Revisiting Preoperative Evaluation of the Inferior Vena Cava in Abdominal Malignancies: A Pictorial Essay
PICTORIAL ESSAY
Hong Kong J Radiol 2026;29:Epub 6 March 2026
Revisiting Preoperative Evaluation of the Inferior Vena Cava in Abdominal Malignancies: A Pictorial Essay
A Mandava1, V Koppula1, M Kandati1, AK Reddy1, H Kacharagadla1, SR Thammineedi2
1 Department of Radiodiagnosis, Basavatarakam Indo American Cancer Hospital and Research Institute,
Hyderabad, India
2 Department of Surgical Oncology, Basavatarakam Indo American Cancer Hospital and Research Institute,
Hyderabad, India
Correspondence: Dr A Mandava, Department of Radiodiagnosis, Basavatarakam Indo American Cancer Hospital and Research
Institute, Hyderabad, India. Email: dranitha@basavatarakam.org
Submitted: 6 August 2025; Accepted: 23 November 2025. This version may differ from the final version when published in an issue.
Contributors: All authors designed the study, acquired the data, analysed the data, drafted the manuscript, and critically revised the manuscript
for important intellectual content. All authors had full access to the data, contributed to the study, approved the final version for publication, and
take responsibility for its accuracy and integrity.
Conflicts of Interest: All authors have disclosed no conflicts of interest.
Funding/Support: This study received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Data Availability: All data generated or analysed during the present study are available from the corresponding author on reasonable request.
Ethics Approval: The study was approved by the Institutional Ethics Committee of Basavatarakam Indo American Cancer Hospital and Research
Institute, India (Ref No.: IEC/2021/55). A waiver of informed patient consent was granted by the Committee as the study involved minimal risk
and non-identifiable data were used.
Declaration: A few of the images were presented as part of scientific exhibit in Radiological Society of North America Annual Meeting 2023,
Chicago [IL], United States, 26-30 November 2023.
INTRODUCTION
Inferior vena cava (IVC) is the largest vein in the
body, draining blood from the lower extremities,
pelvis, and abdomen into the right atrium. Accurate
anatomical assessment is crucial when planning
vascular interventions, resections, anastomoses,
and reconstructions that form an integral part of the
surgical management of abdominopelvic malignancies.
Anomalies and variants can complicate access to the IVC
and its tributaries during interventional procedures and
filter placement. Given that abdominopelvic oncological
surgeries require extensive dissections, unawareness
of vascular involvement and congenital anomalies
can lead to inadvertent injuries with catastrophic
outcomes. Contrast-enhanced computed tomography
with reconstruction is the gold-standard non-invasive
investigation for presurgical mapping; ultrasound with colour Doppler, magnetic resonance imaging, and
positron emission tomography/computed tomography
often play complementary roles in evaluating the IVC
and its draining veins. This pictorial essay presents
several illustrative cases from our experience at a tertiary
care cancer centre in India.
ANATOMY AND VARIANTS
The embryogenesis and development of the IVC is a
complex process, and multiple congenital variations
can arise from abnormal persistence or regression of
embryological veins (Table 1).[1] [2] [3] [4] [5] [6] [7] [8] These congenital
anomalies are collectively present in 4% of the
population.[2] [3] [4] [5] [6] [7] [8] The most common clinically significant
variations include duplication of the IVC and absence
or agenesis (interruption) of the IVC with prominent
hemiazygos-azygos pathways[2] (Figures 1, 2, 3, 4 and 5). Because visceral thoracic and abdominal organs demonstrate leftright
anatomical asymmetry, awareness of discrepancies
in laterality and venous drainage into the IVC—such
as in situs inversus and heterotaxy syndromes—is
critical before undertaking biliary, hepatic, and gastric
surgeries (Figure 6). Variations in renal vein anatomy
are often asymptomatic and overlooked but are crucial
during renal or adrenal surgeries and retroperitoneal dissections. Anomalous veins and collateral vessels may
be misdiagnosed as lymphadenopathy; hence, contrast
imaging is essential in all cases of malignancy (Figures 7, 8 and 9).
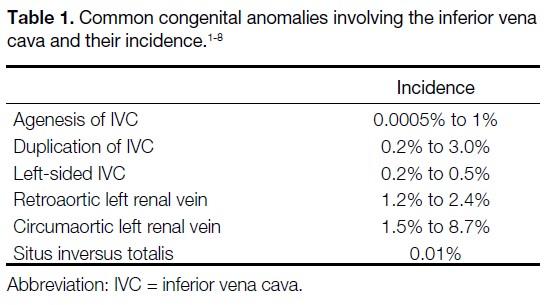
Table 1. Common congenital anomalies involving the inferior vena cava and their incidence.[1] [2] [3] [4] [5] [6] [7] [8]
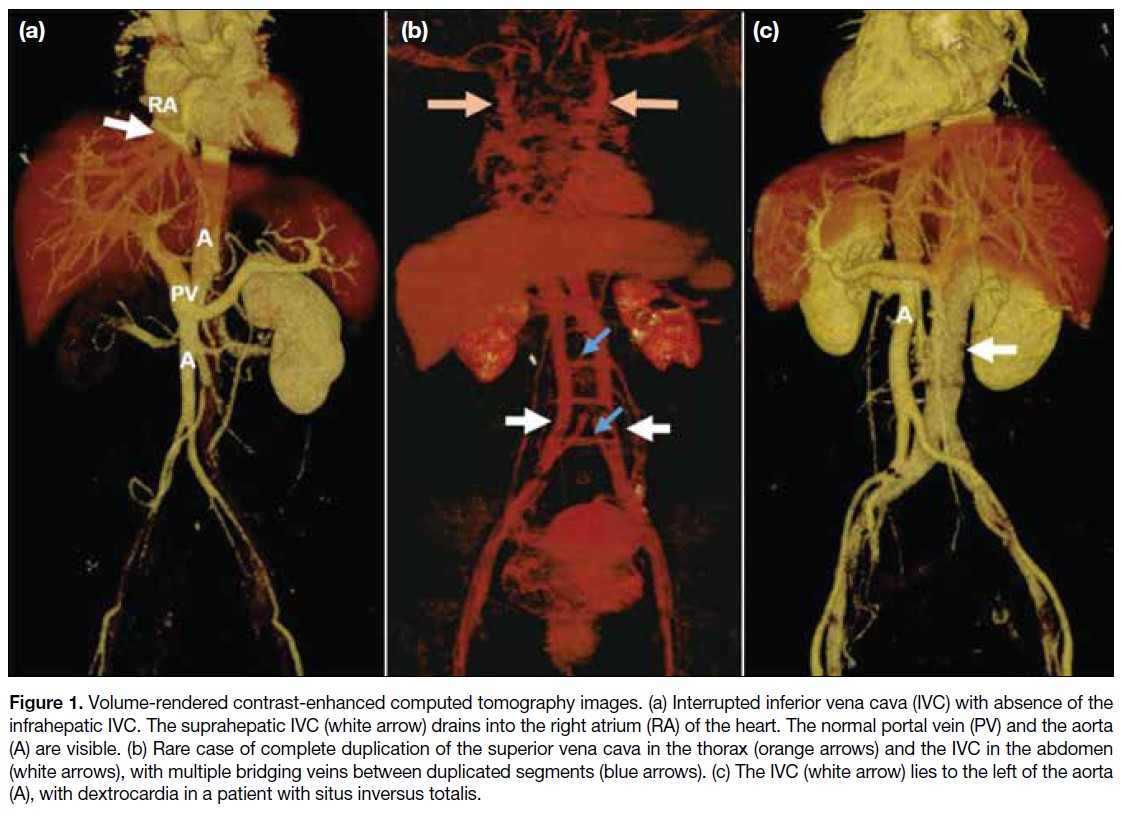
Figure 1. Volume-rendered contrast-enhanced computed tomography images. (a) Interrupted inferior vena cava (IVC) with absence of the
infrahepatic IVC. The suprahepatic IVC (white arrow) drains into the right atrium (RA) of the heart. The normal portal vein (PV) and the aorta
(A) are visible. (b) Rare case of complete duplication of the superior vena cava in the thorax (orange arrows) and the IVC in the abdomen
(white arrows), with multiple bridging veins between duplicated segments (blue arrows). (c) The IVC (white arrow) lies to the left of the aorta
(A), with dextrocardia in a patient with situs inversus totalis.
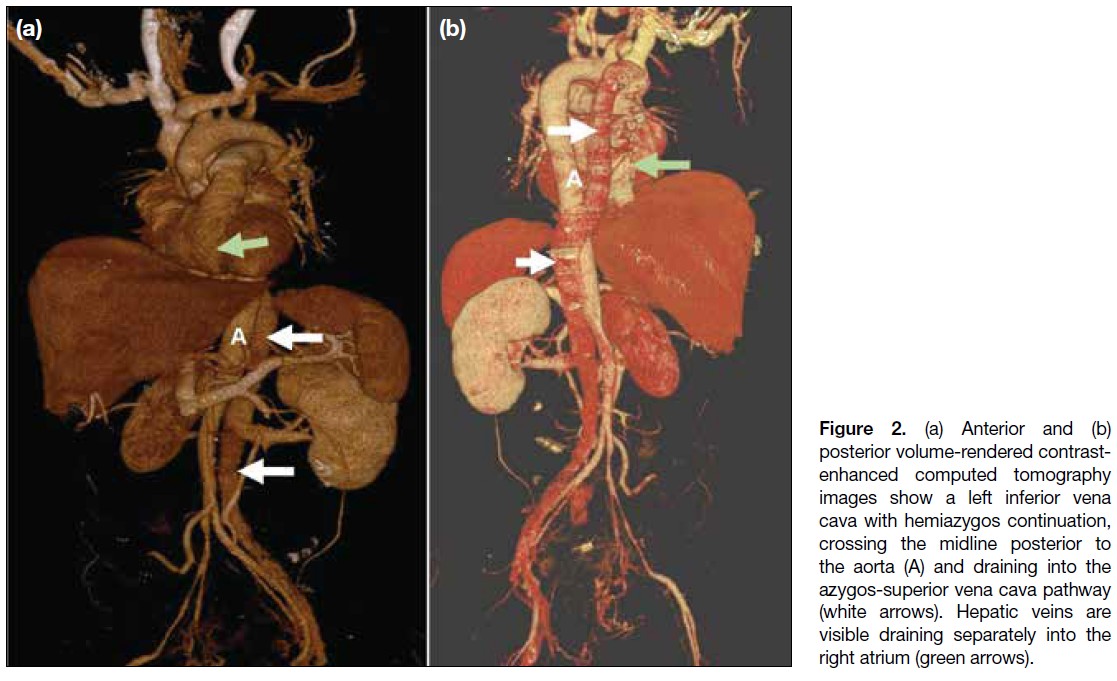
Figure 2. (a) Anterior and (b)
posterior volume-rendered contrast-enhanced
computed tomography
images show a left inferior vena
cava with hemiazygos continuation,
crossing the midline posterior to
the aorta (A) and draining into the
azygos-superior vena cava pathway
(white arrows). Hepatic veins are
visible draining separately into the
right atrium (green arrows).
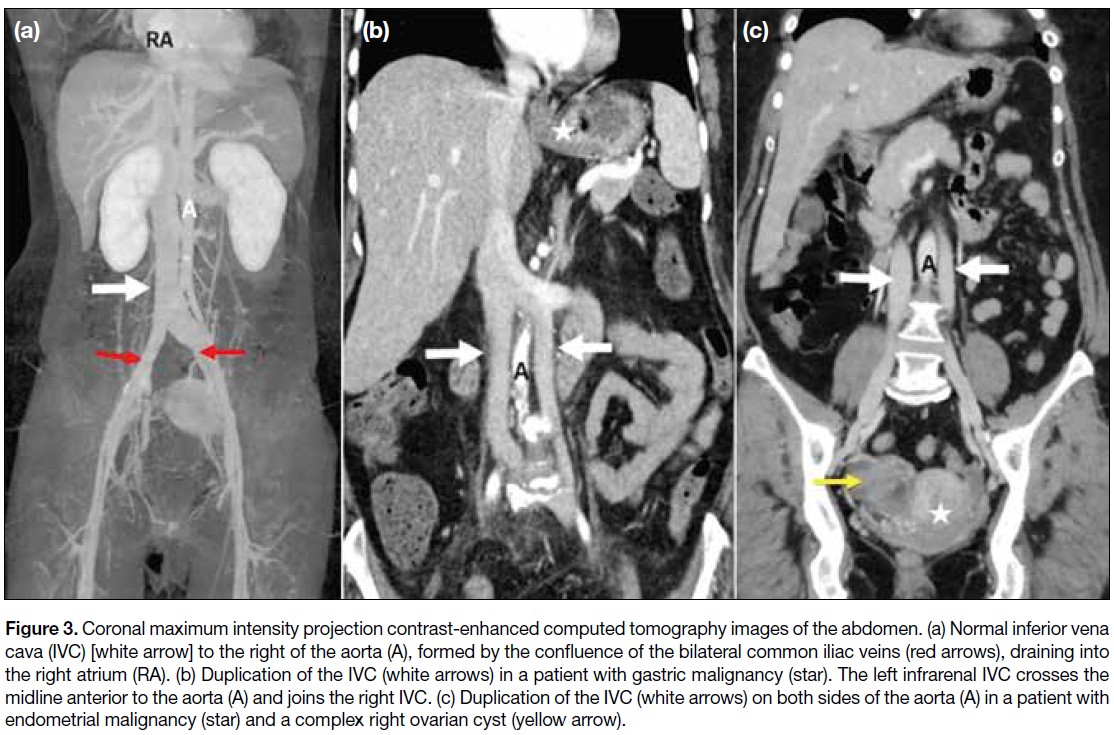
Figure 3. Coronal maximum intensity projection contrast-enhanced computed tomography images of the abdomen. (a) Normal inferior vena
cava (IVC) [white arrow] to the right of the aorta (A), formed by the confluence of the bilateral common iliac veins (red arrows), draining into
the right atrium (RA). (b) Duplication of the IVC (white arrows) in a patient with gastric malignancy (star). The left infrarenal IVC crosses the
midline anterior to the aorta (A) and joins the right IVC. (c) Duplication of the IVC (white arrows) on both sides of the aorta (A) in a patient with
endometrial malignancy (star) and a complex right ovarian cyst (yellow arrow).
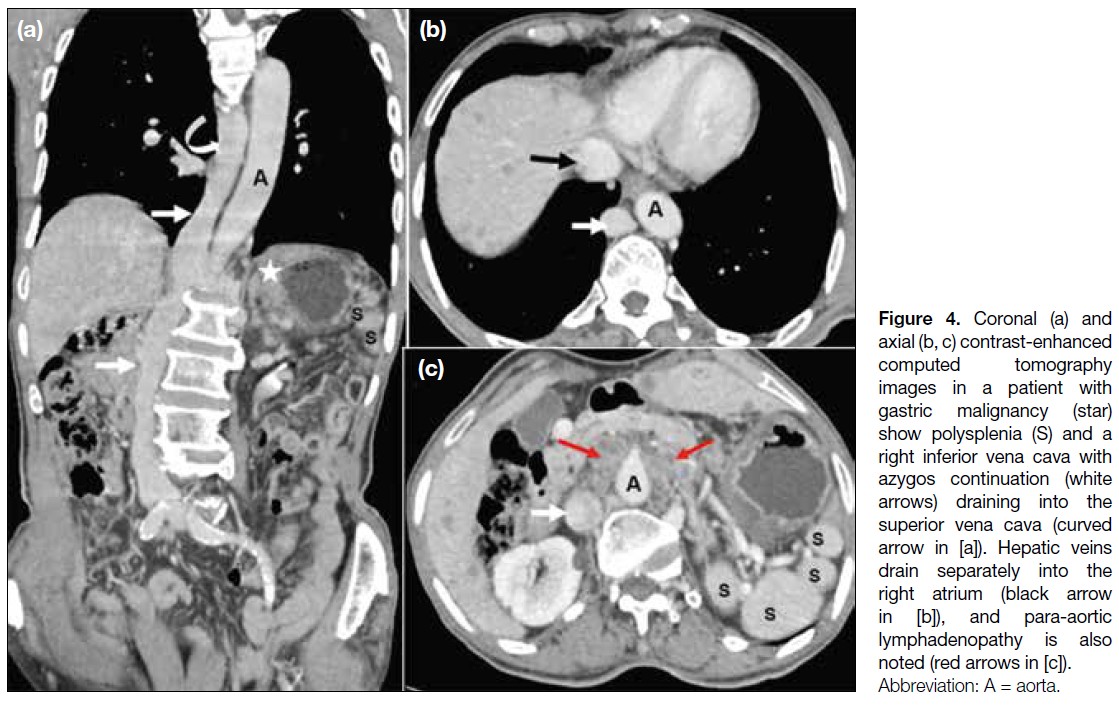
Figure 4. Coronal (a) and
axial (b, c) contrast-enhanced
computed tomography
images in a patient with
gastric malignancy (star)
show polysplenia (S) and a
right inferior vena cava with
azygos continuation (white
arrows) draining into the
superior vena cava (curved
arrow in [a]). Hepatic veins
drain separately into the
right atrium (black arrow
in [b]), and para-aortic
lymphadenopathy is also
noted (red arrows in [c]).
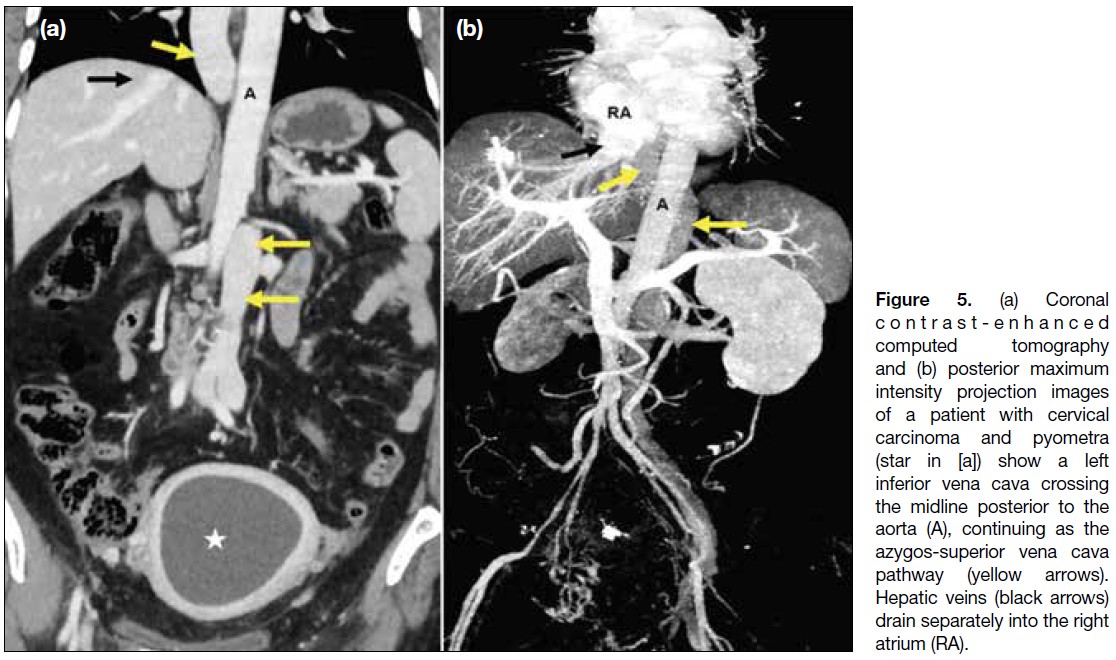
Figure 5. (a) Coronal
contrast-enhanced computed tomography
and (b) posterior maximum
intensity projection images
of a patient with cervical
carcinoma and pyometra
(star in [a]) show a left
inferior vena cava crossing
the midline posterior to the
aorta (A), continuing as the
azygos-superior vena cava
pathway (yellow arrows).
Hepatic veins (black arrows)
drain separately into the right
atrium (RA).
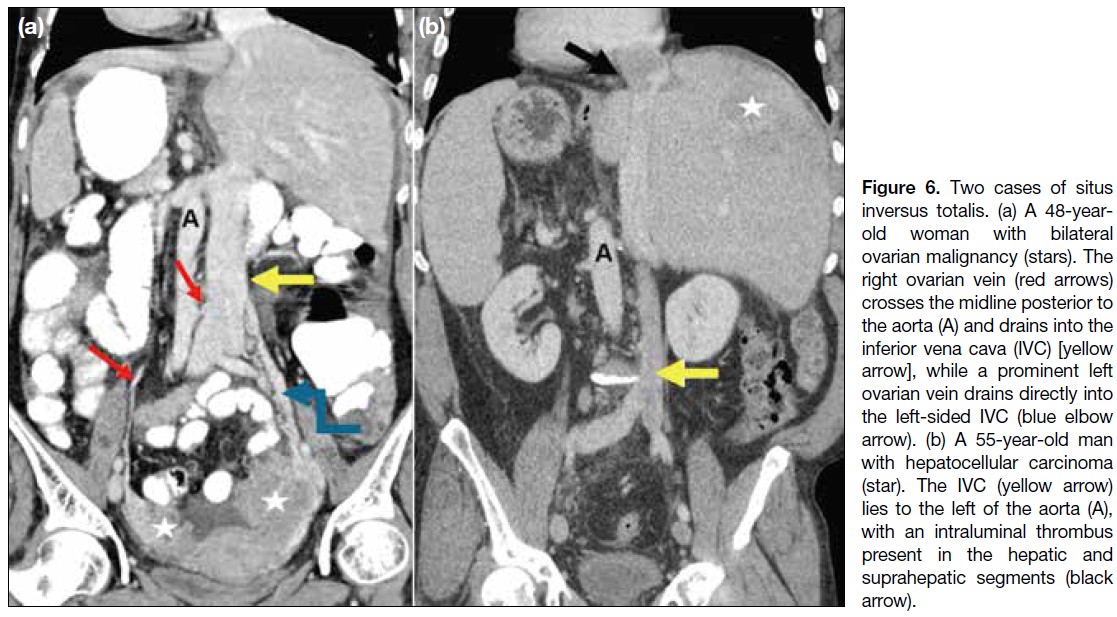
Figure 6. Two cases of situs
inversus totalis. (a) A 48-year-old
woman with bilateral
ovarian malignancy (stars). The
right ovarian vein (red arrows)
crosses the midline posterior to
the aorta (A) and drains into the
inferior vena cava (IVC) [yellow
arrow], while a prominent left
ovarian vein drains directly into
the left-sided IVC (blue elbow
arrow). (b) A 55-year-old man
with hepatocellular carcinoma
(star). The IVC (yellow arrow)
lies to the left of the aorta (A),
with an intraluminal thrombus
present in the hepatic and
suprahepatic segments (black arrow).
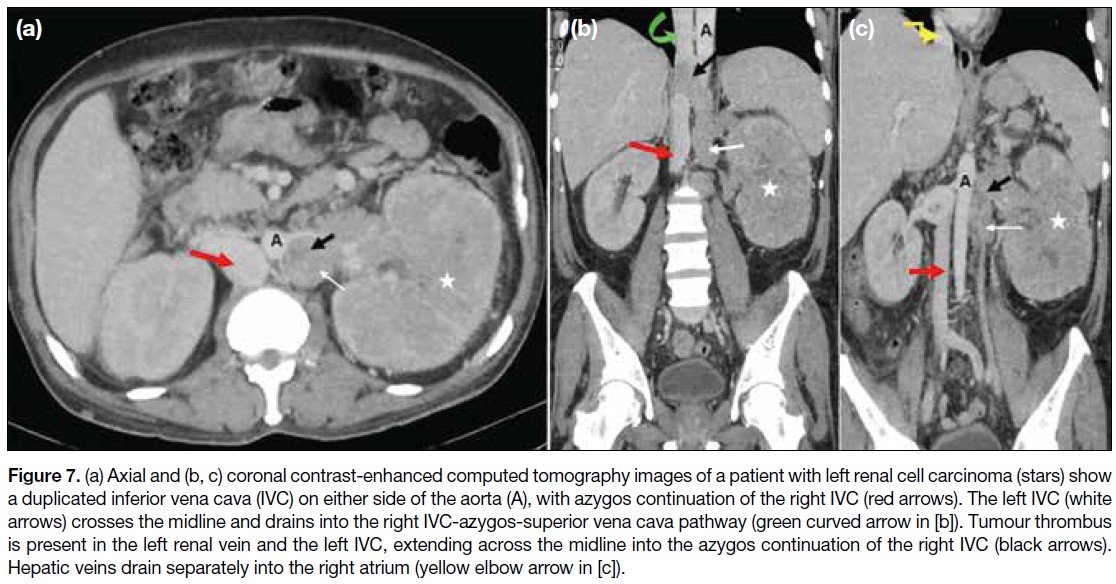
Figure 7. (a) Axial and (b, c) coronal contrast-enhanced computed tomography images of a patient with left renal cell carcinoma (stars) show
a duplicated inferior vena cava (IVC) on either side of the aorta (A), with azygos continuation of the right IVC (red arrows). The left IVC (white
arrows) crosses the midline and drains into the right IVC-azygos-superior vena cava pathway (green curved arrow in [b]). Tumour thrombus
is present in the left renal vein and the left IVC, extending across the midline into the azygos continuation of the right IVC (black arrows).
Hepatic veins drain separately into the right atrium (yellow elbow arrow in [c]).
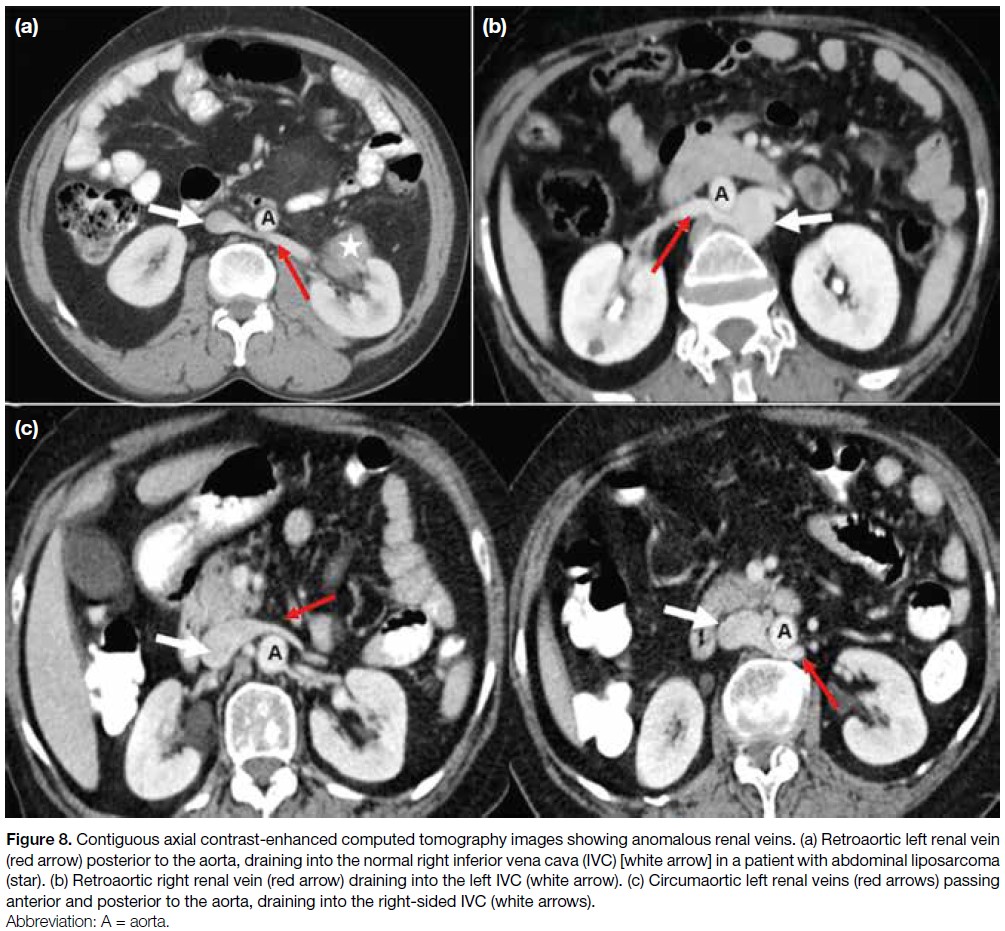
Figure 8. Contiguous axial contrast-enhanced computed tomography images showing anomalous renal veins. (a) Retroaortic left renal vein
(red arrow) posterior to the aorta, draining into the normal right inferior vena cava (IVC) [white arrow] in a patient with abdominal liposarcoma (star). (b) Retroaortic right renal vein (red arrow) draining into the left IVC (white arrow). (c) Circumaortic left renal veins (red arrows) passing anterior and posterior to the aorta, draining into the right-sided IVC (white arrows).
Figure 9. (a) Tortuous left renal vein draining into the left common iliac vein (white arrow) instead of the inferior vena cava (IVC) [black arrow].
(b) Anterior and (c) posterior views show ‘horseshoe’ kidneys (K) with vertically oriented renal veins (white arrows in [b]) and gonadal veins
(red arrows in [c]) draining into the IVC (black arrow in [b]) in a patient with endometrial malignancy (stars).
ACQUIRED PATHOLOGIES
The major acquired venous pathologies in abdominopelvic
malignancies include external compression or infiltration
of the IVC and its draining veins by neoplasms (Figures 10 and 11), metastatic lymph nodes (Figure 12), and/or
intraluminal thrombosis.
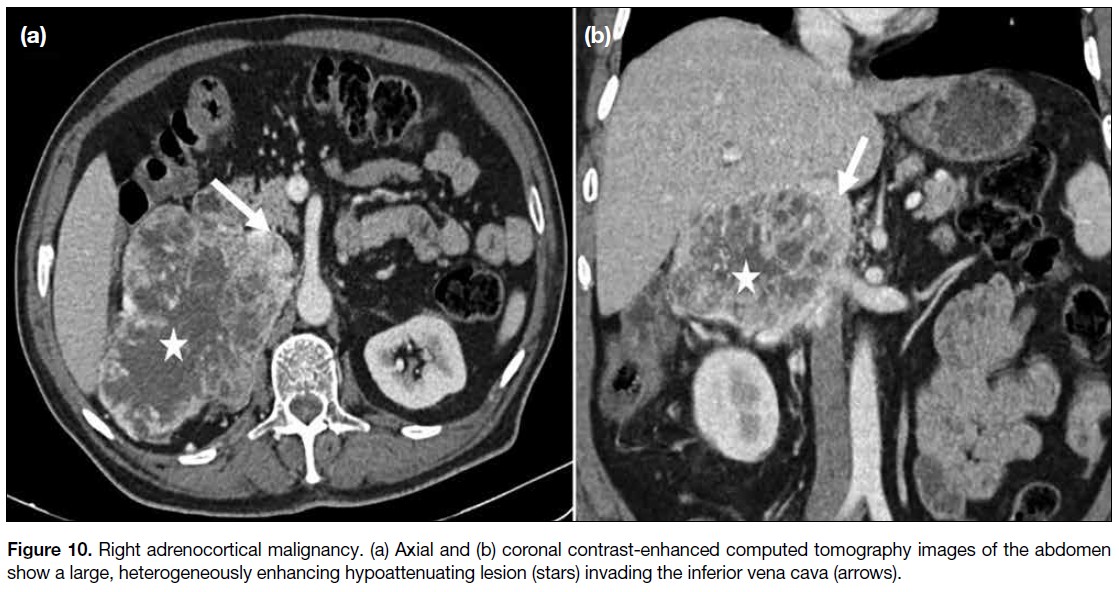
Figure 10. Right adrenocortical malignancy. (a) Axial and (b) coronal contrast-enhanced computed tomography images of the abdomen
show a large, heterogeneously enhancing hypoattenuating lesion (stars) invading the inferior vena cava (arrows).
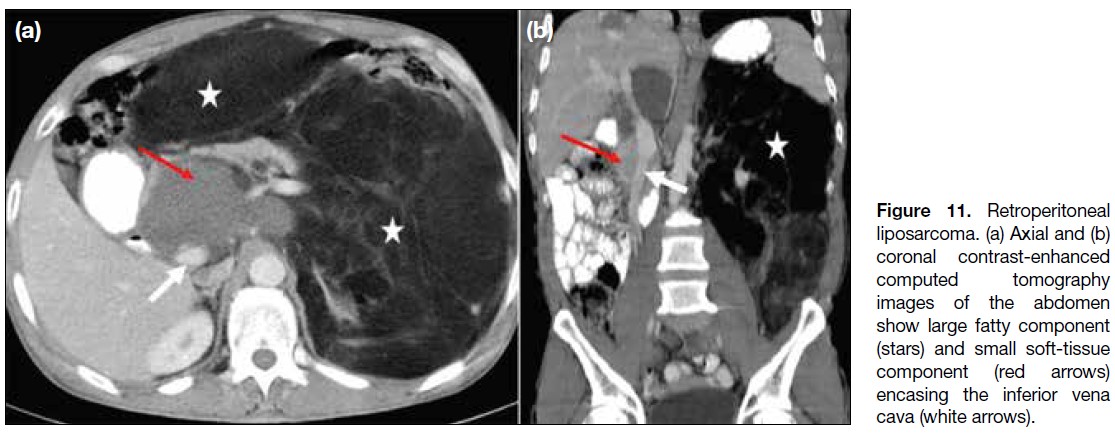
Figure 11. Retroperitoneal
liposarcoma. (a) Axial and (b)
coronal contrast-enhanced
computed tomography images of the abdomen show large fatty component (stars) and small soft-tissue component (red arrows) encasing the inferior vena cava (white arrows).
Figure 12. A 24-year-old
man with lymphoma.
(a) Axial and (b) coronal
contrast-enhanced
computed tomography
images of the abdomen
show splenomegaly (stars)
and conglomerated nodal
mass (red arrows) encasing
and causing narrowing
of the inferior vena cava
(IVC) [white arrows], aorta,
and their branches. (c,
d) Corresponding post-chemotherapy
images show
a significant decrease in the
size of the nodal mass (red
arrows) and spleen (star
in [d]), with expansion and
visualisation of the IVC (white
arrows).
Malignancies most commonly involving the IVC
include those of the liver (4.0%-5.9%), kidney (4%-10%), and adrenal glands (9%-19%).[4] [8] Although the
portal veins are more frequently involved, abnormalities
of the hepatic artery, hepatic veins, and IVC may occur
in hepatocellular carcinomas; accordingly, triphasic
computed tomography should be performed in the
evaluation of liver malignancies (Figure 13).
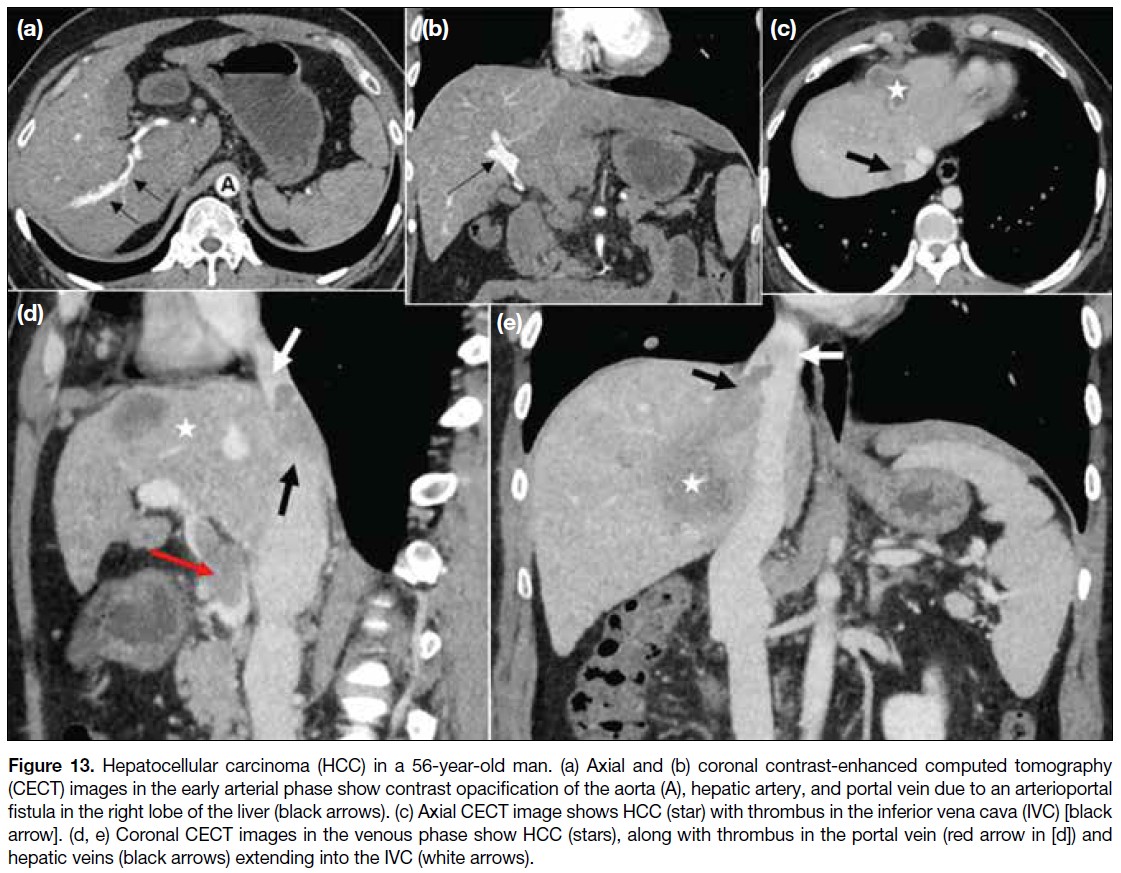
Figure 13. Hepatocellular carcinoma (HCC) in a 56-year-old man. (a) Axial and (b) coronal contrast-enhanced computed tomography
(CECT) images in the early arterial phase show contrast opacification of the aorta (A), hepatic artery, and portal vein due to an arterioportal
fistula in the right lobe of the liver (black arrows). (c) Axial CECT image shows HCC (star) with thrombus in the inferior vena cava (IVC) [black arrow]. (d, e) Coronal CECT images in the venous phase show HCC (stars), along with thrombus in the portal vein (red arrow in [d]) and
hepatic veins (black arrows) extending into the IVC (white arrows).
Cancer-associated thrombosis is recognised as the
most common complication of cancer and is attributed
to several factors (Table 2).[7] [8] [9] [10] [11] [12] Compared with the
general population, patients with cancer have a 12-fold increased risk of developing venous thrombosis, as well
as a significantly worse prognosis[9] [10] (Figure 14). The
IVC and its tributaries, especially the renal and gonadal
veins, should be assessed in all abdominal malignancies to exclude thrombosis (Figure 15). Postsurgical venous
thromboembolism is the leading cause of postoperative
death in cancer patients, and IVC thrombosis is associated
with substantial morbidity and mortality.[11] [12]
Table 2. Risk factors associated with increased incidence of thrombosis in patients with cancer. [7] [8] [9] [10] [11] [12]
Figure 14. (a) Coronal contrast-enhanced computed tomography (CECT) image of a 56-year-old man with adenocarcinoma of the stomach
shows antropyloric gastric malignancy (stars), metastatic lymph nodes (white arrows), and multiple intraluminal tumour thrombi (black
arrows) in the inferior vena cava (IVC) [red arrow], with extraluminal infiltration of the IVC by right iliac lymph nodes (yellow arrow). (b) Coronal
and (c, d) axial CECT images of a 42-year-old woman with mucinous adenocarcinoma of the stomach show diffuse thickening of the gastric
wall (stars in [b] and [c]) and widespread metastatic lymph nodes with multiple tiny calcifications (blue arrows in [b] and [c]). A focal intraluminal
thrombus in the IVC (black arrows in [b] and [c]) and a long-segment thrombus in a dilated, non-enhancing right ovarian vein (green curved
arrows in [b] and [d]) are evident. The left ovarian vein (yellow arrows in [b] and [d]) is compressed by retroperitoneal lymphadenopathy.
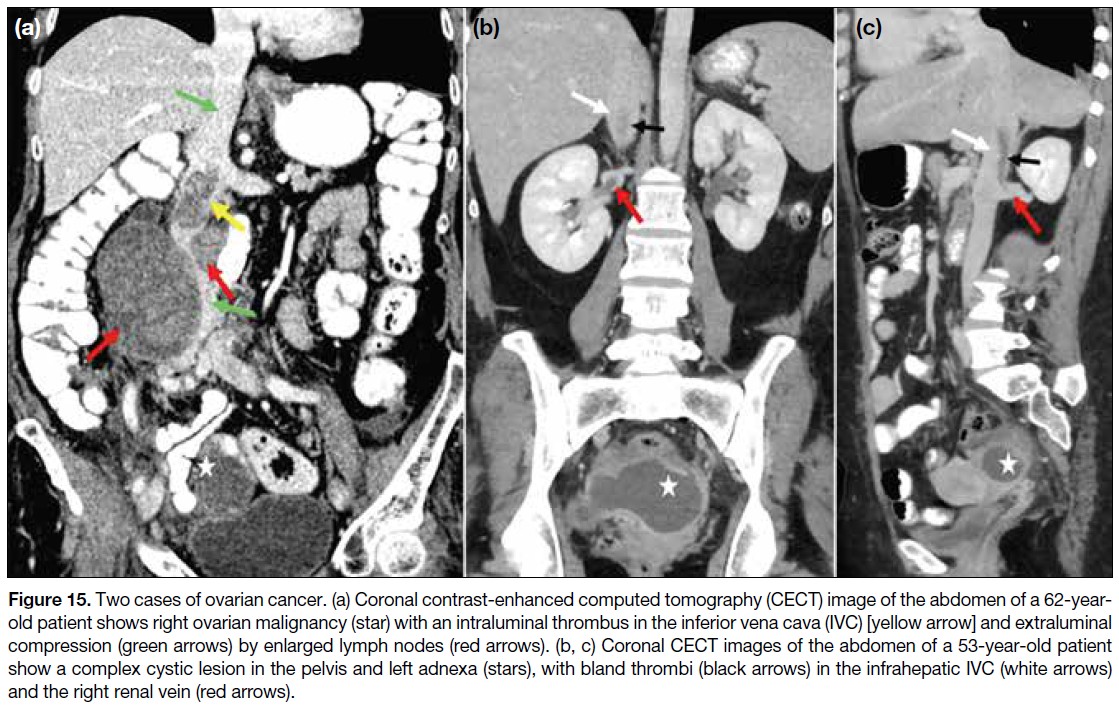
Figure 15. Two cases of ovarian cancer. (a) Coronal contrast-enhanced computed tomography (CECT) image of the abdomen of a 62-year-old
patient shows right ovarian malignancy (star) with an intraluminal thrombus in the inferior vena cava (IVC) [yellow arrow] and extraluminal
compression (green arrows) by enlarged lymph nodes (red arrows). (b, c) Coronal CECT images of the abdomen of a 53-year-old patient
show a complex cystic lesion in the pelvis and left adnexa (stars), with bland thrombi (black arrows) in the infrahepatic IVC (white arrows)
and the right renal vein (red arrows).
Tumour thrombus results either from direct extension of
the malignancy or embolisation of neoplastic cells into the
abdominal veins and/or the IVC. Differentiation between
bland and tumour thrombi is crucial for management:
anticoagulation or catheter-directed thrombolysis is
the mainstay of treatment for bland thrombus, whereas
tumour thrombus may require surgical resection (Table 3).[8] [13] [14] [15] In addition to tumour thrombectomy, adherent
tumour thrombus invading the IVC wall necessitates
en bloc excision, segmental resection, and vascular
reconstruction.[15] Magnetic resonance imaging is superior
to computed tomography in detecting and characterising
tumour thrombus, as well as in identifying vessel wall
invasion[8] (Figure 16). The extent of tumour thrombus
within the IVC and the right atrium, along with vessel
wall invasion, determines staging and resectability.
These two factors are also independent predictors of
adverse prognosis and poor survival rates in abdominal
malignancies.[7] [8]
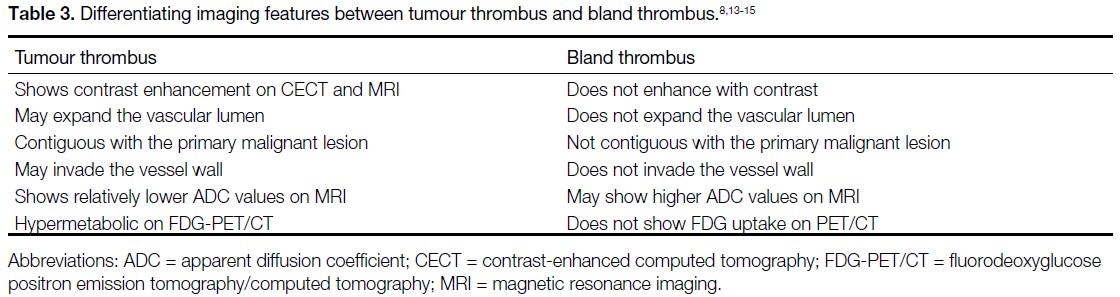
Table 3. Differentiating imaging features between tumour thrombus and bland thrombus.Table 3).[8] [13] [14] [15]
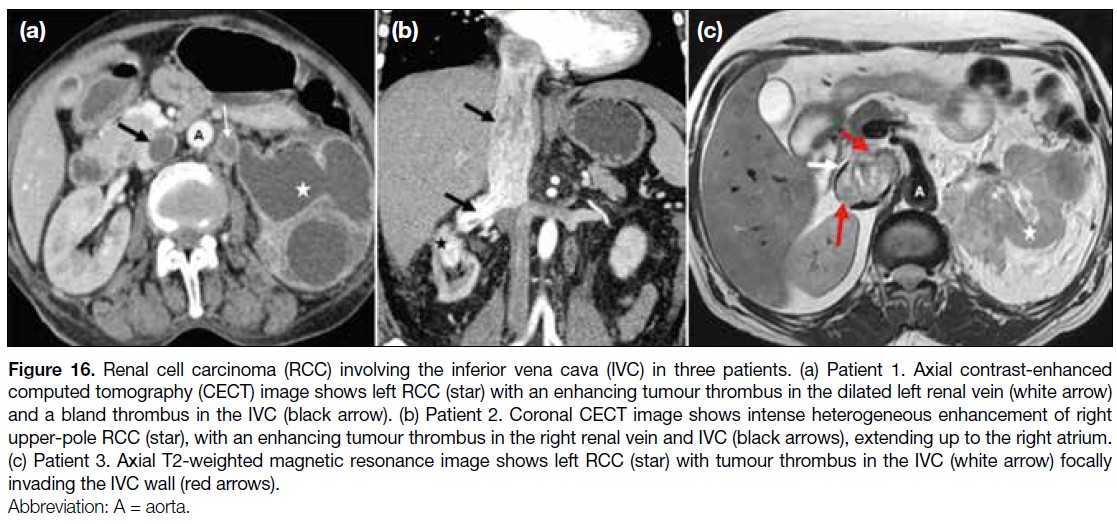
Figure 16. Renal cell carcinoma (RCC) involving the inferior vena cava (IVC) in three patients. (a) Patient 1. Axial contrast-enhanced
computed tomography (CECT) image shows left RCC (star) with an enhancing tumour thrombus in the dilated left renal vein (white arrow)
and a bland thrombus in the IVC (black arrow). (b) Patient 2. Coronal CECT image shows intense heterogeneous enhancement of right
upper-pole RCC (star), with an enhancing tumour thrombus in the right renal vein and IVC (black arrows), extending up to the right atrium.
(c) Patient 3. Axial T2-weighted magnetic resonance image shows left RCC (star) with tumour thrombus in the IVC (white arrow) focally
invading the IVC wall (red arrows).
INFERIOR VENA CAVA IN
PAEDIATRIC MALIGNANCIES
Anatomical variants in the hepatic vasculature and the
IVC should be identified before segmental resection
in hepatoblastoma (Figure 17). Retroperitoneal
malignancies in children may involve the abdominal
vasculature, including the IVC (Figure 18). Thrombosis
and vascular displacement are more common in Wilms
tumours than vessel encasement, whereas vascular
invasion occurs more frequently in neuroblastomas[4] (Figure 19).
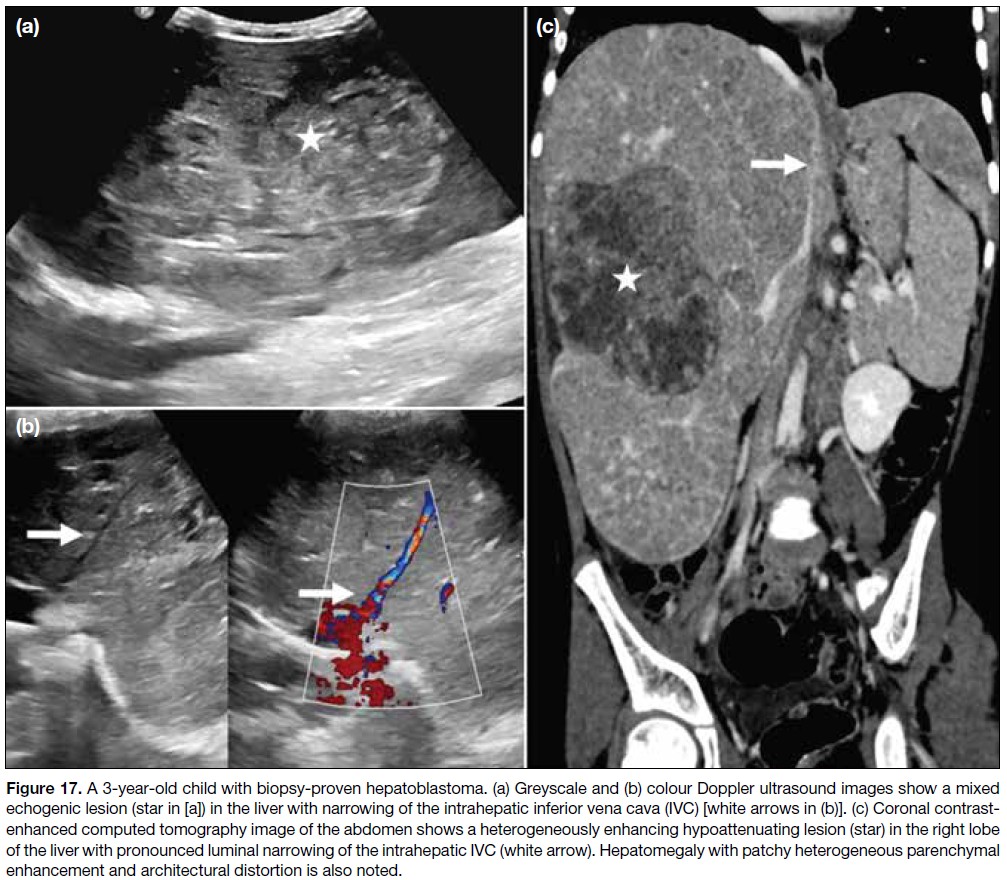
Figure 17. A 3-year-old child with biopsy-proven hepatoblastoma. (a) Greyscale and (b) colour Doppler ultrasound images show a mixed
echogenic lesion (star in [a]) in the liver with narrowing of the intrahepatic inferior vena cava (IVC) [white arrows in (b)]. (c) Coronal contrast-enhanced
computed tomography image of the abdomen shows a heterogeneously enhancing hypoattenuating lesion (star) in the right lobe
of the liver with pronounced luminal narrowing of the intrahepatic IVC (white arrow). Hepatomegaly with patchy heterogeneous parenchymal
enhancement and architectural distortion is also noted.
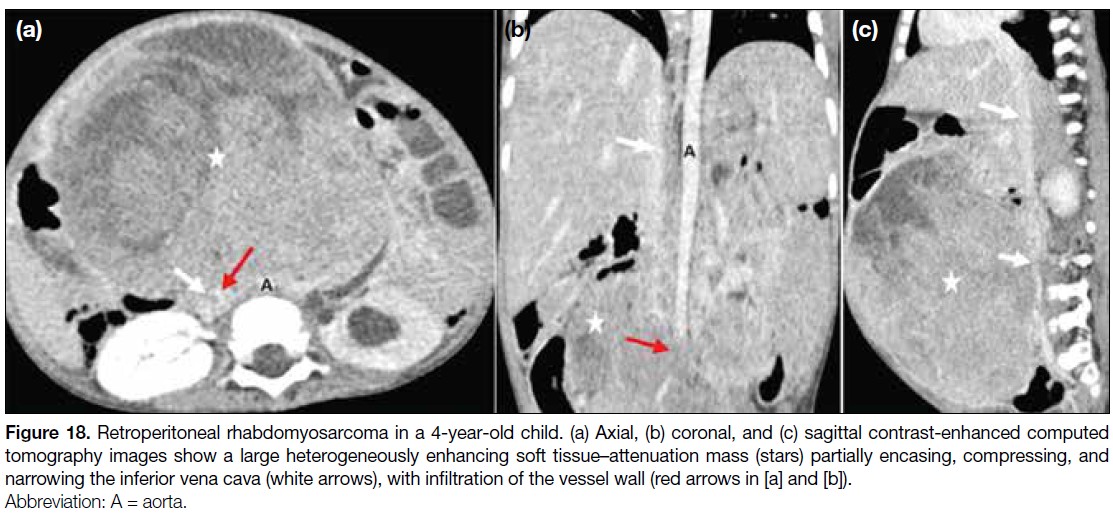
Figure 18. Retroperitoneal rhabdomyosarcoma in a 4-year-old child. (a) Axial, (b) coronal, and (c) sagittal contrast-enhanced computed
tomography images show a large heterogeneously enhancing soft tissue–attenuation mass (stars) partially encasing, compressing, and narrowing the inferior vena cava (white arrows), with infiltration of the vessel wall (red arrows in [a] and [b]).
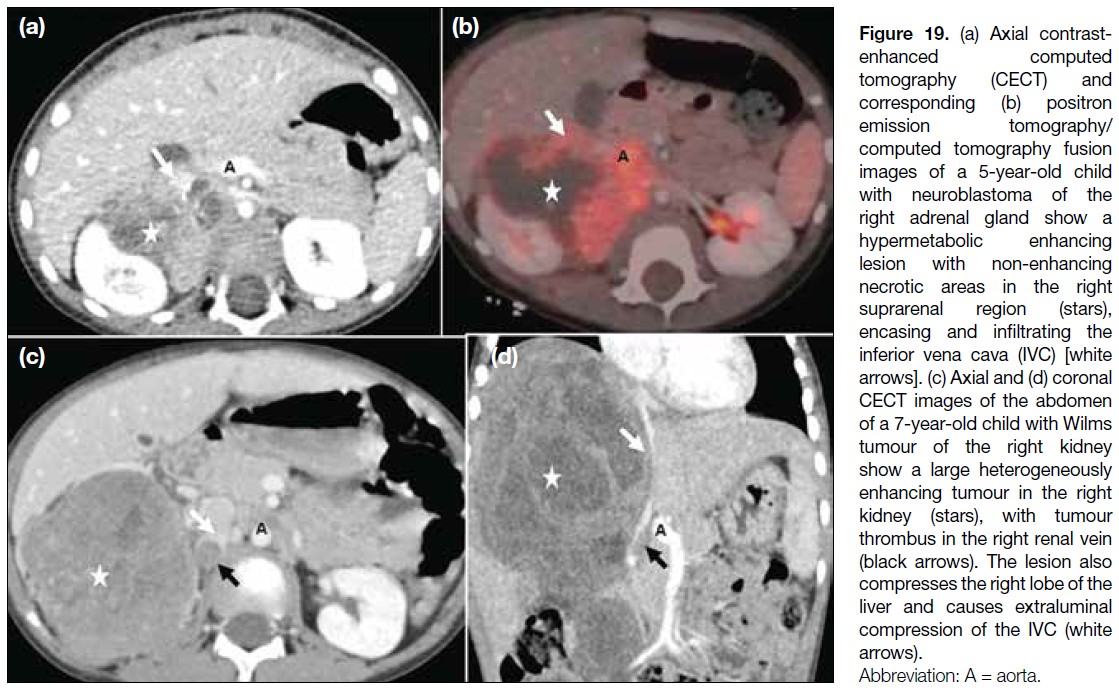
Figure 19. (a) Axial contrast-enhanced
computed tomography (CECT) and
corresponding (b) positron
emission tomography/computed tomography fusion
images of a 5-year-old child
with neuroblastoma of the
right adrenal gland show a
hypermetabolic enhancing
lesion with non-enhancing
necrotic areas in the right
suprarenal region (stars),
encasing and infiltrating the
inferior vena cava (IVC) [white
arrows]. (c) Axial and (d) coronal
CECT images of the abdomen
of a 7-year-old child with Wilms
tumour of the right kidney
show a large heterogeneously
enhancing tumour in the right
kidney (stars), with tumour
thrombus in the right renal vein
(black arrows). The lesion also
compresses the right lobe of the
liver and causes extraluminal
compression of the IVC (white arrows).
POSTSURGICAL COMPLICATIONS
Compression and narrowing of the IVC may occur
as immediate or delayed complications in patients
undergoing extensive retroperitoneal surgeries and
abdominal lymph node dissections (Figure 20).
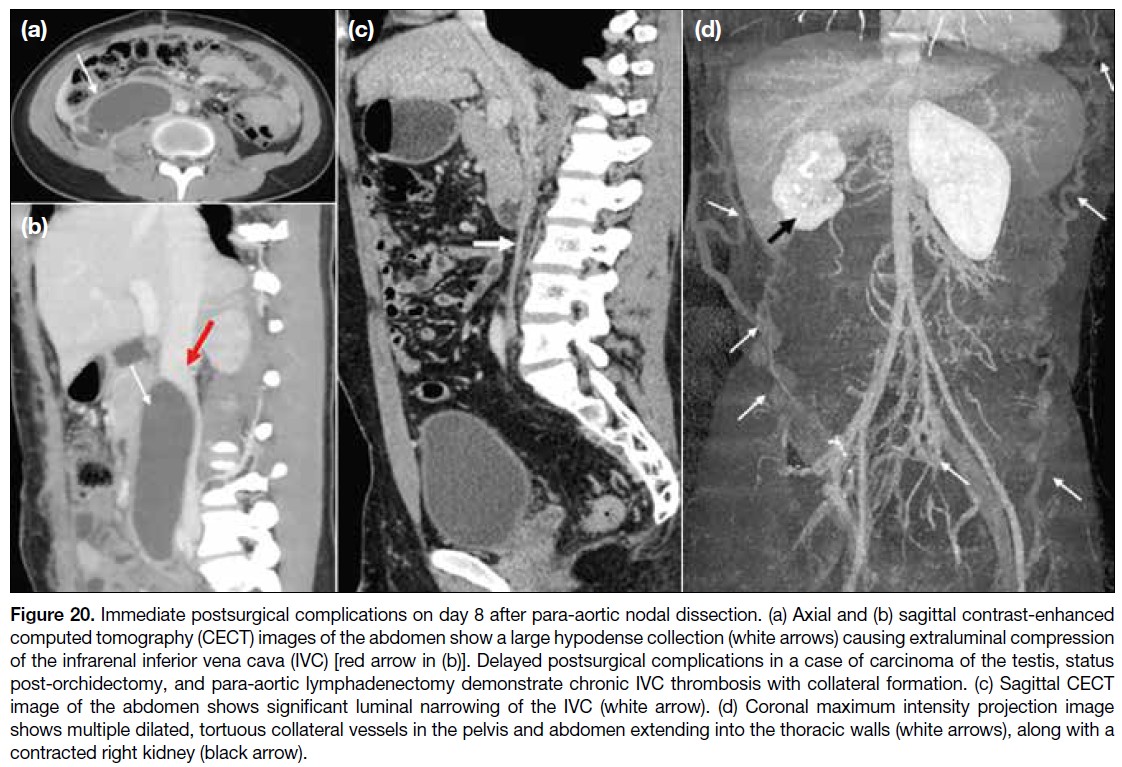
Figure 20. Immediate postsurgical complications on day 8 after para-aortic nodal dissection. (a) Axial and (b) sagittal contrast-enhanced
computed tomography (CECT) images of the abdomen show a large hypodense collection (white arrows) causing extraluminal compression
of the infrarenal inferior vena cava (IVC) [red arrow in (b)]. Delayed postsurgical complications in a case of carcinoma of the testis, status
post-orchidectomy, and para-aortic lymphadenectomy demonstrate chronic IVC thrombosis with collateral formation. (c) Sagittal CECT
image of the abdomen shows significant luminal narrowing of the IVC (white arrow). (d) Coronal maximum intensity projection image
shows multiple dilated, tortuous collateral vessels in the pelvis and abdomen extending into the thoracic walls (white arrows), along with a
contracted right kidney (black arrow).
CONCLUSION
Comprehensive evaluation of the IVC and its tributaries
is a critical component of pre-surgical imaging. Cancer-associated
thrombosis of the IVC and abdominal veins
remains underrecognised and requires a high index
of clinical suspicion due to non-specific symptoms.
Identifying abnormal drainage patterns and congenital
variations, along with recognising intrinsic or extrinsic
involvement of the IVC by abdominopelvic malignancies,
is vital before undertaking major oncological surgery.
REFERENCES
1. Eitler K, Bibok A, Telkes G. Situs inversus totalis: a clinical review. Int J Gen Med. 2022;15:2437–49.
Crossref
2. Bass JE, Redwine MD, Kramer LA, Huynh PT, Harris JH Jr. Spectrum of congenital anomalies of the inferior vena cava: cross-sectional imaging findings. Radiographics. 2000;20:639–52.
Crossref
3. Malaki M, Willis AP, Jones RG. Congenital anomalies of the inferior vena cava. Clin Radiol. 2012;67:165–71.
Crossref
4. Smillie RP, Shetty M, Boyer AC, Madrazo B, Jafri SZ. Imaging evaluation of the inferior vena cava. Radiographics. 2015;35:578–92.
Crossref
5. Onbaș O, Kantarci M, Koplay M, Olgun H, Alper F, Aydınlı B, et al. Congenital anomalies of the aorta and vena cava: 16-detector-row CT imaging findings. Diagn Interv Radiol. 2008;14:163–71.
6. Alkhouli M, Morad M, Narins CR, Raza F, Bashir R. Inferior vena
cava thrombosis. JACC Cardiovasc Interv. 2016;9:629-43
Crossref
7. Jacob T, Modgil V, Rana KK, Das S. Multiple vascular anomalies in the abdomen—a gross anatomical study. Int J Morphol. 2008;26:563–6.
Crossref
8. Li SJ, Lee J, Hall J, Sutherland TR. The inferior vena cava: anatomical variants and acquired pathologies. Insights Imaging. 2021;12:123.
Crossref
9. Mahajan A, Brunson A, Adesina O, Keegan TH, Wun T. The incidence of cancer-associated thrombosis is increasing over time. Blood Adv. 2022;6:307–20.
Crossref
10. Abdol Razak NB, Jones G, Bhandari M, Berndt MC, Metharom P. Cancer-associated thrombosis: an overview of mechanisms, risk factors, and treatment. Cancers (Basel). 2018;10:380.
Crossref
11. Agnelli G, Bolis G, Capussotti L, Scarpa RM, Tonelli F, Bonizzoni E, et al. A clinical outcome–based prospective study on venous thromboembolism after cancer surgery: the @RISTOS project. Ann Surg. 2006;243:89–95.
Crossref
12. Teter K, Schrem E, Ranganath N, Adelman M, Berger J, Sussman R, et al. Presentation and management of inferior vena cava thrombosis. Ann Vasc Surg. 2019;56:17–23.
Crossref
13. Akin O, Dixit D, Schwartz L. Bland and tumor thrombi in abdominal malignancies: magnetic resonance imaging assessment in a large oncologic patient population. Abdom Imaging. 2011;36:62–8.
Crossref
14. Catalano OA, Choy G, Zhu A, Hahn PF, Sahani DV. Differentiation of malignant thrombus from bland thrombus of the portal vein in patients with hepatocellular carcinoma: application of diffusion‑weighted MR imaging. Radiology. 2010;254:154–62.
Crossref
15. Adams LC, Ralla B, Bender YY, Bressem K, Hamm B, Busch J,
et al. Renal cell carcinoma with venous extension: prediction
of inferior vena cava wall invasion by MRI. Cancer Imaging.
2018;18:17.
Crossref