Salvaging Inadvertent Subintimal Stenting with Double-Barrel Subintimal Stenting: A Case Report
CASE REPORT
Hong Kong J Radiol 2026;29:Epub 10 March 2026
Salvaging Inadvertent Subintimal Stenting with Double-Barrel Subintimal Stenting: A Case Report
ES Lo1, SC Woo1, SKH Wong1, LF Cheng1, KM Chan2, WK Ng2
1 Department of Radiology, Princess Margaret Hospital, Hong Kong SAR, China
2 Vascular Surgery Department, Princess Margaret Hospital, Hong Kong SAR, China
Correspondence: Dr ES Lo, Department of Radiology, Princess Margaret Hospital, Hong Kong SAR, China. Email: les474@ha.org.hk
Submitted: 9 July 2025; Accepted: 29 September 2025. This version may differ from the final version when published in an issue.
Contributors: ESL, SCW and LFC designed the study. All authors acquired and analysed the data. ESL and SKHW drafted the manuscript.
All authors critically revised the manuscript for important intellectual content. All authors had full access to the data, contributed to the study,
approved the final version for publication, and take responsibility for its accuracy and integrity.
Conflicts of Interest: All authors have disclosed no conflicts of interest.
Funding/Support: This study received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Data Availability: All data generated or analysed during the present study are available from the corresponding author on reasonable request.
Ethics Approval: This study was approved by the Central Institutional Review Board of Hospital Authority, Hong Kong (Ref No.: CIRB-
2024-555-4). The patient was treated in accordance with the Declaration of Helsinki and provided written informed consent for all treatments,
procedures, and the publication of all clinical images.
Declaration: Part of this study was presented as a poster at the 18th Annual Scientific Meeting of Asia Pacific Society of Cardiovascular and Interventional Radiology, 3-5 May 2024, Bangkok, Thailand.
Supplementary Material: The supplementary material was provided by the authors and some information may not have been peer reviewed. Any opinions or recommendations discussed are solely those of the author(s) and are not endorsed by the Hong Kong College of Radiologists. The
Hong Kong College of Radiologists disclaims all liability and responsibility arising from any reliance placed on the content.
CASE PRESENTATION
A 59-year-old male patient with a history of smoking,
metabolic syndrome, ischaemic heart disease, and long-standing
peripheral arterial disease presented to our
institution in October 2022 with recurrent claudication.
He had previously undergone multiple lower limb
angioplasties and stenting procedures at various
institutions between 2018 and 2021 for recurrent in-stent
restenosis. These included an EverFlex (Medtronic,
Plymouth, [MN], US) stent in the left external iliac
artery (EIA), a Protégé (Medtronic, Plymouth, [MN],
US) stent in the left common iliac artery (CIA), an
EverFlex stent in the right EIA, a Supera (Abbott, Santa
Clara [CA], US) stent from the right common femoral
artery to the proximal superficial femoral artery (CFA-pSFA),
a Zilver (Cook Medical, Limerick, Ireland) stent in the right mid superficial femoral artery (mSFA), and
a Supera stent from the right distal superficial femoral
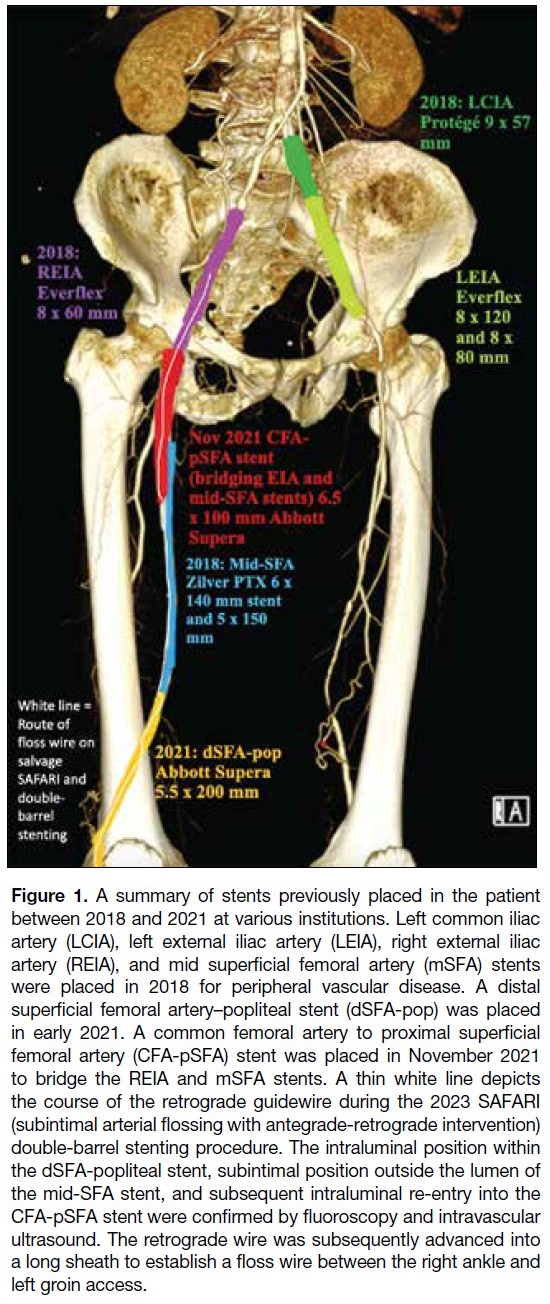
artery to the popliteal artery (dSFA-pop) [Figure 1].
Figure 1. A summary of stents previously placed in the patient
between 2018 and 2021 at various institutions. Left common iliac
artery (LCIA), left external iliac artery (LEIA), right external iliac
artery (REIA), and mid superficial femoral artery (mSFA) stents
were placed in 2018 for peripheral vascular disease. A distal
superficial femoral artery–popliteal stent (dSFA-pop) was placed
in early 2021. A common femoral artery to proximal superficial
femoral artery (CFA-pSFA) stent was placed in November 2021
to bridge the REIA and mSFA stents. A thin white line depicts
the course of the retrograde guidewire during the 2023 SAFARI
(subintimal arterial flossing with antegrade-retrograde intervention)
double-barrel stenting procedure. The intraluminal position within
the dSFA-popliteal stent, subintimal position outside the lumen of
the mid-SFA stent, and subsequent intraluminal re-entry into the
CFA-pSFA stent were confirmed by fluoroscopy and intravascular
ultrasound. The retrograde wire was subsequently advanced into
a long sheath to establish a floss wire between the right ankle and
left groin access.
The patient presented in 2022 with recurrent claudication
following placement of a bridging CFA-pSFA stent
between the right EIA and mid-SFA stents, with
claudication distance reduced to 10 metres. In view of
his recurrent symptoms, the authors were consulted
for suspected stent occlusion of the previously placed
multi-stent system. Computed tomography angiography
revealed an in-stent occlusion due to misalignment of
the CFA-pSFA and mid-SFA stents (Figure 2), likely
resulting from subintimal placement of the CFA-pSFA
stent.
Figure 2. Computed tomography
angiogram in 2022 showing stent
occlusion, likely resulting from
malalignment of the common
femoral artery to the Supera stent
(blue arrows) of the proximal
superficial femoral artery (CFA-pSFA)
and the Zilver stent (black
arrows) of the mid superficial
femoral artery (mid-SFA). The
distal margin of the CFA-pSFA
stent is seen within the subintimal
space, external to the mid-SFA
stent. (a) Axial view. (b) Sagittal
reconstruction.
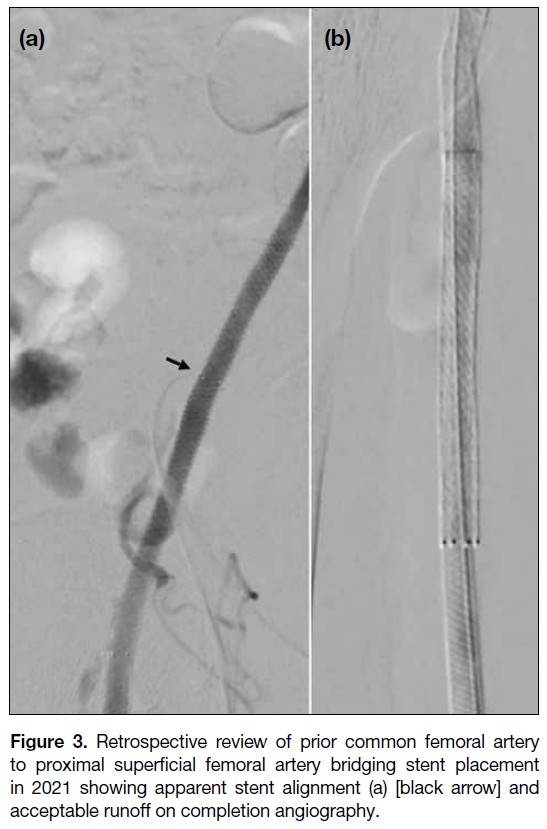
Digital subtraction angiography images in the
anteroposterior projection from the previous procedure
in November 2021 revealed apparent alignment of
the CFA-pSFA and mSFA stents, with improved
runoff post-stenting (Figure 3). Lateral views were unavailable. In view of the recurrent claudication and
the in-stent occlusion, repeat angioplasty was performed
in October 2023. Left CFA access with crossover was
performed. A 6-Fr Destination (Terumo, Somerset,
[NJ], US) long sheath was placed in the right CIA. A
Terumo (Tokyo, Japan) 0.035-inch guidewire was
advanced through the lumen of the occluded right
CFA-pSFA stent, encountering resistance (Figure 4a).
Inability to negotiate the wire into the right mid-SFA
stent led to a decision to obtain retrograde access via
the right posterior tibial artery (PTA). With the aid of
a 2.6-Fr CXI (Cook medical, Bloomington [IN], US)
microcatheter, a 0.018-inch Advantage (Terumo, Tokyo,
Japan) wire was advanced retrogradely through the PTA
and the dSFA-pop Supera stent intraluminally. The wire
was manipulated at the junction of the mid-SFA Zilver
stent and dSFA-pop Supera stent, entering the subintimal
space. After further subintimal manipulation, re-entry
of the retrograde wire into the lumen of the occluded
CFA-pSFA stent was achieved. The wire was then
advanced into the right EIA/CIA stent lumen (Figures 1 and 4b). Wire position was confirmed by intravascular
ultrasound (IVUS) [Visions PV 0.018-inch catheter
(Phillips, Rancho Cordova [CA], US)] and angiography.
The retrograde wire was externalised through the 6-Fr
crossover sheath and retrieved via the left groin access to
establish through-and-through access.
Figure 3. Retrospective review of prior common femoral artery
to proximal superficial femoral artery bridging stent placement
in 2021 showing apparent stent alignment (a) [black arrow] and
acceptable runoff on completion angiography.
Figure 4. Digital subtraction angiography images of angioplasty and double-barrel stenting performed in November 2023. (a) Crossover
wire from left femoral access, with the tip positioned within the right common femoral artery to the proximal superficial femoral artery (CFA-pSFA)
stent, encountering resistance due to occlusion. The occlusion was eventually navigated; however, in view of failure to re-enter the
mid superficial femoral artery (SFA) stent lumen, a retrograde approach was employed. (b) A 0.018-inch retrograde wire via right posterior
tibial artery access was advanced intraluminally through the occluded distal superficial femoral artery (dSFA)–pop stent, with subsequent
entry into the subintimal space outside the Zilver SFA stent and re-entry into the intraluminal occluded CFA stent. The long sheath from
the left groin access was cannulated by the retrograde wire and subsequently externalised, establishing a through-and-through floss wire.
Position was confirmed by intravascular ultrasound (Figure 6). (c) After establishment of the floss wire, angioplasty of the intraluminal-subintimal-
intraluminal wire tract was performed. (d, e) Angioplasty of the posterior tibial artery and tibioperoneal trunk was performed,
followed by mid-SFA stenting with double-barrel exclusion of the Zilver stent. Completion angiography showed significant restoration of
flow between the newly deployed mid-SFA stent and adjacent stents.
Angioplasty was performed along the wire path from the
right popliteal stent to the left EIA stent with an Armada
(Abbott, Santa Clara, [CA], US) 6 × 200 mm2 balloon,
expanding the subintimal space for subsequent stenting.
Following IVUS sizing, double-barrel subintimal
stenting was performed by deploying a Supera 5.5 × 80 mm2 stent to bridge the CFA-pSFA and dSFA-pop
stents (Figure 4c). Additional angioplasty of the newly
deployed stent, as well as the PTA and tibioperoneal trunk,
was performed with an Armada 2.5 × 200 mm2 balloon.
Completion angiography demonstrated re-establishment
of flow through the double-barrel subintimal stent, with a
patent intraluminal-subintimal-intraluminal channel and
crush exclusion of the Zilver mSFA stent (Figure 4d and e). Postoperatively, the patient resumed apixaban 5 mg twice daily and aspirin 80 mg daily.
At 1-month follow-up, symptoms improved from
claudication after walking 20 steps (Rutherford grade
III) to no claudication (Rutherford grade 0). There was
no evidence of tissue loss or vascular compromise. At
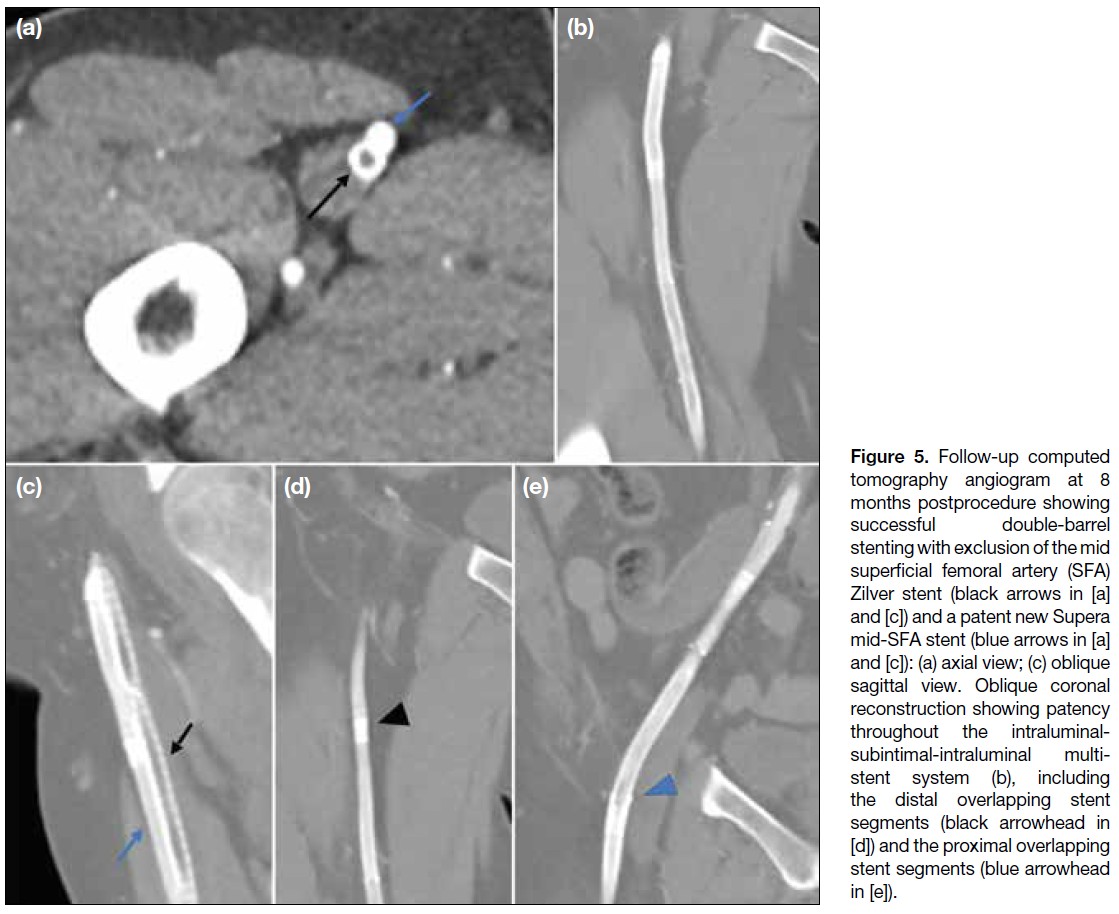
8 months, follow-up computed tomography showed
successful crush exclusion of the mid-SFA Zilver stent (Figure 5). There was complete alignment of the mid-SFA Supera stent with adjacent proximal and distal stents, with preserved patency and no significant in-stent restenosis (Figure 5). However, mild-to-moderate instent
restenosis was noted in the previously implanted
popliteal and iliac stents. The patient remains under
surveillance and is scheduled for repeat angioplasty
to preserve the patency of the multi-stent system
(supplementary Figure).
Figure 5. Follow-up computed
tomography angiogram at 8 months postprocedure showing successful double-barrel stenting with exclusion of the mid superficial femoral artery (SFA) Zilver stent (black arrows in [a] and [c]) and a patent new Supera mid-SFA stent (blue arrows in [a] and [c]): (a) axial view; (c) oblique sagittal view. Oblique coronal reconstruction showing patency throughout the intraluminal-subintimal-intraluminal multi-stent
system (b), including the distal overlapping stent segments (black arrowhead in [d]) and the proximal overlapping stent segments (blue arrowhead in [e]).
DISCUSSION
Our case highlights several important considerations for
interventionists. In retrospect, inadvertent subintimal
stent placement could have been avoided through several
measures. First, routine biplanar imaging could prevent
false assurance from a single anteroposterior projection
and detect stent misalignment. Attention should also
be paid to contrast pooling around the stent tips and
the rate of contrast runoff; delayed clearance may alert
the operator to possible distal outflow impairment
or subintimal entry. Second, careful observation of
the guidewire tip behaviour and mobility may alert
interventionists to inadvertent subintimal entry. In
cases of initial intimal dissection and subintimal entry,
the tip load of the guidewire may be exceeded with
the wire tip bending in the reverse direction and a
‘crushing’ sensation commonly reported.[1] Initial entry
into the potential subintimal space may restrict free wire
rotation. Nonetheless, where manipulation continues
and the wire tracks further into an enlarging subintimal
space, guidewire rotation may become freer, with loss
of resistance. Prolonged manipulation should be avoided
if early intraluminal re-entry fails, as this may enlarge
the subintimal space and further complicate luminal
re-entry. Third, in cases of equivocal wire position,
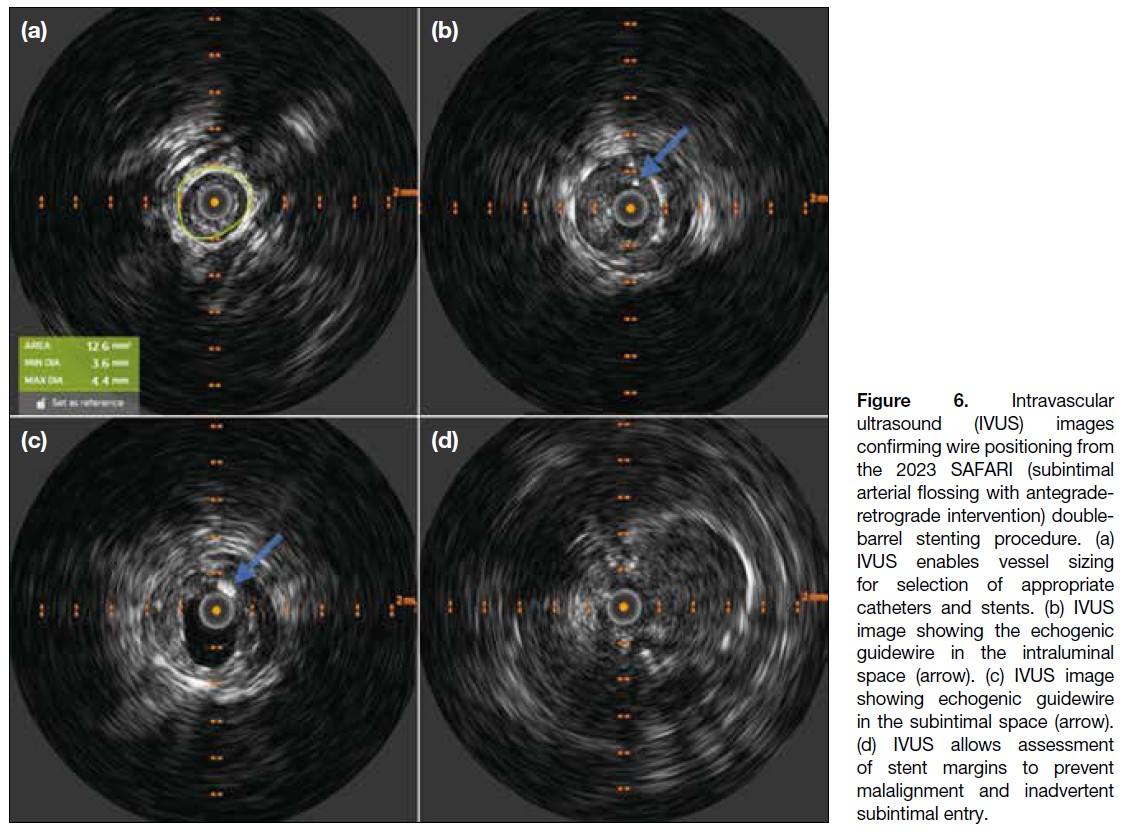
familiarity with IVUS may assist operators in accurately stenting within the true lumen. The IVUS images from
our salvage procedure are shown (Figure 6). Although
resource-intensive and operator-dependent, IVUS
enables more accurate visualisation of the vascular and
subintimal spaces with applications not only in stent
positioning but also in the accurate arterial stent sizing.[2] [3]
Figure 6. Intravascular
ultrasound (IVUS) images
confirming wire positioning from
the 2023 SAFARI (subintimal
arterial flossing with antegrade-retrograde
intervention) double-barrel
stenting procedure. (a)
IVUS enables vessel sizing
for selection of appropriate
catheters and stents. (b) IVUS
image showing the echogenic
guidewire in the intraluminal
space (arrow). (c) IVUS image
showing echogenic guidewire
in the subintimal space (arrow).
(d) IVUS allows assessment
of stent margins to prevent
malalignment and inadvertent
subintimal entry.
In cases of inadvertent subintimal entry or dissection,
achieving luminal re-entry remains a major challenge,
and familiarity with re-entry techniques is essential
for interventionists. If spontaneous re-entry cannot be
achieved with a standard wire, specific re-entry devices
(such as the Outback (Cordis, Miami Lakes [FL], US)
may be utilised. Promising data demonstrated technical
success and primary stent patency rates of up to 92.3% at
12 months with the Outback, as subintimal angioplasty
gains increasing recognition in the treatment of long-segment
TransAtlantic Inter-Society Consensus II class
C/D lesions.[4] Where such devices are unavailable,
several alternative approaches may be considered,
including retrograde access via the distal artery with
establishment of a through-and-through floss wire using the subintimal arterial flossing with antegrade-retrograde
intervention (the SAFARI [subintimal arterial flossing
with antegrade-retrograde intervention] technique[5]
as in our case), the parallel wire technique,[6] the wire
rendezvous technique with ballooning of subintimal
space as seen in CART (controlled antegrade and
retrograde subintimal tracking), reverse CART, or the
double-balloon technique.[7] [8]
In our experience, SAFARI can be performed in several
ways once luminal re-entry has been achieved. First,
a nitinol snare system may be deployed via antegrade
access to capture the retrograde wire intraluminally and
establish a through-and-through access.[9] Alternatively,
retrograde manipulation of the wire tip within the sheath
or catheter via groin access may be performed (as in our
current case).
To the best of our knowledge, cases of double-barrel
subintimal stenting are sparsely reported in the literature
and have not been reported locally. Several case reports
describe double-barrel stenting (DBS) for exclusion of occluded stents in salvage procedures for lower limb and
coronary arterial occlusions,[10] [11] [12] although this remains
an uncommonly utilised technique. One case series of
three patients with peripheral arterial disease following
DBS reported varying degrees of success, with the
longest assisted secondary patency of up to 85 months,[13]
supporting its feasibility and long-term patency.
CONCLUSION
We report a case of prior inadvertent subintimal stenting
of a bridging CFA-pSFA stent, followed by successful
salvage with subintimal DBS using the SAFARI
technique within a multi-stent system. Methods to reduce
the risk of inadvertent subintimal stenting are discussed.
Subintimal manipulation and re-entry techniques with
antegrade-retrograde approaches are also discussed as important tools for interventionists. Although not
commonly employed, DBS has been described in several
case reports and small case series. Our case affirms the
feasibility of this technique where salvage of inadvertent
subintimal stenting is necessary.
REFERENCES
1. Dash D. Guidewire crossing techniques in coronary chronic total occlusion intervention: A to Z. Indian Heart J. 2016;68:410–20.
Crossref
2. Loffroy R, Falvo N, Galland C, Fréchier L, Ledan F, Midulla M, et al. Intravascular ultrasound in the endovascular treatment of patients with peripheral arterial disease: current role and future perspectives. Front Cardiovasc Med. 2020;7:551861.
Crossref
3. Ying LH, Fan YS, Lu Y, Xu K, Li CJ. Intravascular ultrasound guided retrograde guidewire true lumen tracking technique for chronic total occlusion intervention. J Geriatr Cardiol. 2018;15:199–202.
Crossref
4. Gandini R, Fabiano S, Spano S, Volpi T, Morosetti D, Chiaravalloti A, et al. Randomized control study of the outback LTD reentry catheter versus manual reentry for the treatment of chronic total occlusions in the superficial femoral artery. Catheter Cardiovasc Interv. 2013;82:485–92.
Crossref
5. Zhuang KD, Tan SG, Tay KH. The “SAFARI” technique using retrograde access via peroneal artery access. Cardiovasc Intervent Radiol. 2012;35:927–31.
Crossref
6. Taniguchi Y, Sakakura K, Ban S, Fujita H. IVUS-assisted parallel wiring for coronary chronic total occlusion. Postępy Kardiol Interwencyjnej. 2022;18:79–80.
Crossref
7. Michael TT, Papayannis AC, Banerjee S, Brilakis ES. Subintimal dissection/reentry strategies in coronary chronic total occlusion interventions. Circ Cardiovasc Interv. 2012;5:729–38.
Crossref
8. Lee CH, Lee SW. Advancements in endovascular therapy for chronic limb-threatening ischemia: a focus on below-the-ankle interventions and wound healing strategies. J Cardiovasc Interv. 2023;2:220–31.
Crossref
9. Spinosa DJ, Harthun NL, Bissonette EA, Cage D, Leung DA, Angle JF, et al. Subintimal arterial flossing with antegrade-retrograde intervention (SAFARI) for subintimal recanalization to treat chronic critical limb ischemia. J Vasc Interv Radiol. 2005;16:37–44.
Crossref
10. Duterloo D, Lohle PN, Lampmann LE. Subintimal double-barrel restenting of an occluded primary stented superficial femoral artery. Cardiovasc Intervent Radiol. 2007;30:474–6.
Crossref
11. Somsen YB, Nap A, Henriques JP, Knaapen P. Double barrel in CTO PCI. PCRonline.com. Available from: https://www.pcronline.com/Cases-resources-images/Images-interventional-cardiology/EuroIntervention-images/2024/Double-barrel-in-CTO-PCI. Accessed 1 Jun 2025.
12. Capretti G, Mitomo S, Giglio M, Carlino M, Colombo A, Azzalini L. Subintimal crush of an occluded stent to recanalize a chronic total occlusion due to in-stent restenosis: insights from a multimodality imaging approach. JACC Cardiovasc Interv. 2017;10:e81–3.
Crossref
13. Asfoura S, Farooq I, Siddiqui W, Khatib Y. Long-term patency of double-barrel endovascular stenting for occlusive peripheral vascular disease. Available from: https://javelinjournal.org/long-term-patency-of-double-barrel-endovascular-stenting-for-occlusive-peripheral-vascular-disease/. Accessed 1 Jun 2025.