clinical documentation, imaging
findings, and its temporal relationship to treatment.
For example, aspiration pneumonia occurring with
documented dysphagia secondary to progressive local
NPC was classified as an NPC-related death, whereas
aspiration pneumonia in the absence of documented
treatment-related dysphagia or residual tumour was
classified as a non-NPC death.
Statistical Analyses
OS was defined as the interval from the date of
histological diagnosis to the date of death. Progression-free
survival was defined as the interval from histological
diagnosis to the date of disease progression (including
local, regional, or distant progression) or death. Cancer-specific
survival (CSS) was defined as the interval from
histological diagnosis to the date of NPC-related death.
Survival rates were estimated using the Kaplan–Meier
method. Univariable and multivariable Cox proportional
hazards regression models were used to identify factors
associated with survival. Variables with p < 0.05 in
univariable analysis and those deemed clinically relevant
were considered for multivariable modelling. To reduce
multicollinearity, closely related clinical variables
were not included simultaneously in the multivariable
model, such as individual TNM components and overall
stage or measures of performance status and frailty.
Hazard ratios (HRs) with 95% confidence intervals
(95% CIs) were reported. The Mann–Whitney U test
was used to compare distributions of ordinal variables
between patient cohorts. For categorical variables, the
Chi squared test or Fisher’s exact test was applied, as
appropriate. All statistical tests were two-sided, with a
significance threshold of p < 0.05. Statistical analyses
were performed using SPSS (Windows version 26.0;
IBM Corp, Armonk [NY], United States).
RESULTS
Patient Characteristics and Treatment
In total, 42 patients were included. Patient characteristics are summarised in Table 1. The median age was 83
years (range, 80-94) and 29 patients (69.0%) were men.
Most patients presented with stage III disease (33.3%),
followed by stage II (26.2%), stage IVa (19.0%), and
stage I (11.9%). A higher proportion of patients in Cohort
A underwent PET-CT for distant metastasis screening
compared with Cohort B (29.6% vs. 6.7%). Staging
information was unavailable for four patients (9.5%), all
of whom were in Cohort B.
Table 1. Baseline patient and disease characteristics.
Overall, 27 patients (64.3%) received radical RT to the
nasopharynx (Cohort A), while 15 patients (35.7%) did
not (Cohort B) [Table 1]. Reasons for not undergoing
radical RT included patient refusal (n = 9), concomitant
malignancy (n = 1), and medical unfitness for radical
treatment (n = 5). Of the 15 patients in Cohort B, two
(13.3%) received palliative RT. Chemotherapy was not
administered to any patients in either cohort.
Cohort A had significantly more patients with a KPS
score ≥70% compared with Cohort B. No significant
differences were observed in ACE-27 scores or CCI
scores. Although a higher proportion of patients in
Cohort B had a mFI-11 score ≥0.27 (categorised as
frail) compared with Cohort A, this difference was not
statistically significant (Table 1).
Survival Outcome and Prognostic Factors
At the time of analysis, eight patients (19.0%) were alive.
The median follow-up duration was 20.3 months (range,
1.5-138) for the entire cohort, and 28.2 months for
those who were alive. The median OS was 22.8 months
(95% CI = 14.6-30.9).
Among patients who received radical RT (Cohort A), the
median OS was 41.3 months (95% CI = 27.7-55.0). The
median CSS was not reached. The median progression-free
survival was 39.6 months (95% CI = 22.4-56.7).
The 5-year OS and CSS rates were 38.1% and 74.2%,
respectively (Figure 1).
Figure 1. (a) Overall survival and (b) cancer-specific survival in Cohort A.
Among patients who did not receive radical RT (Cohort
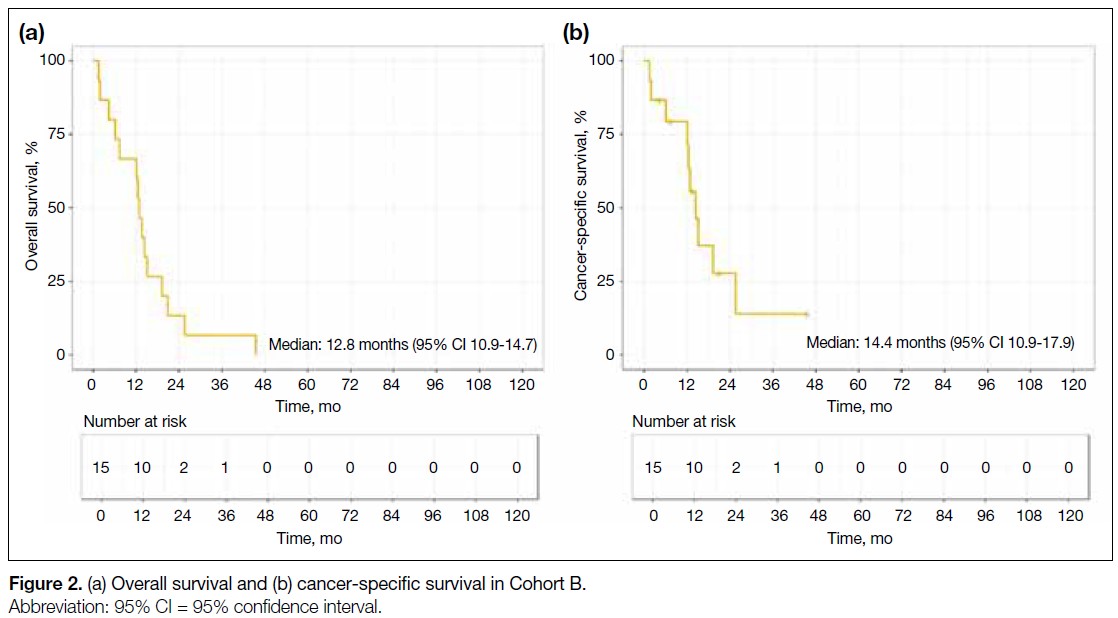
B), the median OS was 12.8 months (95% CI = 10.9-14.7) and the median CSS was 14.4 months (95% CI = 10.9-17.9). No patient in Cohort B survived to 5 years
(Figure 2).
Figure 2. (a) Overall survival and (b) cancer-specific survival in Cohort B.
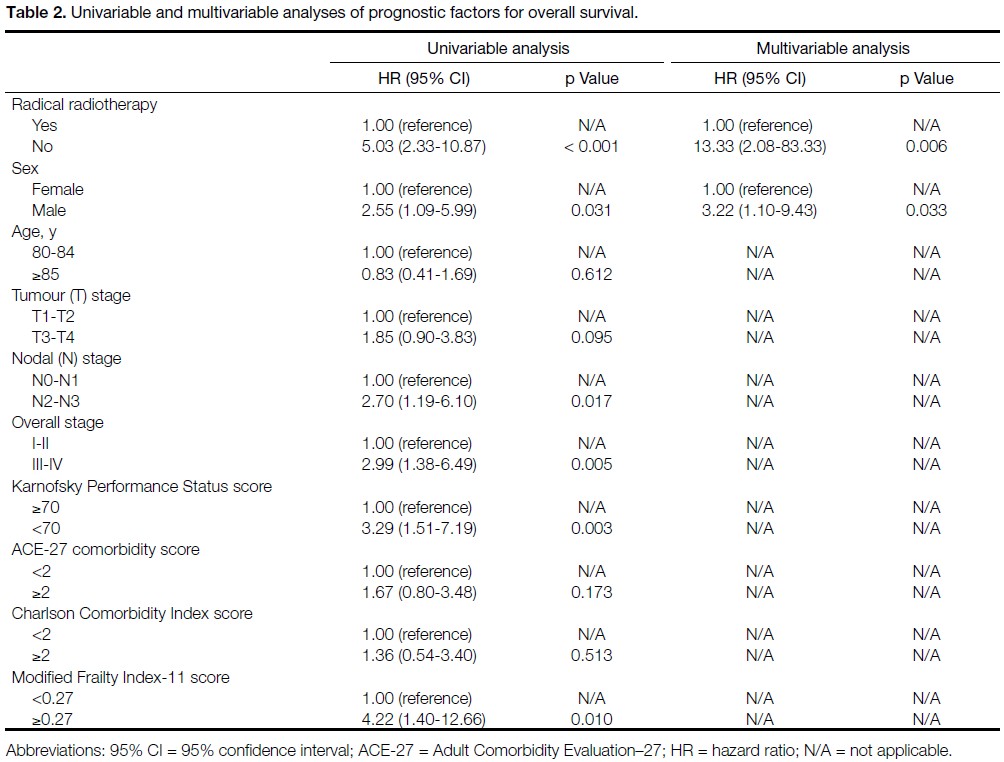
Univariable analysis identified several factors
significantly associated with worse OS, including
absence of radical RT (no vs. yes; HR = 5.03, p < 0.001),
male sex (male vs. female; HR = 2.55, p = 0.031),
advanced nodal stage (N2-3 vs. N0-N1; HR = 2.70,
p = 0.017), advanced overall AJCC stage (stage III-IV vs.
stage I-II; HR = 2.99, p = 0.005), poor KPS score (<70%
vs. ≥70%; HR = 3.29, p = 0.003), and frailty based on the
mFI-11 (mFI-11 ≥0.27 vs. <0.27; HR = 4.22, p = 0.010).
On multivariable analysis, no receipt of radical RT
(HR = 13.33, p = 0.006) and male sex (HR = 3.22,
p = 0.033) were independently associated with worse OS
(Table 2).
Table 2. Univariable and multivariable analyses of prognostic factors for overall survival.
Cause-of-Death Analysis
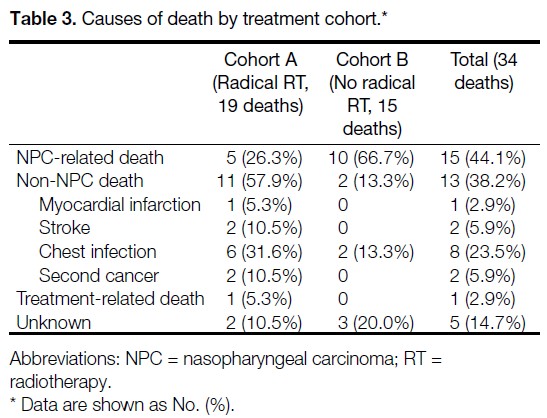
Among the 34 patients who died, the most common
cause of death was NPC-related death (n = 15, 44.1%),
followed by non-NPC death (n = 13, 38.2%). Treatment-related
mortality occurred in one patient (2.9% of
deaths), and the cause of death was unknown in five patients (14.7%). The causes of death among patients
who underwent radical RT (Cohort A) and those who
did not (Cohort B) are summarised in Table 3. The two
cohorts demonstrated distinct cause-of-death profiles. In
Cohort A, the most common cause of death was non-NPC death (n = 11, 57.9%), followed by NPC-related death (n = 5, 26.3%), unknown causes (n = 2, 10.5%),
and treatment-related death (n = 1, 5.3%). Among
patients in Cohort A who died of non-NPC causes, the
median interval from the last day of RT to death was
36.9 months (interquartile range, 16.1-71.0). In Cohort B, the majority of patients died of NPC-related causes
(n = 10, 66.7%); two patients (13.3%) died of non-NPC
causes and three patients (20%) died of unknown causes.
Detailed descriptions of the circumstances of death for
individual cases are provided in the online supplementary Table.
Table 3. Causes of death by treatment cohort.
Radical Radiotherapy
Treatment Outcomes
Among the 27 patients in Cohort A who underwent
radical RT, the majority (96.3%) completed the planned
course of treatment. Local treatment response to RT was
documented in 22 patients; of these, 95.5% achieved a
complete response. One patient had persistent disease
in the nasopharynx and achieved successful salvage
with brachytherapy. No local or regional relapse was
observed. Five patients (18.5%) developed distant
recurrence, with a median time to onset of distant
metastasis of 17.6 months (range, 8.3-34.0). None of
these patients received further systemic anticancer
therapy for metastatic disease.
Acute and Late Treatment Toxicities
Table 4 summarises the acute toxicities observed in
Cohort A. Grade ≥3 acute RT toxicities, defined as
those occurring during RT or within 3 months after
RT, were observed in six of 27 patients (22.2%). The most frequently reported acute toxicities were mucositis
(all grades, 96.3%; grade ≥3, 14.8%) and radiation
dermatitis (all grades, 77.8%; grade ≥3, 3.7%). Seven
patients (25.9%) required unplanned hospital admission during treatment: four for grade 3 mucositis, one for
grade 3 dermatitis, one for feeding tube insertion to
support nutrition in the absence of clinically significant
mucositis, and one for a chest infection during the sixth
RT fraction (this patient subsequently died). The fatal
chest infection resulted in a treatment-related mortality
rate of 3.7%. Two patients (7.4%) died within 90 days of
completing RT.
Table 4. Acute treatment-related toxicities in Cohort A (n = 27).
Grade ≥3 late RT toxicities (defined as those occurring
more than 3 months after RT) were observed in 14.8% of
patients, the majority of which involved severe hearing
loss. One patient (3.7%) required long-term feeding tube
support due to dysphagia.
DISCUSSION
In this retrospective study of patients aged 80 years or
above with NPC, radical RT using IMRT resulted in
a median OS of 41.3 months and a 5-year OS rate of
38.1%, with manageable toxicity. To our knowledge,
this is the first study to specifically evaluate treatment
outcomes and toxicities in this group of patients, thereby
addressing a critical knowledge gap.
The treatment of NPC in older adults is challenging
and frequently overlooked, as this population is
often excluded from or underrepresented in clinical
trials. Older adults represent a heterogeneous group
characterised by a wide range of co-morbidities and
varying degrees of frailty. Management of NPC in
this group is often complex, and survival outcomes are
generally worse compared with those of their younger
counterparts.
Yang et al[8] reported outcomes in patients aged 70
years or above with NPC, most of whom received RT
combined with chemotherapy, achieving a 5-year OS rate of 59.5%. Notably, only 65.3% of patients in that
cohort received IMRT, and most were younger than 75
years.[8] Jin et al[7] examined a similar cohort of patients
aged 70 years or above with NPC who were treated
exclusively with IMRT and reported a 5-year OS rate
of 54%; however, chemotherapy was administered to
42.8% of patients, and the maximum age in that cohort
was 73 years. Patients aged 80 years or above represent
an especially challenging subgroup, even within the
broader geriatric population. In a National Cancer
Database analysis by Huang et al,[10] patients aged 80
years or above with NPC who received radical RT had
a 5-year OS rate of 31.3%. Toxicity outcomes were not
reported in that study.
Due to prevalent co-morbidities and reduced bone
marrow reserve, older patients with NPC often
have limited tolerance for chemotherapy, whether
administered as induction therapy or concurrently with
RT. The benefit of chemotherapy in this population
remains a subject of debate. While some retrospective
studies have reported improved outcomes with the
addition of chemotherapy to RT in older adults,[12] [22] [23]
others have shown no clear survival advantage.[7] [24] [25] In
clinical practice, chemotherapy is seldom administered
to patients aged 80 years or above.[8] Indeed, in our cohort,
no patient in this age-group received chemotherapy.
High-dose RT to the head and neck region can be
potentially morbid, and treatment tolerance is a
significant concern, particularly among older adults. A
study by Sze et al[9] reported significantly higher rates of
acute grade 3 toxicities, RT incompletion, and 90-day
mortality in patients aged 70 years or above with NPC
compared with younger patients. As a result, clinicians
may be hesitant to offer radical RT to patients aged 80
years or above.
Our findings demonstrated that radical RT is associated
with meaningful survival outcomes in patients aged 80
years or above. Among those who received radical RT,
a median OS exceeding 3 years and a 5-year OS rate of
38.1% are encouraging, suggesting that radical RT can
provide reasonable survival even for octogenarians.
Our study also showed that patients who did not receive
radical RT had poorer outcomes, with a median OS of
only 12.8 months. However, direct survival comparisons
between these two cohorts should be interpreted with
caution due to important baseline differences. Patients
in Cohort B had significantly worse performance status, with a greater proportion exhibiting a KPS score below
70 compared with Cohort A. Although no significant
differences were observed between cohorts in terms of
co-morbidity indices, inherent disparities undoubtedly
existed. These differences may introduce confounding
bias, whereby the observed survival advantage of
radical RT may be partially attributable to baseline
patient characteristics. Despite these limitations, the
considerable difference in outcomes suggests a potential
benefit of radical RT in appropriately selected older
adults.
Perhaps more importantly, the cause-of-death analysis
offers additional insight into the potential benefit of
radical RT. Among patients who received radical RT,
most deaths were due to medical conditions unrelated
to NPC or its treatment, whereas in the non-radical RT
group, the majority of deaths were attributable to NPC
progression.
These findings may assist clinicians in discussions
with patients and caregivers, facilitating personalised
management strategies. It is important for clinicians
to recognise the potential benefits of radical RT in
appropriately selected patients, ensuring that advanced
age alone does not preclude access to potentially curative
treatment.
IMRT has become the standard of care for NPC, providing
optimal tumour coverage while sparing critical organs
at risk.[26] It is associated with improved tumour control
and a reduction in both acute and late toxicities.[27] [28] In
our study, however, grade ≥3 acute toxicities remained
common (22.2%) among patients undergoing radical RT
with IMRT. It is important to recognise that older adults
are at increased risk of developing severe treatment-related
toxicities; all toxicities should be identified
promptly and managed proactively. In particular,
RT-induced mucositis and dysphagia can lead to life-threatening
infectious complications, as demonstrated
by the single grade 5 toxicity observed in our cohort.
Intensive clinical monitoring throughout treatment—combined with appropriate supportive medications
and multidisciplinary collaboration involving nurses,
dietitians, and speech therapists—is essential. Vigilance
in nutritional management is particularly important, as
older adults may already be at high risk of sarcopenia and
have limited physiological reserves.[29] Clinicians should
maintain a low threshold for feeding tube insertion
during RT, and a prophylactic approach to nutritional
support may be considered.
Although the incidence of grade ≥3 acute toxicities was
relatively high, it was not prohibitive. In our study, the
rates of grade ≥3 dermatitis and mucositis were 3.7%
and 14.8%, respectively, both of which appear lower
than previously reported figures of 21.6% to 22.3%
for grade ≥3 dermatitis and 18.9% to 68% for grade ≥3
mucositis.[9] [25] This difference is likely attributable to our
institutional protocol, which routinely includes a 3-mm
skin clip and the creation of midline structure avoidance
volumes. In the present study, the treatment-related
mortality rate was 3.7% and the 90-day mortality rate
was 7.4%, a figure comparable to the 7.8% reported by
Sze et al[9] in patients aged above 70 years.
Late grade ≥3 RT toxicities were also infrequent in
our study; only one patient remained dependent on a
feeding tube. This observation may be partly explained
by the relatively short follow-up period and limited
survival duration, which may have precluded the full
manifestation of late toxicities. Another contributing
factor is that all patients received IMRT, which delivers
a more conformal dose distribution to the target volume
while better sparing adjacent normal tissues.[30]
Although this study focuses on patients aged 80 years
or above, it is essential for clinicians to recognise that
chronological age alone should not serve as the sole
criterion for risk stratification. Co-morbidity and frailty
assessments provide critical information to guide the
management of older patients with NPC. Comprehensive
geriatric assessment, considered the gold standard for
evaluating older adults, is recommended by both the
International Society of Geriatric Oncology[31] and the
American Society of Clinical Oncology[32] to support
treatment decision making. However, comprehensive
geriatric assessment is not widely implemented due to
its time-consuming nature. Several tools are available
for co-morbidity assessment, including the CCI,[17] the
ACE-27,[16] and the mFI-11.[18] Notably, both ACE-27
and CCI have been associated with survival outcomes.
For example, Huang et al[10] identified CCI score ≥2 was
an independent prognostic factor for mortality, while
higher ACE-27 scores have been associated with poorer
survival outcomes.[7] [9] [33] In our study, there was a trend
towards worse survival outcomes in patients with higher
CCI, ACE-27, and mFI-11 scores; however, none of
these associations reached statistical significance in
multivariable analysis, likely due to the small sample
size.
Several questions remain unanswered. Although radical RT of 70 Gy remains the current standard of care,[4]
it is unclear whether this ‘one-size-fits-all’ approach
is appropriate for older adults with NPC. A logical
consideration is RT dose de-escalation, aiming to balance
optimal tumour control with minimised toxicity. Wang
et al[34] demonstrated comparable outcomes between
standard-dose RT (70 Gy) and reduced-dose RT (53-67
Gy) in patients with T1 to T3 NPC. However, there is
currently no robust evidence supporting RT dose de-escalation
specifically in older adults with NPC. Future
studies are warranted to explore the optimal dose and
fractionation schedules for this population.
Strengths and Limitations
This study has several strengths. To our knowledge, it
is the first to specifically report treatment outcomes and
toxicities in patients aged 80 years or above with NPC.
All treatments were delivered using modern IMRT
techniques, and acute and late treatment-related adverse
events were prospective documented.
This study has several important limitations. First,
inherent selection bias exists in this retrospective cohort
comparison, as patients who received radical RT were
likely to have been healthier overall, despite similar co-morbidity
scores, and treatment decisions were influenced
by unmeasured factors, including clinician judgement
and patient preference. Second, comprehensive screening
for distant metastases was not performed in some
patients, particularly those who did not receive radical
RT. It is therefore possible that a higher proportion of
patients in Cohort B had undiagnosed stage IVb disease
at presentation, which may have contributed to poorer
outcomes. Third, the relatively small sample size limits
the statistical power of the analysis and precludes the
application of more sophisticated statistical methods,
such as causal inference approaches (e.g., propensity
score matching). Fourth, the follow-up duration was
relatively short and some late toxicities may not yet have
emerged. Fifth, formal geriatric assessments (such as
comprehensive geriatric assessment) and quality-of-life
evaluations were not conducted. Prospective multicentre
studies with larger sample sizes, standardised geriatric
assessments, and quality-of-life measurements are
warranted to validate these findings and better inform
clinical practice.
CONCLUSION
In appropriately selected patients aged 80 years or above
with NPC, radical RT using modern IMRT techniques
represents a viable treatment option, offering reasonable survival outcomes with an acceptable toxicity profile.
Chronological age alone should not be regarded as a
barrier to radical treatment in NPC.
REFERENCES
1. International Agency for Research on Cancer, World Health Organization. Ferlay J, Ervik M, Lam F, Laversanne M, Colombet M, Mery L, et al. Nasopharyngeal cancer statistics. Global Cancer Observatory: Cancer Today. 2024. Available from: https://gco.iarc.who.int/media/globocan/factsheets/cancers/4-nasopharynx-fact-sheet.pdf. Accessed 9 Feb 2026.
2. Chang ET, Ye W, Zeng YX, Adami HO. The evolving epidemiology of nasopharyngeal carcinoma. Cancer Epidemiol Biomarkers Prev. 2021;30:1035–47.
Crossref
3. Hospital Authority. Hong Kong Cancer Registry. Cancer statistics query system (All ages). Available from: https://www3.ha.org.hk/cancereg/allages.asp. Accessed 9 Feb 2025.
4. Lee VH, Lam KO, Lee AW. Chapter 10—Standard of Care for Nasopharyngeal Carcinoma (2018–2020). In: Lee AW, Lung ML, Ng WT, editors. Nasopharyngeal Carcinoma: From Etiology to Clinical Practice. London: Academic Press; 2019: 205–38.
Crossref
5. Bossi P, Chan AT, Licitra L, Trama A, Orlandi E, Hui EP, et al. Nasopharyngeal carcinoma: ESMO–EURACAN Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2021;32:452–65.
Crossref
6. Blanchard P, Lee A, Marguet S, Leclercq J, Ng WT, Ma J, et al. Chemotherapy and radiotherapy in nasopharyngeal carcinoma: an update of the MAC–NPC meta-analysis. Lancet Oncol. 2015;16:645–55.
Crossref
7. Jin YN, Zhang WJ, Cai XY, Li MS, Lawrence WR, Wang SY, et al. The characteristics and survival outcomes in patients aged 70 years and older with nasopharyngeal carcinoma in the intensity-modulated radiotherapy era. Cancer Res Treat. 2019;51:34–42.
Crossref
8. Yang G, Huang J, Sun J, Wang L. Elderly nasopharyngeal carcinoma patients (aged ≥70 years): survival and treatment strategies. Cancer Med. 2023;12:19523–9.
Crossref
9. Sze HC, Ng WT, Chan OS, Shum TC, Chan LL, Lee AW. Radical radiotherapy for nasopharyngeal carcinoma in elderly patients: the importance of co-morbidity assessment. Oral Oncol. 2012;48:162–7.
Crossref
10. Huang Y, Chen W, Haque W, Verma V, Xing Y, Teh BS, et al. The impact of comorbidity on overall survival in elderly nasopharyngeal carcinoma patients: a National Cancer Data Base analysis. Cancer Med. 2018;7:1093–101.
Crossref
11. Mascarella MA, Vendra V, Sultanem K, Tsien C, Shenouda G, Sridharan S, et al. Predicting short-term treatment toxicity in head and neck cancer through a systematic review and meta-analysis. J Geriatr Oncol. 2024;15:102064.
Crossref
12. Liu H, Chen QY, Guo L, Tang LQ, Mo HY, Zhong ZL, et al. Feasibility and efficacy of chemoradiotherapy for elderly patients
with locoregionally advanced nasopharyngeal carcinoma: results
from a matched cohort analysis. Radiat Oncol. 2013;8:70.
Crossref
13. Wen YF, Sun XS, Yuan L, Zeng LS, Guo SS, Liu LT, et al. The impact of Adult Comorbidity Evaluation–27 on the clinical
outcome of elderly nasopharyngeal carcinoma patients treated with
chemoradiotherapy or radiotherapy: a matched cohort analysis. J Cancer. 2019;10:5614–21.
Crossref
14. Amin MB, Edge S, Greene F, Byrd DR, Brookland RK, Washington MK, et al (eds). AJCC Cancer Staging Manual. 8th ed. New York: Springer; 2017.
15. MDCalc. Karnofsky Performance Status Scale. Available from: https://www.mdcalc.com/calc/3168/karnofsky-performance-status-scale. Accessed 6 Feb 2026.
16. British Geriatrics Society. Adult Comorbidity Evaluation–27. Available from: https://www.bgs.org.uk/sites/default/files/content/attachment/2018-07-05/adult_comorbidity_evaluation.pdf. Accessed 6 Feb 2026.
17. MDCalc. Charlson Comorbidity Index (CCI). Available from: https://www.mdcalc.com/calc/3917/charlson-comorbidity-index-cci. Accessed 6 Feb 2026.
18. Evidencio. Modified Frailty Index. Available from: https://www.evidencio.com/models/show/1777. Accessed 6 Feb 2026.
19. Lee AW, Ng WT, Pan JJ, Poh SS, Ahn YC, AlHussain H, et al. International guideline for the delineation of the clinical target
volumes (CTV) for nasopharyngeal carcinoma. Radiother Oncol. 2018;126:25–36.
Crossref
20. Grégoire V, Ang K, Budach W, Grau C, Hamoir M, Langendijk JA, et al. Delineation of the neck node levels for head and neck tumors:
a 2013 update. DAHANCA, EORTC, HKNPCSG, NCIC CTG,
NCRI, RTOG, TROG consensus guidelines. Radiother Oncol.
2014;110:172-81.
Crossref
21.United States Department of Health and Human Services. Common
Terminology Criteria for Adverse Events (CTCAE) Version 5.0.
2017 Nov 27. Available from: https://dctd.cancer.gov/research/ctep-trials/for-sites/adverse-events/ctcae-v5-5x7.pdf. Accessed 6 Feb 2026.
22. Zeng Q, Xiang YQ, Wu PH, Lv X, Qian CN, Guo X. A matched
cohort study of standard chemo-radiotherapy versus radiotherapy
alone in elderly nasopharyngeal carcinoma patients. PLoS One.
2015;10:e0119593.
Crossref
23. Lu Y, Hua J, Yan F, Jiang C, Piao Y, Ye Z, et al. Combined
radiotherapy and chemotherapy versus radiotherapy alone in elderly
patients with nasopharyngeal carcinoma: a SEER population-based
study. Medicine (Baltimore). 2021;100:e26629.
Crossref
24. Lyu Y, Ni M, Zhai R, Kong F, Du C, Hu C, et al. Clinical
characteristics and prognosis of elderly nasopharyngeal carcinoma
patients receiving intensity-modulated radiotherapy. Eur Arch
Otorhinolaryngol. 2021;278:2549-57.
Crossref
25. Sommat K, Yit NL, Wang F, Lim JH. Impact of comorbidity
on tolerability and survival following curative intent intensity
modulated radiotherapy in older patients with nasopharyngeal
cancer. J Geriatr Oncol. 2018;9:352-8.
Crossref
26. Lee AW, Ng WT, Chan LL, Hung WM, Chan CC, Sze HC, et al.
Evolution of treatment for nasopharyngeal cancer—success and
setback in the intensity-modulated radiotherapy era. Radiother
Oncol. 2014;110:377-84.
Crossref
27. Fatima K, Andleeb A, Sofi MA, Rasool MT, Fir A, Nasreen S, et al.
Clinical outcome of intensity-modulated radiotherapy versus
two-dimensional conventional radiotherapy in locally advanced
nasopharyngeal carcinoma: comparative study at SKIMS Tertiary
Care Institute. J Cancer Res Ther. 2022;18:133-9.
Crossref
28. Peng G, Wang T, Yang KY, Zhang S, Zhang T, Li Q, et al.
A prospective, randomized study comparing outcomes and
toxicities of intensity-modulated radiotherapy vs. conventional
two-dimensional radiotherapy for the treatment of nasopharyngeal
carcinoma. Radiother Oncol. 2012;104:286-93.
Crossref
29. Morse RT, Ganju RG, Gan GN, Cao Y, Neupane P, Kakarala K,
et al. Sarcopenia and treatment toxicity in older adults undergoing
chemoradiation for head and neck cancer: identifying factors to
predict frailty. Cancers (Basel). 2022;14:2094.
Crossref
30. Lai SZ, Li WF, Chen L, Luo W, Chen YY, Liu LZ, et al. How
does intensity-modulated radiotherapy versus conventional
two-dimensional radiotherapy influence the treatment results in
nasopharyngeal carcinoma patients? Int J Radiat Oncol Biol Phys.
2011;80:661-8.
Crossref
31. Wildiers H, Heeren P, Puts M, Topinkova E, Janssen-Heijnen ML,
Extermann M, et al. International Society of Geriatric Oncology
consensus on geriatric assessment in older patients with cancer. J
Clin Oncol. 2014;32:2595-603.
Crossref
32. Mohile SG, Dale W, Somerfield MR, Hurria A. Practical assessment
and management of vulnerabilities in older patients receiving
chemotherapy: ASCO Guideline for Geriatric Oncology Summary.
J Oncol Pract. 2018;14:442-6.
Crossref
33. Guo R, Chen XZ, Chen L, Jiang F, Tang LL, Mao YP, et al.
Comorbidity predicts poor prognosis in nasopharyngeal carcinoma:
development and validation of a predictive score model. Radiother
Oncol. 2015;114:249-56.
Crossref
34. Wang X, Wang Y, Jiang S, Zhao J, Wang P, Zhang X, et al.
Safety and effectiveness of de-escalated radiation dose in T1-3
nasopharyngeal carcinoma: a propensity matched analysis. J
Cancer. 2019;10:5057-64.
Crossref